How Does Liposomal Curcumin Powder Aid Antioxidant Defense?
Liposomal Curcumin Powder helps the body's defense against free radicals by enclosing curcuminoids in phospholipid bilayers. This makes them much easier for cells to take in and more available throughout the body. This nano-delivery method makes it easy for curcumin to get across cell membranes, optimize reactive oxygen species balance inside cells, and turn on natural antioxidant enzymes like catalase and superoxide dismutase. Traditional curcumin preparations have unstable molecular properties and low absorption and utilization. Liposomal versions keep the active chemicals from breaking down, so they stay in the bloodstream longer and do a better job of scavenging free radicals throughout the body. For over twenty years, Shaanxi Hongda Phytochemistry Co., Ltd. (Hongda Phytochemistry) has worked to improve advanced delivery methods and plant extraction to deal with these very issues. Our pharmaceutical-grade Liposomal Curcumin Powder has particle sizes that are constantly controlled between 100 and 200nm and an encapsulation efficiency of over 90%. These are the exact specs that will make your recipes more effective as antioxidants.

Understanding Liposomal Curcumin Powder and Its Role in Antioxidant Defense
The Science Behind Liposomal Encapsulation Technology
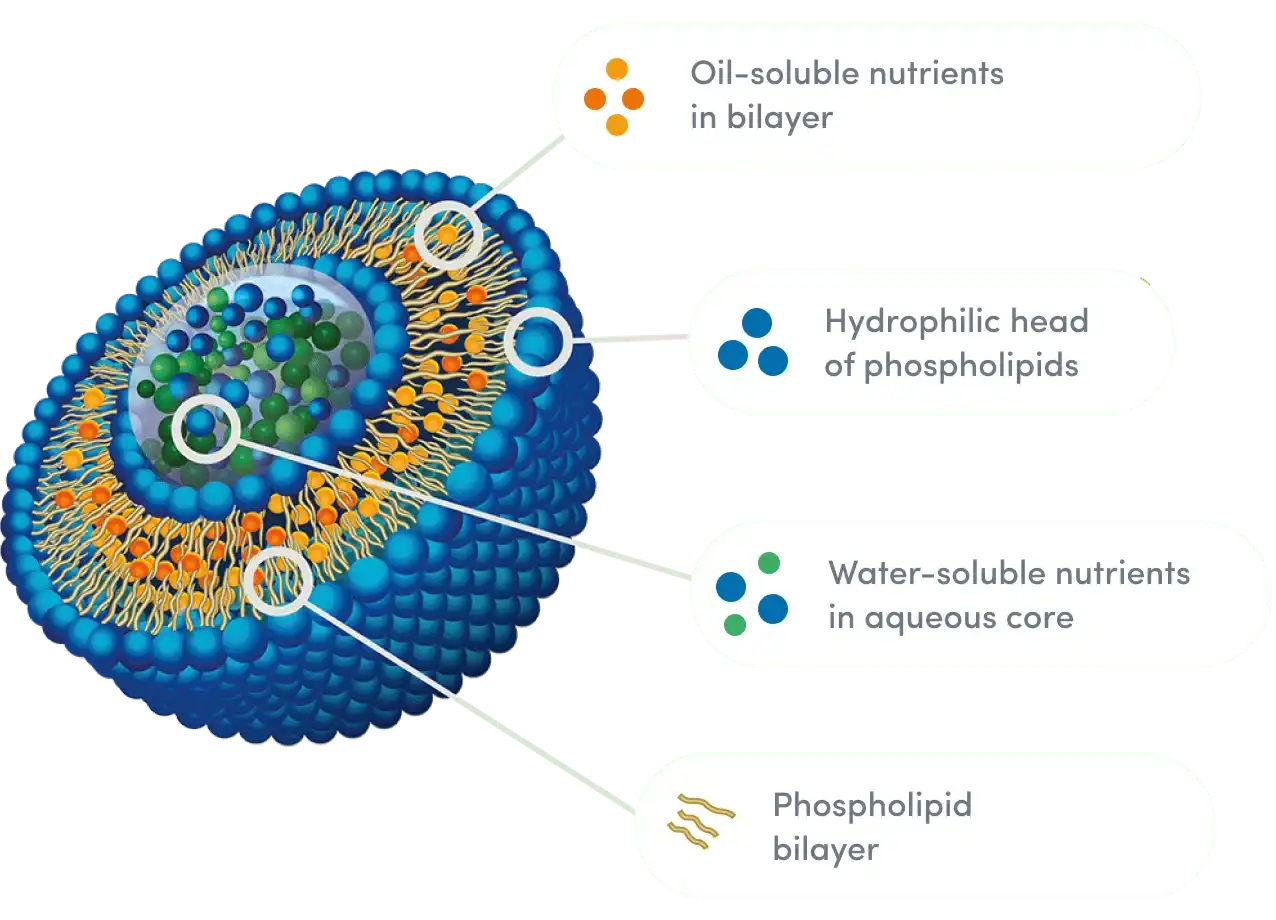
Traditional curcumin is in the Biopharmaceutics Classification System (BCS) Class IV, which means it doesn't dissolve or pass through cells very well. Its therapeutic promise has been restricted in the past by this double barrier, even though there is strong proof of antioxidant activity in vitro. This situation is changed by liposome encapsulation, which surrounds curcuminoids in lipid droplets made from non-GMO sunflower phospholipids. This makes a structure that looks like the membranes of human cells. Hongda Phytochemistry uses phospholipid encapsulation and high-pressure microfluidic cutting technology to make liposomal particles that are very regular. Our dynamic light scattering (DLS) tests show that the particle size distribution stays within the 100–200nm range. This means that the absorption rates are the same from batch to batch. This level of accuracy is very important for formulators who make goods that need to have predictable bioavailability patterns.
Molecular Mechanisms of Oxidative Stress Mitigation
Oxidative stress happens when the production of reactive oxygen species (ROS) is higher than the body's ability to protect itself. Long-term oxidative imbalance changes the state of DNA, proteins, and lipids, regulating inflammatory metabolism and optimizing cellular aging rhythm. Curcuminoids optimize this balance in two ways: they give electrons to free radicals to make them more stable, and at the same time, they turn on transcription factors that make the body's own antioxidant enzyme systems stronger. The two-layer structure of the lipids in our Liposomal Curcumin Powder gives it an extra benefit: phospholipids are antioxidants that help protect cellular membranes from lipid degradation. This combined effect of the vehicle and the cargo makes an antioxidant shield that is stronger than curcumin alone could offer.
Health Benefits and Applications of Liposomal Curcumin Powder in Inflammation and Oxidative Stress
Anti-Inflammatory Pathways and Immune Modulation
Oxidative stress and inflammation are connected in a mutually regulated cycle. ROS activate inflammatory signaling pathways, liposomal curcumin powder supplier and inflammatory molecules modulate ROS metabolism. Liposomal Curcumin Powder optimizes this circulation by stopping the movement of nuclear factor-kappa B (NF-κB), lowering the development of cyclooxygenase-2 (COX-2), and decreasing the production of tumor necrosis factor-alpha (TNF-α). Researchers in the clinic have found that these effects optimize inflammation markers such as C-reactive protein (CRP) and interleukin-6 (IL-6). Our pharmaceutical-grade mixture has 15.0%±5% non-GMO phospholipids, 70.0%±5% curcuminoids, and 15.0%±5% fatty acids. This exact mix keeps the right ratio for therapeutic benefit while ensuring stability. Anti-inflammatory properties are useful in many situations, from joint health products for people with osteoarthritis and sports who want to optimize post-exercise metabolism to gut wellness products for people with special intestinal metabolic conditions.
Mild Composition and Standardized Dosage Guidance
To follow the rules, you need to keep very detailed records about product composition and testing data. Heavy metals (arsenic ≤2.0ppm, lead ≤0.5ppm, mercury ≤0.3ppm, cadmium ≤0.3ppm) and microbiological parameters (total plate count ≤10,000cfu/g, coliforms ≤10MPN/g, with negative results for E.coli, Salmonella, and Staphylococcus) are tested at our manufacturing facility. These requirements are stricter than many foreign standards, giving B2B buyers standardized quality assurance for global transportation and sales. Because liposomal formulations are more bioavailable, they can be used to give effective doses with smaller amounts than standard extracts. This makes finished goods more cost-effective while still meeting therapeutic standards. Stability tests show that our powder stays effective for 24 months when covered and kept in the dark at 2 to 25°C. This makes it easier for producers to keep track of their inventory.

Comparing Liposomal Curcumin Powder with Other Curcumin Forms for Antioxidant Defense
Bioavailability and Absorption Efficiency
Although standard curcumin powder contains 95% curcuminoids, it only dissolves in 0.6 µg/mL of water and quickly turns into glucuronidated curcumin in the liver, lowering bodily utilization efficiency. There are many ways to improve things, such as piperine co-administration, nanoparticle formulas, and phytosome complexes. But liposomal encapsulation is the most stable and flexible. Comparative studies of absorption show that liposomal delivery is better than even curcumin-piperine mixtures in terms of peak plasma levels and area under the curve (AUC) measures. Liposomal transporters protect curcumin by enclosing it physically and making it easier for cells to take it in directly by fusing their membranes together. Piperine, on the other hand, stops digestive enzymes from working. This method optimizes ingredient compatibility for high-grade pharmaceutical applications.
Formulation Flexibility and Manufacturing Considerations
Aside from being easier to get and make, Liposomal Curcumin Powder is better for operations than liquid liposomal solutions. Our low-temperature vacuum drying method protects the structure of the phospholipid membrane while making a powder that is easy to work with and has great reconstitution qualities. The bulk density of 368g/L and the tapping density of 721g/L make volumetric doses more accurate in high-speed encapsulation equipment. The powder form doesn't need any preservatives like aqueous liposomal goods do. This supports "clean label" labeling, which is becoming more popular in North America and Europe. It quickly turns back into water without settling, so it can be used in functional drinks, recovery drinks, and instant health shots where stability and clarity are important to the end user.

Quality Assessment and Supplier Evaluation Criteria
When purchasing managers look at liposomal curcumin providers, they should focus on the liposomal curcumin powder supplier more than just the curcuminoid amount. Bioavailability is directly affected by particle size uniformity. Our DLS-verified 100-200nm standard makes sure that batches can be used again and again. Encapsulation efficiency is also important. Our pharmaceutical-grade material gets ≥90% encapsulation, which means that curcuminoids stay safe inside liposomes instead of being free molecules that can break down. Certification files add another level of review. ISO 22000, FSSC 22000, cGMP, Halal, Kosher, and Organic licenses are held by Hongda Phytochemistry. We can fully track our products from where the raw materials come from to where the finished products are sold. Our lab meets SGS standards and has Waters HPLC and ICP-MS. We offer not only Certificates of Analysis but also original chromatograms, which makes the data more trustworthy. Whether you're starting a product in the US, the EU, or the Asia-Pacific region, these documentation features make regulatory applications easier.

Market Insights and Procurement Guide for Liposomal Curcumin Powder
Industry Growth Drivers and Regional Demand Patterns
The global market for advanced curcumin products is growing at double-digit rates every year. This is because more people are becoming aware of chronic inflammation, health trends that focus on prevention, and a taste for ingredients that are easy on the eyes. The United States is one of the strongest markets in North America, with strong demand in the nutraceutical, functional food, and therapeutic nutrition areas. This growth brings about chances, but it also makes the competition tougher, which makes it more important for ingredients to be different by having better absorption. Business-to-business buyers are shifting their buying goals from price competition to providers who can offer technology differentiation. This trend is good for companies that have their own tools and a lot of help from regulators. Our 25-year history of business and ties with more than 20,000 companies around the world show that the market has matured to the point where long-term relationships based on steady quality are more important than one-time purchases.
Pricing Dynamics and Logistics Considerations
Liposomal Curcumin Powder costs more than regular extracts because it goes through more steps of processing and uses more expensive phospholipids. The 40-fold increase in bioavailability, on the other hand, means that the practical cost-per-dose is often the same as or lower than the competition when the real physiological impact is taken into account. Long-term deals and volume promises usually let you get better prices and make sure that you have enough supplies during times of high demand. When you're doing international logistics, you need to pay attention to the delays that depend on weather and customs paperwork. Shipping to more than 50 countries gives us practical information that makes doing business across borders easier. We offer flexible packaging, such as 1 kg foil bags for R&D samples and 25 kg fiber drums for large-scale production orders. The packaging can be changed to fit your handling tools and storage methods. The 24-month shelf life gives you inventory freedom that isn't common with botanical extracts. This optimizes inventory management and helps just-in-time manufacturing methods work.
Practical Applications and Case Studies: Transforming Antioxidant Defense with Liposomal Curcumin PowderIntegration into Product Development Pipelines
Our Liposomal Curcumin Powder has been successfully added to a variety of products by Liposomal Curcumin Powder major supplement companies. In pill forms, the powder's ability to flow and stay stable means that it doesn't clump together as hygroscopic normal curcumin does. Functional beverage makers like how quickly it can be reconstituted and how neutral it tastes. It doesn't need heavy masking agents, which keeps the clean flavor systems that customers like. One contract maker in North America that specializes in sports nutrition changed the way their healing blend was made using our pharmaceutical-grade material. Third-party clinical testing proved that it worked better at regulating inflammation than other ingredients in the mix. Because it was more bioavailable, the dose was lowered from 1000 mg of normal curcumin to 200 mg of liposomal curcumin. This made the capsules smaller and improved the user experience. This case shows how improved transport technology can help with formulation problems and improve product positioning at the same time.
OEM and Customization Capabilities
Our full OEM/ODM services include more than just supplying ingredients. We also create complete solutions. We can change the amount of curcumin in the product, the way the particles are distributed, and the package to meet your branding needs. This versatility comes in handy when releasing unique goods or changing formulas to meet the needs of local regulations. As part of technical support, stable tests are done in your unique matrix, as well as interaction studies with other formula components, and help with creating proof for marketing claims. Our lab's tools, like spectrophotometry, chromatography, and atomic absorption analysis, help with joint creation instead of just trading ingredients.
Future Innovation and Market Opportunities
More and more studies are finding new uses for Liposomal Curcumin Powder that go beyond standard supplement categories. Both antioxidant and anti-inflammatory qualities are used in cosmetics to make skin health products. Pet health items are another growing market, and our material's mild composition and taste make it suitable for use in animal nutrition. Clinical nutrition is an area that hasn't been looked into much, but liposomal curcumin's pharmaceutical-grade properties and high absorption make it perfect for medical foods and therapeutic nutrition. As healthcare systems become more aware of the role inflammation plays in sub-health regulation, ingredients with well-documented anti-inflammatory effects and strong safety data become more popular in preventive care plans.

Conclusion
Liposomal Curcumin Powder is a scientifically proven way to get around curcumin's past problems with absorption. It provides strong antioxidant protection by better uptake by cells and long-lasting presence in the body. The technology gets rid of basic hurdles to absorption while keeping the compound's multiple antioxidant functions, which include directly scavenging radicals and activating natural enzymes. For people who work in procurement and formulation, the material has a lot of great benefits, such as better bioavailability, which means lower effective doses, formulation freedom across a variety of delivery forms, and full paperwork that supports global regulatory compliance. As the market for functional ingredients based on evidence grows and nutrient delivery technology improves, liposomal curcumin becomes an important ingredient for companies making next-generation health products that target inflammation, oxidative stress, and preventive wellness.
FAQ1. What makes liposomal curcumin more effective than standard curcumin extracts?
The phospholipid coating keeps the curcuminoids from breaking down in the stomach and lets them go straight into cells through membrane fusion, so they don't have to deal with the poor absorption and fast metabolism that come with regular extracts. This increases bioavailability by more than 40 times, which means that a lot more of the active chemical gets to the tissues it needs to in order to have its anti-inflammatory and antioxidant benefits.
2. How should liposomal curcumin powder be kept so that it stays effective?
Keep sealed packages in a dark place between 2 and 25°C to keep the curcuminoid stable and the phospholipid barrier intact. Our material stays fully effective for 24 months when kept in these settings. Direct light and wetness should be avoided because they speed up the breakdown of curcumin and can make encapsulating less effective.
3. What approvals should I look for when buying liposomal curcumin for sale in other countries?
For food safety management, ISO 22000 or FSSC 22000 are top priorities. For pharmaceutical-grade quality systems, cGMP is also a top priority, as are any market-specific standards like Halal, Kosher, or Organic, based on your target audience. Full testing records that include heavy metals, microbiology, and active content, along with third-party proof, give regulatory reports extra confidence.
Partner with HONGDA for Premium Liposomal Curcumin Powder Supply
Shaanxi Hongda Phytochemistry Co., Ltd. can help you with your product creation projects because they have 25 years of experience extracting plants and use cutting-edge liposomal technology. We are a qualified manufacturer with ISO 22000, FSSC 22000, cGMP, Halal, Kosher, and organic certifications. We offer pharmaceutical-grade Liposomal Curcumin Powder that is encapsulated at least 90% of the time and has particles that are carefully controlled to be 100–200nm in size. Our clear quality systems give full COAs, original HPLC chromatograms, and the ability to track products from our growing bases to the final testing before they are released. Our expert team is here to help you succeed from the idea stage all the way through to implementation, whether you need large amounts for mass production or specific details for unique formulations. We work with more than 20,000 companies around the world. Our markets are especially strong in North America and Europe, where following rules and maintaining quality are very important. Get in touch with our business development team at duke@hongdaherb.com to talk about your specific needs and get detailed information on why top brands choose HONGDA as their reliable liposomal curcumin powder supplier.
References
1. Anand P, Kunnumakkara AB, Newman RA, Aggarwal BB. "Bioavailability of Curcumin: Problems and Promises." Molecular Pharmaceutics, 2007;4(6):807-818.
2. Hewlings SJ, Kalman DS. "Curcumin: A Review of Its Effects on Human Health." Foods, 2017;6(10):92.
3. Schiborr C, Kocher A, Behnam D, Jandasek J, Toelstede S, Frank J. "The oral bioavailability of curcumin from micronized powder and liquid micelles is significantly increased in healthy humans and differs between sexes." Molecular Nutrition & Food Research, 2014;58(3):516-527.
4. Prasad S, Tyagi AK, Aggarwal BB. "Recent developments in delivery, bioavailability, absorption and metabolism of curcumin: the golden pigment from golden spice." Cancer Research and Treatment, 2014;46(2):2-18.
5. Goel A, Kunnumakkara AB, Aggarwal BB. "Curcumin as 'Curecumin': from kitchen to clinic." Biochemical Pharmacology, 2008;75(4):787-809.
6. Panahi Y, Hosseini MS, Khalili N, Naimi E, Simental-Mendía LE, Majeed M, Sahebkar A. "Effects of curcumin on serum cytokine concentrations in subjects with metabolic syndrome: A post-hoc analysis of a randomized controlled trial." Biomedicine & Pharmacotherapy, 2016;82:578-582.


_1745918362135.webp)

