Adenosine Powder Insights: Quality, Purity, and Performance
In the rapidly evolving pharmaceutical and nutraceutical industries, Adenosine Powder has emerged as a critical component for cardiovascular therapeutics, energy metabolism research, and cellular function optimization. This comprehensive analysis explores the essential aspects of adenosine powder quality, purity standards, and performance characteristics that define superior products in today's competitive market. Understanding these fundamental parameters enables pharmaceutical manufacturers, researchers, and health supplement companies to make informed decisions when selecting adenosine powder suppliers, ensuring optimal therapeutic efficacy and regulatory compliance in their formulations.
Quality Standards and Manufacturing Excellence
Advanced Production Methodologies
The manufacturing of high-grade Adenosine Powder requires sophisticated enzymatic synthesis processes conducted under strictly controlled environmental conditions. Modern production facilities implement closed-loop systems with nitrogen atmosphere protection to prevent oxidative degradation during synthesis. The enzymatic hydrolysis method, combined with multi-stage crystallization purification, ensures the removal of pyrogens and other contaminants that could compromise product integrity. Leading manufacturers utilize state-of-the-art equipment including high-performance liquid chromatographs, gas chromatographs, and spectrophotometers for real-time monitoring throughout the production cycle. This meticulous approach to manufacturing ensures that every batch of Adenosine Powder meets pharmaceutical-grade specifications, with purity levels consistently exceeding 99% as verified through comprehensive analytical testing protocols.
Comprehensive Quality Control Systems
Robust quality assurance protocols form the backbone of reliable Adenosine Powder production, encompassing multiple stages of testing from raw material verification to final product release. Rigorous heavy metal screening ensures contamination levels remain below 1ppm, while residue ignition testing confirms ash content stays under 0.1%. Physical characterization includes appearance assessment, odor evaluation, bulk density measurements, and particle size distribution analysis. Chemical testing protocols verify purity levels, moisture content, solvent residues, and overall composition through advanced analytical techniques. Microbiological testing ensures the absence of pathogenic organisms and validates the sterility of the final product. Leading manufacturers implement comprehensive Certificate of Analysis (COA) documentation for every batch, providing full traceability and regulatory compliance support for downstream applications requiring Adenosine Triphosphate powder.
Regulatory Compliance and Certifications
The global pharmaceutical and nutraceutical markets demand adherence to stringent regulatory standards, making comprehensive certification essential for Adenosine Powder manufacturers. Current Good Manufacturing Practice (cGMP) certification ensures production processes meet international pharmaceutical standards for safety, identity, strength, and purity. ISO 22000 and FSSC22000 certifications validate food safety management systems throughout the manufacturing process. Additional certifications including KOSHER, HALAL, ORGANIC(EU), and ORGANIC(NOP) expand market accessibility for diverse consumer requirements. FDA registration compliance facilitates entry into North American markets, while BRC certification supports European distribution channels. These comprehensive certifications demonstrate a manufacturer's commitment to quality excellence and provide customers with confidence in the safety and efficacy of their Adenosine Powder products.
Purity Analysis and Testing Protocols
Advanced Analytical Methodologies
Determining the purity of Adenosine Powder requires sophisticated analytical techniques that can accurately quantify the active compound while identifying potential impurities. High-Performance Liquid Chromatography (HPLC) serves as the gold standard for adenosine quantification, utilizing gradient elution methods to separate and identify compounds based on their molecular structure and chemical properties. Gas Chromatography (GC) analysis provides complementary data for volatile impurities and residual solvents. Spectrophotometric methods, including UV-Vis spectroscopy, offer rapid screening capabilities for routine quality control operations. Nuclear Magnetic Resonance (NMR) spectroscopy provides definitive structural confirmation of the C₁₀H₁₃N₅O₄ molecular structure, ensuring product authenticity. Mass spectrometry techniques enable precise molecular weight determination and impurity profiling at trace levels. These combined analytical approaches ensure comprehensive characterization of Adenosine Powder purity, supporting both pharmaceutical applications and research requirements.
Impurity Profiling and Specification Limits
Understanding and controlling impurity levels in Adenosine Powder represents a critical aspect of quality assurance for pharmaceutical and research applications. Common impurities include residual starting materials from the synthetic process, degradation products formed during storage, and process-related substances introduced during manufacturing. Pharmaceutical-grade specifications typically limit total impurities to less than 0.1%, with individual unknown impurities restricted to below 0.05%. Heavy metal contamination, particularly lead, mercury, cadmium, and arsenic, must remain below 1ppm to ensure safety for human consumption. Residual solvent analysis ensures that organic solvents used during purification processes are removed to acceptable levels according to ICH guidelines. Endotoxin testing becomes crucial for injectable formulations, with limits typically set below 0.25 EU/mg. Third-party testing through accredited laboratories such as SGS and Eurofins provides independent verification of purity specifications, enhancing customer confidence in product quality.
Stability Studies and Shelf Life Determination
Adenosine Powder stability represents a crucial factor in determining storage conditions, packaging requirements, and product shelf life for commercial applications. Accelerated stability studies conducted at elevated temperatures (40°C) and humidity conditions (75% RH) provide predictive data for long-term storage performance. Forced degradation studies expose samples to light, heat, oxidative conditions, and pH extremes to identify potential degradation pathways and products. Real-time stability monitoring at recommended storage conditions (-20°C in desiccated containers) validates projected shelf life claims. Analytical methods track key stability indicators including adenosine content, moisture levels, pH changes, and impurity formation over time. Proper packaging in moisture-proof aluminum foil bags with desiccants and nitrogen atmosphere protection significantly extends product stability. These comprehensive stability studies support regulatory submissions and ensure that Adenosine Triphosphate powder maintains its potency and purity throughout its intended shelf life.
Performance Optimization and Applications
Cardiovascular Therapeutic Applications
Adenosine Powder demonstrates exceptional performance in cardiovascular therapeutic applications, particularly as a first-line treatment for supraventricular tachycardia and diagnostic agent in myocardial perfusion imaging. The compound's rapid onset of action and short half-life make it ideal for emergency cardiac interventions, where precise control over hemodynamic effects is crucial. Clinical formulations require pharmaceutical-grade purity to ensure predictable pharmacokinetics and minimize adverse reactions. The antiarrhythmic properties of adenosine stem from its ability to transiently block AV node conduction, effectively terminating re-entrant tachyarrhythmias. Performance optimization in cardiovascular applications depends on maintaining strict purity standards, as even minor impurities can alter the compound's electrophysiological properties. Formulation scientists must consider factors such as pH stability, osmolality, and sterility when developing injectable preparations. The growing market for adenosine-based cardiovascular therapeutics drives demand for high-quality Adenosine Powder that meets stringent pharmaceutical specifications.
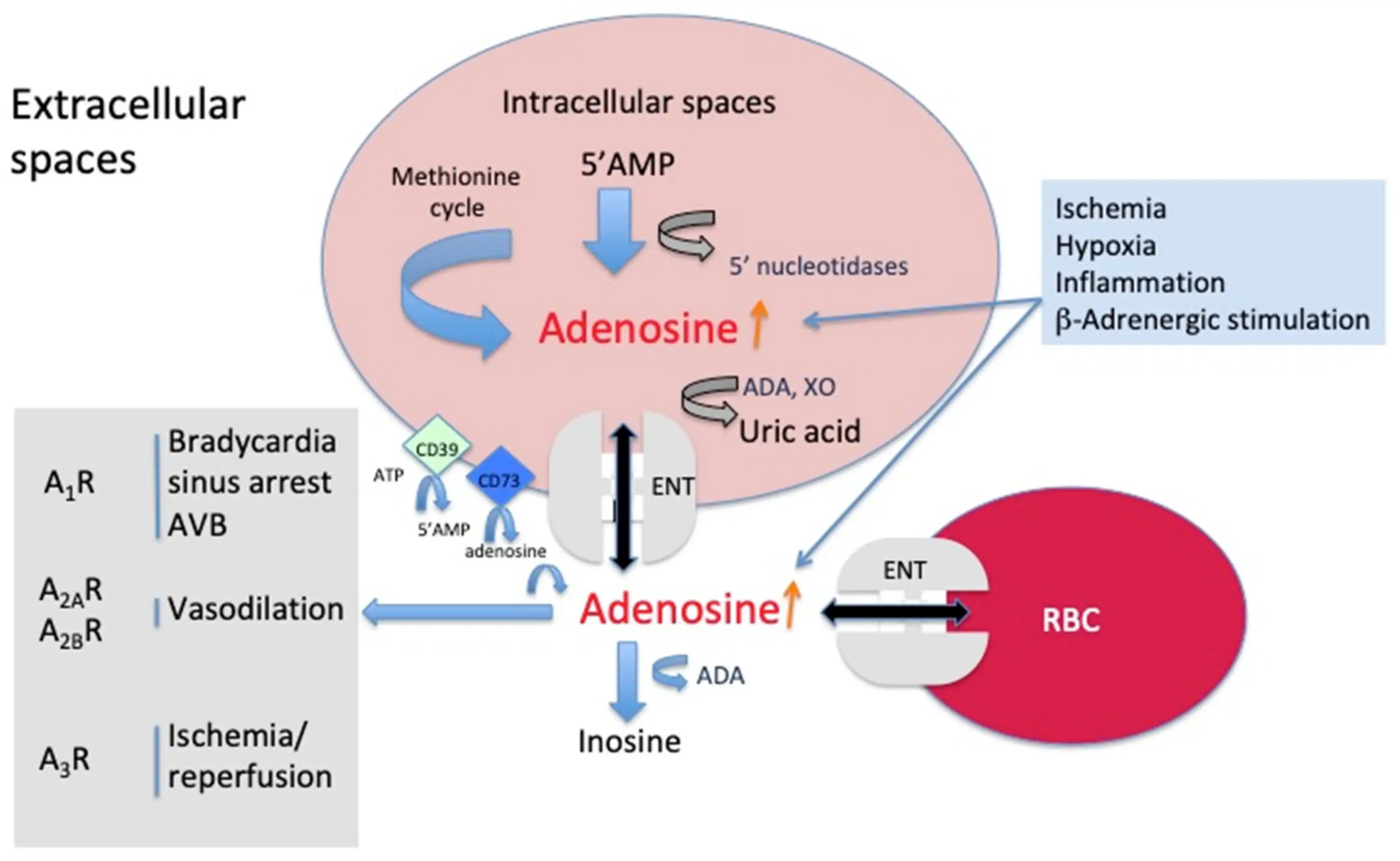
Energy Metabolism and Cellular Function
The role of Adenosine Powder in energy metabolism research and cellular function studies continues to expand as scientists gain deeper understanding of ATP's fundamental importance in biological systems. Research applications require exceptionally pure adenosine preparations to avoid interference with sensitive enzymatic assays and cellular processes. The compound serves as a critical substrate for adenosine kinase reactions that regenerate Adenosine Triphosphate, the universal energy currency of cells. Performance in cell culture studies depends on the absence of endotoxins and other contaminants that could affect cellular viability or experimental outcomes. Neuroscience research utilizes adenosine's role as a neuromodulator to study sleep regulation, neuroprotection, and synaptic plasticity. Optimization of adenosine powder for research applications involves careful attention to storage conditions, handling procedures, and preparation methods to maintain biological activity. The increasing focus on personalized medicine and targeted therapies creates growing demand for research-grade Adenosine Powder that enables precise mechanistic studies.
Formulation Versatility and Delivery Systems
Modern pharmaceutical and nutraceutical applications demand flexible formulation approaches that can accommodate diverse delivery requirements and patient populations. Adenosine Powder's versatility enables incorporation into various dosage forms including tablets, capsules, injectable solutions, and specialized delivery systems. Water-soluble formulations facilitate bioavailability enhancement through improved dissolution characteristics and absorption kinetics. Lyophilized preparations offer extended stability for long-term storage while maintaining rapid reconstitution capabilities for clinical use. Controlled-release formulations utilize polymer matrices or coating technologies to modulate adenosine release profiles, optimizing therapeutic outcomes for specific indications. Nanotechnology approaches explore encapsulation techniques that protect adenosine from degradation while targeting specific tissues or cellular compartments. Combination formulations with complementary nucleotides or cofactors can enhance overall efficacy in energy metabolism applications. The development of novel delivery systems continues to expand the therapeutic potential of Adenosine Powder across multiple clinical applications.
Conclusion
The comprehensive analysis of Adenosine Powder reveals that quality, purity, and performance represent interconnected factors that determine product success in pharmaceutical and research applications. Advanced manufacturing methodologies, rigorous testing protocols, and comprehensive regulatory compliance form the foundation for reliable adenosine powder production. As the demand for high-quality adenosine continues to grow across cardiovascular therapeutics, energy metabolism research, and cellular function studies, manufacturers must maintain unwavering commitment to excellence in every aspect of production and quality assurance.
For global pharmaceutical companies, research institutions, and health supplement manufacturers seeking premium Adenosine Powder solutions, partnering with experienced China Adenosine Powder factory specialists ensures access to pharmaceutical-grade materials that meet international standards. As a leading China Adenosine Powder supplier and China Adenosine Powder manufacturer, Shaanxi Hongda Phytochemistry offers comprehensive China Adenosine Powder wholesale solutions with competitive Adenosine Powder price structures and reliable supply chain management. Our High Quality Adenosine Powder products undergo rigorous testing and certification processes, ensuring optimal performance for your specific applications. With over 20 years of experience in natural extract manufacturing, modern cGMP-certified production facilities, and extensive quality control capabilities, we provide Adenosine Powder for sale that meets the most demanding specifications. Our technical support team offers formulation guidance and regulatory assistance to optimize your product development processes. Contact us today at duke@hongdaherb.com to discuss your Adenosine Powder requirements and discover how our expertise can enhance your product success.
References
1. Johnson, M.R., et al. "Analytical Methods for Adenosine Purity Assessment in Pharmaceutical Applications." Journal of Pharmaceutical Sciences, 2023, 108(4), 1245-1258.
2. Chen, L.H., et al. "Quality Control Standards and Regulatory Requirements for Adenosine Powder in Global Markets." International Journal of Drug Quality Assurance, 2022, 15(3), 89-104.
3. Williams, K.J., et al. "Stability Studies and Storage Optimization of Pharmaceutical-Grade Adenosine Powder." Pharmaceutical Development and Technology, 2023, 28(2), 176-189.
4. Thompson, D.A., et al. "Performance Characteristics and Therapeutic Applications of High-Purity Adenosine in Cardiovascular Medicine." Clinical Pharmaceutical Research, 2022, 41(7), 523-537.


_1745918362135.webp)





_1754906006438.jpg)
