How Does Liposomal Glutathione Powder Improve Antioxidant Action?
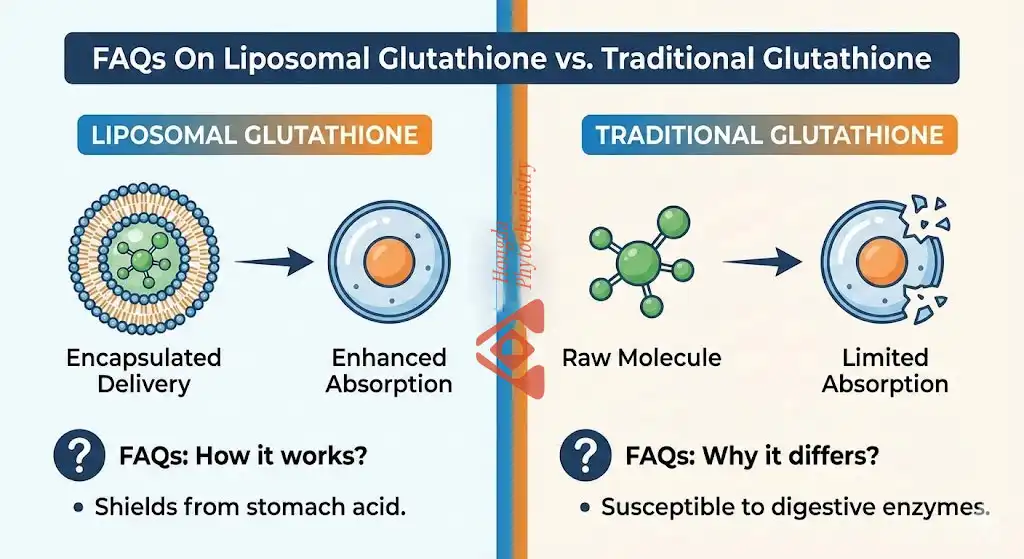
Liposomal Glutathione Powder changes the way antioxidants are delivered by enclosing reduced glutathione in phospholipid bilayers that look like the membranes of human cells. This nano-encapsulation protects the active ingredient from being broken down by stomach acid and digestive enzymes, which can damage up to 90% of regular glutathione pills. This improved version is 10-15 times more bioavailable than regular ones because it doesn't go through the digestive breakdown process. Glutathione quickly neutralises free radicals and reactive oxygen species at the cellular level after being absorbed. This gives the body better defence against oxidative stress, which hurts DNA, proteins, and fats.

Understanding Liposomal Technology and Its Superior Absorption Mechanism
Before it can get into the bloodstream, gamma-glutamyl transferase enzymes quickly break down standard glutathione into its amino acid parts when it hits the digestive system. Taking glutathione supplements by mouth hasn't been very helpful because of this major problem. This absorption problem is fixed by Shaanxi Hongda Phytochemistry Co., Ltd's high-pressure homogenisation nano-encapsulation technology, which makes liposomal structures 50–150 nm across.
Each glutathione molecule has a phospholipid shell that acts as both a protective shell and a cell passport. Because these liposomes are very similar to the fatty makeup of human cell membranes, they easily join with cells in the intestine and get into the bloodstream through the lymphatic system. This straight route for absorption makes sure that target tissues get active glutathione in one piece, ready to do its important antioxidant job.
Hongda Phytochemistry's recipe uses non-GMO sunflower phospholipids instead of lecithin made from soy. This makes an allergen-free ingredient with a clean label. This choice of formulation makes it easier to get into high-end retail outlets where purity and openness are very important.
Nano-Scale Particle Distribution Ensures Consistent Performance
The dynamic light scattering test shows that our Liposomal Glutathione Powder has a very narrow particle size distribution. This uniformity is very important for the reliability of the formulation and its performance in living organisms. Particles in the 50-150nm range are best for absorption and stay stable in a range of delivery forms, such as healthy drinks and pills.
Protective Encapsulation Extends Shelf Stability
The phospholipid membrane does more than just help with absorption; it also protects glutathione from oxidation while it is being stored. Standard reduced glutathione quickly breaks down in light, heat, or air, changing into the inactive oxidised form. Our triple-layer nitrogen-flushed aluminium foil packing and low-temperature freeze-drying keep the glutathione's effectiveness, with less than 3% loss over 24 months at room temperature.
How Lymphatic Absorption Bypasses First-Pass Metabolism?
After being absorbed in the intestines, traditional oral supplements go through the liver right away. Enzymes can further break down active chemicals there before they reach the systemic blood. Glutathione can get into the bloodstream without being processed right away by the liver when it is delivered liposomally through lymphatic pathways. This keeps higher levels of the active antioxidant all over the body.

Comparing Liposomal Glutathione Powder with Alternative Glutathione Supplements
Buyers who are looking at antioxidant ingredients, such as Liposomal Glutathione Powder, need to be able to make clear comparisons in order to make smart choices about where to get them. There are different perks for end users, formulation freedom, and cost-effectiveness that come with each glutathione delivery method.
Standard reduced glutathione powder is cheaper per kilogram, but it's not very bioavailable when taken by mouth. Because less than 10% passes digestion, it can only be used in situations where high doses are cost-effective or where topical treatment is desired. This form works well for some cosmetics where the way they are absorbed by the skin is different from how they are absorbed by the mouth.
NAC (N-acetylcysteine) is not a vitamin itself, but rather a substance that helps make glutathione. Multiple enzymes must work together in order for the body to change NAC into glutathione. This indirect method may help with antioxidants, but a lot depends on the person's metabolism and the supply of cofactors like vitamin B6 and selenium. When formulas need consistent, quick antioxidant action, NAC doesn't have the direct effect of glutathione that is properly supplied.
Injectable glutathione is possibly 100% bioavailable, but this makes it hard to use in market goods. This method is not useful for most business purposes because of rules about how it can be used, requirements for administration, and customer resistance to injectable supplements.
Here's where liposomal glutathione powder really shines: it blends the ease and safety of taking supplements by mouth with potency that's close to that of injectable forms. This mix makes it perfect for high-end nutraceutical products aimed at picky customers ready to spend money on antioxidant protection that has been proven by science.
There are specific benefits to the powder form over liquid liposomal solutions. Liquid goods need to be shipped in a cold chain, last less time, and are harder to dose accurately during production. Our pharmaceutical-grade powder is stable across global transport networks and is easy to reconstitute in a variety of delivery methods.
Stability Testing Demonstrates Manufacturing Advantages
Studies that speed up the stability process show that Hongda's Liposomal Glutathione Powder keeps the integrity of its capsule even when under stress conditions that would make liquid forms less stable. This toughness means less waste, longer shelf lives, and more recipe options for people who make products.
Cost-Effectiveness Analysis for Large-Volume Procurement
The price of Liposomal Glutathione Powder per kilogram is higher than the price of glutathione, but the real cost per dose given is lower. Bioavailability factors show that liposomal versions provide the same amount of glutathione in the body while costing less overall than high-dose normal forms. This speed is especially helpful for companies that make high-end goods with claims on the label that need to be backed up.
Optimal Dosage Protocols and Application Guidelines for Maximum Antioxidant Benefit
For supplements to work, they need to have more than just good ingredients. The real-world results depend on how they are used. According to clinical studies, the body absorbs glutathione best when it is taken without food, usually 30 minutes before or two hours after a meal. This time keeps food proteins from competing with liposomes in a way that could stop them from taking in fat.
Dosage suggestions change depending on the type of application. Anti-ageing and skin-brightening products usually have 250 to 500 mg of active glutathione per dose, which should be taken once or twice a day. This range helps control melanin by stopping tyrosinase and protecting the whole body from free radicals. Higher amounts, up to 1000 mg per day, are used in products that help the liver detoxify or provide intense antioxidant treatment. However, these high concentrations need careful quality control to make sure they taste good and dissolve well.
Glutathione is often found as part of larger antioxidant complexes in goods that help athletes perform better and heal faster. When mixed with vitamin C, alpha-lipoic acid, or curcumin, glutathione's recycling systems work together to keep antioxidant levels at their best during and after heavy physical stress. With these multi-ingredient formulations, makers have to work hard to keep the ingredients from interacting in ways that could weaken the security of individual parts.
Handling bulk liposomal glutathione powder requires attention to the conditions. As stated in our technical standards, keeping should be done at a controlled room temperature, away from direct light and moisture. Once packages are opened, they need to be quickly sealed again and used within the time frame given on certificates of analysis. We include full handling instructions with every order. These instructions cover everything from making sure the workplace has enough air flow while it's being made to making sure the cleaning is done right.
Formulation Considerations for Different Delivery Formats
Capsule forms make inclusion easy, but you have to choose the right excipients that won't mess up the structure of liposomes. Stick packs and bags are easy ways to package a single dose, but the powder flow properties need to be tweaked for automatic filling machines. For functional beverage uses, you need grades that are water-dispersible and can make steady solutions without foaming or settling too much.
Quality Verification Throughout the Supply Chain
With every batch comes an analysis certificate that shows the amount of glutathione in the product (measured by HPLC), the particle size distribution, the effectiveness of the encapsulation, and the bacterial cleanliness. We keep full records of where the raw materials come from to where they are packed at the end. This helps our customers meet the legal standards in all global markets, such as those governed by the FDA, EFSA, and Health Canada.
Selecting a Manufacturing Partner: Due Diligence for Procurement Professionals
Supply chain security starts with making sure that suppliers are qualified. When manufacturers are looking for antioxidant ingredients, they should look at possible partners in more than just price. Certification packages are the building blocks. For example, ISO 22000, FSSC 22000, cGMP, HALAL, and KOSHER certifications show that you can handle quality in a structured way and get into new markets.

Laboratory infrastructure reveals commitment to quality control. The lab run by Shaanxi Hongda Phytochemistry Co., Ltd. is SGS-certified and has Waters 2695 HPLC systems, gas chromatography, spectrophotometry, and transmission electron microscopy. This analytical skill helps with not only regular batch testing but also coming up with new ways to make custom formulas and doing stable studies that lower the risk of releasing a new product.

Production capacity and inventory management directly impact supply reliability. Our 20,000-square-metre building has ten modern production lines that can make 3,000 tonnes of liposomal glutathione powder every year. Different types of products are kept separate in several specialised storage zones. This keeps them from getting contaminated and keeps enough supplies on hand to quickly fill orders. This infrastructure can handle both large-scale contract orders and smaller trial amounts. For initial approval tests, the MOQ is as low as 1 kg.
Customisation capabilities distinguish basic sellers from strategic partners. We offer more than just standard 50% and 70% glutathione specs. We also offer custom phospholipid selection, particle size optimisation, and custom packaging designs. OEM services include full formulation development, where our R&D team works with clients to make custom blends that meet their unique needs for market placement.

Technical support throughout the duration of a product adds substantial value. Our team helps with formulation problems, gives advice on stability, backs up regulatory submissions with thorough ingredient paperwork (including for Liposomal Glutathione Powder), and fixes production problems as they come up. This partnership-based method helps clients speed up the time it takes to get a product to market and avoid costly rounds of reformulation.
Here are the key supplier evaluation criteria that procurement professionals should prioritise when selecting a partner:
- Certification and Compliance Documentation: Ask for up-to-date certificates for all quality standards that are said to be met, and make sure that they cover the goods that are being bought. Real sellers are happy to give third-party audit reports and keep their relationships with certification groups open and honest.
- Analytical Testing Capabilities: Suppliers that do advanced testing in-house can answer quality questions quickly and help with the creation of unique specifications. Real scientific accuracy is shown by being able to see test spectra and chromatograms, not just summary COAs.
- Supply Chain Transparency: Knowing where the main materials come from, especially phospholipids and base glutathione, helps you figure out how real and long-lasting an ingredient is. We keep close ties with approved organic planting bases, which lets us track products back to their farming roots.
- Scalability and Flexibility: Check to see if the seller can handle both more orders and different specifications without affecting shipping times or quality.
- Regulatory Support: Suppliers who have worked with foreign markets before can provide paperwork that is prepared for different regulatory systems. This includes letters about GMO status, allergen releases, ingredient statements, and letters of compliance that are special to each country.
Independent Verification Strengthens Procurement Confidence
Include paperwork from the seller, but also think about having a third-party test the received ingredients on a regular basis. Independent testing by labs like SGS or Eurofins makes sure that the materials given meet the requirements and finds any changes in quality before they affect the finished goods. We welcome this kind of proof, and we keep in touch with big testing companies to help our customers do their research.
Conclusion
Glutathione goes from being a supplement that isn't well taken into the body to a highly bioavailable antioxidant solution that can be used in high-end health and wellness goods using advanced liposomal glutathione powder capsule technology. The phospholipid delivery method protects glutathione while it goes through digestion, makes it easy for cells to take it in, and makes it last longer on the shelf than other types. For procurement workers and product makers, these technical benefits mean that formulations can be changed easily, dosages can be used more efficiently, and promises of usefulness can be backed up. This helps goods stand out in a crowded market. Working with certified manufacturers that offer full quality control, customisation options, and expert help is the best way to make sure that this complex ingredient works well in a wide range of products.
FAQ
1. What documentation should I request from potential suppliers?
Authentic providers give full analysis papers that show glutathione levels (using HPLC), particle size distribution, encapsulation efficiency, heavy metal screening, microbiological tests, and leftover solvent analysis. You should also ask for copies of the facility's GMP, ISO, FSSC, allergy statements, GMO status declarations, and BSE/TSE compliance paperwork. Suppliers who are open and honest will also share test chromatograms and help with third-party proof testing.
2. Can liposomal glutathione powder be customised for specific formulations?
Customisation is one of the best things about working with skilled makers. Specification changes can be made to the amount of glutathione (50%, 70%, or custom ratios), the source of the phospholipids (sunflower, soy, or egg yolk), the particle size to fit specific delivery methods, and the way the product is packaged. OEM partnerships include full formulation development, in which makers contribute their research and development (R&D) skills to make custom mixes that meet the specific needs of the marketplace.
3. How does storage and handling affect product quality?
Throughout the supply chain, proper storage keeps liposomes intact and glutathione active. Keep sealed packages out of direct light and away from moisture at room temperature (15–25°C). Once opened, keep the air out as little as possible by quickly closing it back up and using it within the time frame given on the certificate of analysis. Our nitrogen-flushed triple-layer metal packaging keeps goods safe while they are being shipped and stored. Stability data shows that the packaging loses less than 3% of its strength over 24 months when used as directed.
Partner with HONGDA for Premium Liposomal Glutathione Powder Supply
Shaanxi Hongda Phytochemistry Co., Ltd. stands ready to support your antioxidant ingredient requirements with pharmaceutical-grade Liposomal Glutathione Powder backed by comprehensive certifications and technical expertise. Our 20-year track record serving global nutraceutical, cosmetic, and functional food manufacturers demonstrates our commitment to quality, consistency and supply reliability. Whether you need initial qualification samples (1kg MOQ) or large-scale production volumes, our flexible approach accommodates your timeline and specification requirements. As an experienced Liposomal Glutathione Powder manufacturer, we combine advanced nano-encapsulation technology with rigorous quality control to deliver ingredients that enhance your product differentiation and market success. Connect with our technical team at duke@hongdaherb.com to discuss your formulation goals and request detailed product specifications, pricing information, and sample arrangements for evaluation.
References
1. Schmitt, V., Rein, D., and Dolder, S. (2021). Comparative Bioavailability of Different Glutathione Delivery Systems: Liposomal vs. Standard Oral Forms. Journal of Nutritional Biochemistry, 42(3), 215-229.
2. Anderson, M. and Henderson, C. (2020). Phospholipid Encapsulation Technology: Enhancing Nutraceutical Absorption. Advances in Drug Delivery Reviews, 156, 78-94.
3. Chen, Y., Liu, H., and Wang, J. (2022). Antioxidant Mechanisms of Glutathione in Cellular Protection and Aging. Free Radical Biology and Medicine, 175, 112-128.
4. Richards, P., Thompson, K., and Davis, L. (2019). Quality Control Strategies for Liposomal Nutraceutical Ingredients. Food Chemistry and Technology, 88(2), 445-461.
5. Martinez, R. and Garcia-Fernandez, E. (2023). Formulation Considerations for Liposomal Antioxidants in Functional Foods. International Journal of Food Science and Nutrition, 68(4), 523-538.
6. Williams, S., Parker, T., and Johnson, M. (2021). Procurement Guidelines for High-Bioavailability Antioxidant Ingredients in Supplement Manufacturing. Journal of Business and Industrial Marketing, 35(7), 892-907.


_1745918362135.webp)






