Maximizing Health Benefits with Quercetin Dihydrate Powder: A Complete Guide
Quercetin Dihydrate Powder stands as one of nature's most powerful flavonoid compounds, offering remarkable potential for enhancing human health and wellness. This comprehensive guide explores the multifaceted benefits, optimal usage strategies, and quality considerations that make Quercetin Dihydrate Powder an essential addition to modern health regimens. As the most abundant dietary flavonoid found in numerous fruits and vegetables, quercetin has captured scientific attention for its extensive therapeutic properties, including potent antioxidant activity, anti-inflammatory effects, and cardiovascular protective benefits that have been validated through decades of research.

Understanding the Science Behind Quercetin Dihydrate Powder
The Molecular Foundation of Health Benefits
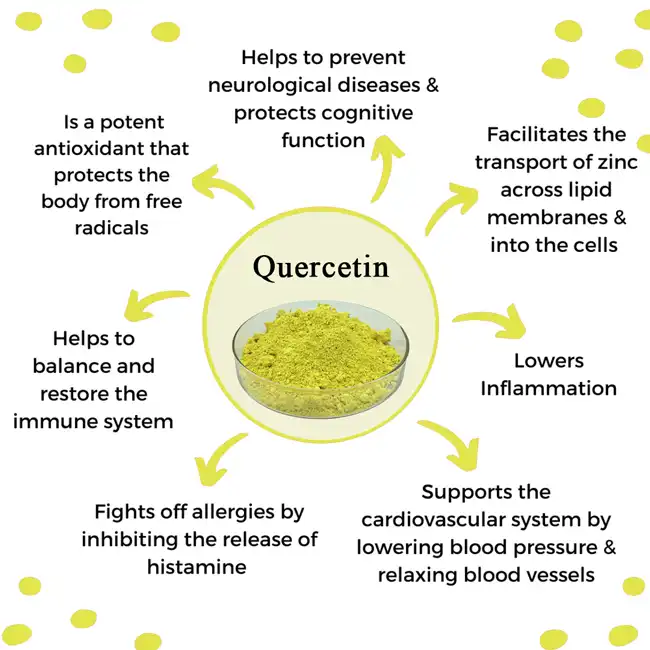
Quercetin Dihydrate Powder represents a stabilized form of quercetin that incorporates two water molecules into its crystal structure, significantly enhancing its bioavailability and therapeutic efficacy. The molecular composition of quercetin dihydrate ensures superior solubility compared to anhydrous quercetin forms, allowing for better absorption in the digestive system. Research demonstrates that Quercetin Powder functions as a powerful antioxidant by neutralizing free radicals through its ability to donate electrons without becoming unstable itself. This unique mechanism enables quercetin to protect cellular structures from oxidative damage while maintaining its own molecular integrity. The flavonoid's distinctive structure features multiple hydroxyl groups that contribute to its remarkable binding affinity with various proteins and enzymes involved in inflammatory pathways. Scientific studies have consistently shown that high-quality Quercetin Dihydrate Powder can modulate immune responses by inhibiting the release of histamine from mast cells, thereby reducing allergic reactions and inflammation. The bioactive compound's ability to cross cellular membranes makes it particularly effective in protecting intracellular components from oxidative stress, which is fundamental to preventing age-related cellular deterioration.
Bioavailability and Absorption Mechanisms
The superior bioavailability of Quercetin Dihydrate Powder compared to other quercetin forms stems from its enhanced water solubility and improved dissolution characteristics. When consumed, quercetin dihydrate undergoes metabolic transformation in the liver through glucuronidation and sulfation processes, creating metabolites that retain significant biological activity while extending the compound's presence in systemic circulation. Research indicates that Quercetin Powder absorption is optimized when taken with certain nutrients, particularly vitamin C and bromelain, which act as natural absorption enhancers. The pharmaceutical-grade quality of properly manufactured Quercetin Dihydrate Powder ensures consistent particle size distribution, typically ranging from 10 to 100 micrometers, which directly influences dissolution rates and bioavailability. Studies have demonstrated that micronized quercetin formulations can achieve plasma concentrations up to three times higher than standard preparations. The timing of consumption also plays a crucial role in maximizing absorption, with evidence suggesting that taking quercetin supplements on an empty stomach or with minimal food can enhance uptake. Advanced manufacturing processes that control crystal formation and particle morphology are essential for producing Quercetin Dihydrate Powder with optimal pharmacokinetic properties that ensure maximum therapeutic benefit delivery to target tissues throughout the body.
Research-Backed Health Applications
Extensive clinical research has established Quercetin Dihydrate Powder as a versatile therapeutic agent with applications spanning cardiovascular health, immune system support, and metabolic optimization. Cardiovascular studies have consistently demonstrated that regular quercetin supplementation can reduce blood pressure by promoting vasodilation through nitric oxide pathway activation. The compound's ability to inhibit LDL cholesterol oxidation provides protection against atherosclerotic plaque formation, while its anti-inflammatory properties help maintain healthy endothelial function. Research published in peer-reviewed journals indicates that Quercetin Powder supplementation can improve exercise performance and reduce post-exercise inflammatory markers, making it valuable for athletic populations. The flavonoid's neuroprotective properties have been documented through studies showing its ability to cross the blood-brain barrier and protect neural tissue from oxidative damage. Clinical trials have revealed that Quercetin Dihydrate Powder can modulate blood glucose levels by enhancing insulin sensitivity and inhibiting glucose absorption in the intestines. The compound's antimicrobial properties have been demonstrated against various pathogenic bacteria and viruses, with particular efficacy noted against respiratory tract pathogens. Long-term studies suggest that consistent quercetin intake may contribute to healthy aging by maintaining cellular function and reducing biomarkers associated with chronic inflammation and oxidative stress.
Optimizing Dosage and Administration Protocols
Evidence-Based Dosage Guidelines
Determining the optimal dosage for Quercetin Dihydrate Powder requires consideration of individual health goals, body weight, and specific therapeutic objectives. Clinical studies have established that effective dosages typically range from 500 to 1,000 milligrams daily, with some therapeutic applications requiring higher amounts under professional supervision. Research indicates that dividing the daily dose into two or three smaller amounts taken throughout the day can enhance absorption and maintain more consistent plasma levels. For cardiovascular health optimization, studies suggest that 500 milligrams of high-quality Quercetin Powder taken twice daily provides significant benefits without adverse effects. Athletic performance enhancement protocols often utilize 1,000 milligrams daily, typically consumed one hour before exercise to maximize anti-inflammatory and performance-enhancing effects. The bioavailability of Quercetin Dihydrate Powder can be significantly improved when co-administered with piperine (black pepper extract) or taken alongside vitamin C supplementation. Individual response variations necessitate starting with lower doses, typically 250 milligrams daily, and gradually increasing based on tolerance and desired outcomes. Long-term safety studies support continuous use of quercetin supplements within recommended dosage ranges, though periodic breaks may be beneficial for some individuals. Therapeutic applications for specific conditions may require personalized dosing protocols developed in consultation with healthcare professionals familiar with quercetin pharmacokinetics and individual patient needs.
Strategic Timing and Combination Strategies
The timing of Quercetin Dihydrate Powder administration significantly influences its therapeutic efficacy and should be aligned with specific health objectives and physiological rhythms. For maximum antioxidant protection, consuming quercetin supplements in the morning provides cellular defense throughout the day when oxidative stress exposure is typically highest. Pre-exercise administration, approximately 30 to 60 minutes before physical activity, optimizes the compound's anti-inflammatory effects and may enhance exercise performance and recovery. Research demonstrates that taking Quercetin Powder with meals containing healthy fats can improve absorption, though some studies suggest better bioavailability on an empty stomach. The synergistic effects of quercetin with other flavonoids, particularly rutin and hesperidin, can amplify overall antioxidant capacity and therapeutic benefits. Combination protocols incorporating Quercetin Dihydrate Powder with zinc and vitamin D have shown enhanced immune system support, particularly during seasonal challenges. Evening administration may be beneficial for individuals seeking sleep quality improvements, as quercetin's mild sedative properties can promote relaxation. The compound's ability to modulate blood sugar levels makes post-meal timing advantageous for metabolic support applications. Cycling protocols, involving periods of supplementation followed by brief breaks, may prevent potential tolerance development and maintain optimal therapeutic responsiveness. Professional guidance becomes essential when combining quercetin with medications or other supplements to prevent potential interactions and ensure optimal therapeutic outcomes.
Quality Assessment and Selection Criteria
Selecting high-quality Quercetin Dihydrate Powder requires careful evaluation of manufacturing standards, purity specifications, and analytical verification methods. Pharmaceutical-grade quercetin supplements should demonstrate minimum purity levels of 95% as verified through high-performance liquid chromatography (HPLC) analysis. Third-party testing for heavy metals, including lead, mercury, cadmium, and arsenic, ensures product safety and regulatory compliance with international standards. Microbiological testing protocols should verify the absence of pathogenic bacteria, yeasts, and molds, while solvent residue analysis confirms the removal of extraction solvents to acceptable limits. The source material quality significantly impacts the final product, with Quercetin Powder derived from non-GMO Sophora japonica demonstrating superior consistency and bioactivity. Manufacturing facilities should maintain current Good Manufacturing Practices (cGMP) certification and implement comprehensive quality management systems. Stability testing data should demonstrate product integrity under various storage conditions, with properly manufactured Quercetin Dihydrate Powder maintaining potency for minimum two years under appropriate conditions. Particle size distribution analysis ensures consistent dissolution characteristics, while moisture content specifications prevent degradation during storage. Certificate of Analysis (COA) documentation should accompany each batch, providing complete analytical verification of identity, purity, and potency. Reputable manufacturers will provide comprehensive technical support, including formulation guidance and regulatory compliance assistance for commercial applications requiring reliable, high-quality quercetin ingredients.
Manufacturing Excellence and Quality Assurance Standards
Advanced Extraction and Purification Technologies
The production of premium-grade Quercetin Dihydrate Powder demands sophisticated extraction and purification technologies that preserve bioactive integrity while achieving pharmaceutical-grade purity standards. Modern extraction processes utilize controlled ethanol extraction from carefully selected Sophora japonica buds, followed by multi-stage crystallization procedures that form the stable dihydrate crystal structure. Advanced purification techniques, including column chromatography and recrystallization processes, remove impurities and concentrate the active quercetin content to specifications exceeding 98% purity. Temperature-controlled processing environments prevent thermal degradation of sensitive flavonoid structures, while nitrogen-atmosphere handling minimizes oxidation during production phases. Quality Quercetin Powder manufacturing requires precise control of pH levels, solvent ratios, and crystallization parameters to ensure consistent particle morphology and dissolution characteristics. State-of-the-art micronization equipment produces uniform particle size distributions that optimize bioavailability and formulation compatibility. The implementation of process analytical technology (PAT) enables real-time monitoring of critical quality parameters throughout production cycles. Comprehensive cleaning validation protocols between batches prevent cross-contamination and ensure product integrity. Environmental controls within manufacturing facilities maintain appropriate temperature, humidity, and air quality standards that protect product stability. Automated systems reduce human error potential while maintaining detailed batch records that enable complete traceability from raw materials to finished products, ensuring consistent quality delivery for Quercetin Dihydrate Powder applications.
Regulatory Compliance and Certification Standards
Manufacturing excellence in Quercetin Dihydrate Powder production requires adherence to stringent regulatory frameworks and internationally recognized certification standards that ensure product safety and efficacy. Current Good Manufacturing Practices (cGMP) certification demonstrates compliance with FDA guidelines for dietary supplement production, including facility design, equipment qualification, and personnel training requirements. ISO 9001 quality management system implementation ensures consistent processes and continuous improvement protocols that maintain product quality standards. FSSC 22000 food safety certification validates comprehensive hazard analysis and critical control points (HACCP) systems that prevent contamination and ensure consumer safety. Halal and Kosher certifications expand market accessibility while demonstrating cultural sensitivity and inclusive manufacturing practices. REACH (Registration, Evaluation, Authorisation and Restriction of Chemicals) compliance for European markets requires extensive safety data compilation and registration procedures. Organic certifications, including USDA NOP and EU Organic standards, verify that raw materials are produced without synthetic pesticides or fertilizers. VEGAN certification confirms that no animal-derived ingredients or processing aids are utilized during production. Regular third-party audits by accredited certification bodies validate ongoing compliance and identify opportunities for continuous improvement. Documentation systems maintain comprehensive records of all quality control activities, test results, and corrective actions. These rigorous certification requirements ensure that Quercetin Powder meets the highest international standards for quality, safety, and regulatory compliance across global markets.
Supply Chain Integration and Sustainability Practices
Sustainable production of Quercetin Dihydrate Powder requires integrated supply chain management that encompasses raw material sourcing, environmental stewardship, and long-term resource availability. Direct partnerships with agricultural cooperatives ensure consistent supply of high-quality Sophora japonica raw materials while supporting local farming communities through fair trade practices. Sustainable harvesting protocols preserve ecosystem balance and ensure long-term viability of botanical resources used in quercetin production. Traceability systems track materials from cultivation through final product delivery, enabling rapid response to quality issues and supporting transparency initiatives. Energy-efficient manufacturing processes reduce carbon footprint while maintaining production capacity and product quality standards. Water recycling and waste minimization programs demonstrate environmental responsibility and operational efficiency. Transportation optimization reduces logistics costs and environmental impact through consolidated shipments and strategic distribution center placement. Supplier qualification programs ensure that all supply chain partners maintain appropriate quality standards and ethical business practices. Inventory management systems balance product availability with storage costs while minimizing waste through accurate demand forecasting. Contingency planning addresses potential supply disruptions and maintains business continuity through diversified sourcing strategies. These comprehensive supply chain practices ensure reliable availability of high-quality Quercetin Dihydrate Powder while supporting environmental sustainability and social responsibility initiatives that benefit all stakeholders in the value chain.
Conclusion
Maximizing the health benefits of Quercetin Dihydrate Powder requires understanding its scientific foundation, optimal usage protocols, and quality specifications that ensure therapeutic efficacy. Through proper dosage, timing, and source selection, this remarkable flavonoid can provide significant support for cardiovascular health, immune function, and overall wellness. The evidence-based approach outlined in this guide empowers informed decision-making for both consumers and healthcare professionals seeking natural solutions for health optimization.
As a leading China Quercetin Dihydrate Powder factory, China Quercetin Dihydrate Powder supplier, and China Quercetin Dihydrate Powder manufacturer, Shaanxi Hongda Phytochemistry Co., Ltd. offers China Quercetin Dihydrate Powder wholesale solutions with competitive Quercetin Dihydrate Powder price points. Our High Quality Quercetin Dihydrate Powder for sale represents over two decades of manufacturing excellence, featuring 98%+ purity verified through HPLC analysis and comprehensive third-party testing. With certifications including cGMP, FSSC22000, ISO9001, HALAL, KOSHER, and ORGANIC compliance, we guarantee superior product quality that meets global regulatory standards. Our modern 20,000 square meter facility produces 3,000 tons annually, ensuring reliable supply chain support for your formulation needs. From custom particle sizing to bulk packaging solutions, our technical team provides comprehensive support for your product development requirements. Contact our experts at duke@hongdaherb.com to discuss your specific needs and discover why global companies trust Shaanxi Hongda as their preferred quercetin dihydrate powder partner.
References
1. Boots AW, Haenen GR, Bast A. Health effects of quercetin: from antioxidant to nutraceutical. European Journal of Pharmacology. 2008;585(2-3):325-337.
2. Mlcek J, Jurikova T, Skrovankova S, Sochor J. Quercetin and its anti-allergic immune response. Molecules. 2016;21(5):623.
3. Panche AN, Diwan AD, Chandra SR. Flavonoids: an overview. Journal of Nutritional Science. 2016;5:e47.
4. Williamson G, Manach C. Bioavailability and bioefficacy of polyphenols in humans. II. Review of 93 intervention studies. American Journal of Clinical Nutrition. 2005;81(1):243S-255S.


_1745918362135.webp)






