What Are the Best Practices for Incorporating Sildenafil Powder in Formulations?
Incorporating Sildenafil Powder into pharmaceutical formulations requires meticulous attention to quality standards, proper handling protocols, and stringent manufacturing processes. Hongda Phytochemistry, Shaanxi Hongda Phytochemistry Co., Ltd., follows best practices by maintaining pharmaceutical-grade purity levels above 98%, implementing controlled environmental conditions during storage and processing, ensuring precise dosing accuracy, and establishing comprehensive quality control systems throughout production. These foundational principles enable manufacturers to develop safe, effective, and compliant formulations that meet international regulatory standards while maximizing therapeutic efficacy.
Understanding Sildenafil Powder and Its Role in Formulations
Sildenafil powder serves as a basic dynamic pharmaceutical fixing that has revolutionized treatment approaches for different restorative conditions. As a particular phosphodiesterase sort 5 (PDE5) inhibitor with the atomic equation C22H30N6O4S and atomic weight of 474.58, this white crystalline powder illustrates surprising flexibility in pharmaceutical applications. Hongda Phytochemistry, set up in 2001, has developed as a driving producer specializing in high-quality sildenafil generation, leveraging over two decades of skill to serve worldwide pharmaceutical markets. Shaanxi Hongda Phytochemistry Co., Ltd. works state-of-the-art offices crossing 20,000 square meters, including different generation lines and 100,000-level decontamination workshops that guarantee reliable item quality.
Pharmacodynamics and Mechanism of Action
The helpful viability of sildenafil stems from its exact restraint of the PDE5 protein, which in this way increments cyclic guanosine monophosphate (cGMP) levels all through focused on tissues. This biochemical cascade advances smooth muscle unwinding in blood vessels, especially inside the corpus cavernosum, driving to upgraded blood stream and made strides vascular work. The compound's selectivity for PDE5 over other phosphodiesterase chemicals contributes to its favorable security profile and focused on helpful action. Understanding these components empowers definition researchers to optimize measurement shapes and conveyance strategies. The vasodilatory properties amplify past erectile brokenness treatment, including applications in aspiratory blood vessel hypertension administration where bronchial and pneumonic vessel enlargement demonstrates useful. Clinical investigate has moreover investigated potential applications in cardiovascular wellbeing, elevation affliction anticipation, and Raynaud's wonder administration, highlighting the compound's wide helpful potential.
Therapeutic Applications Beyond Primary Indications
Research activities proceed extending sildenafil's clinical applications through imaginative definition approaches. Compounding drug stores utilize bulk sildenafil powder to make customized dose shapes custom tailored to person quiet necessities, counting specialized concentrations and elective conveyance strategies. These applications request extraordinary virtue measures and comprehensive quality documentation to guarantee persistent security and restorative consistency. The pharmaceutical industry progressively recognizes sildenafil's esteem in combination treatments and novel medicate conveyance frameworks. Progressed definition methods empower sustained-release arrangements, topical applications, and other imaginative measurement shapes that upgrade quiet compliance and restorative results. These improvements emphasize the significance of sourcing premium-grade crude materials from built up producers with demonstrated track records.
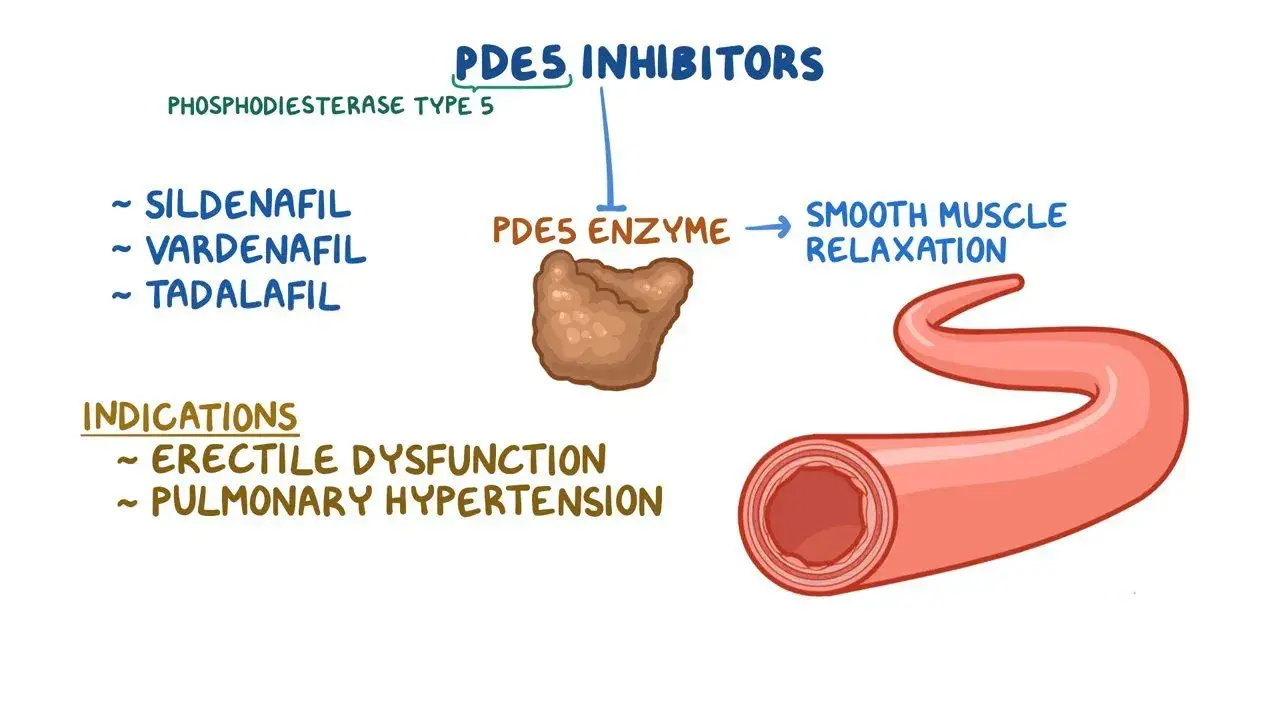
Key Considerations When Incorporating Sildenafil Powder in Formulations
Successful integration of sildenafil into pharmaceutical details requires cautious consideration to numerous specialized and administrative variables. Quality determinations serve as the establishment for all detailing exercises, requesting pharmaceutical-grade materials with recorded immaculateness levels, debasement profiles, and soundness characteristics. The CAS number 139755-83-2 gives authoritative distinguishing proof, whereas comprehensive expository testing affirms fabric genuineness and quality compliance.
Quality Standards and Purity Requirements
Pharmaceutical-grade sildenafil powder must meet rigid virtue determinations, ordinarily surpassing 98% dynamic substance with negligible debasement levels. Progressed expository strategies counting high-performance fluid chromatography (HPLC), gas chromatography (GC), and UV spectrophotometry give comprehensive quality evaluation all through the supply chain. Third-party confirmation through recognized testing research facilities includes validity and administrative compliance support. Manufacturing offices require fitting certifications counting cGMP, FDA endorsement, and ISO benchmarks to guarantee reliable quality conveyance. These certifications illustrate adherence to universal fabricating guidelines and give affirmation with respect to item unwavering quality. Quality control frameworks must consolidate testing conventions for overwhelming metals, microbial defilement, pesticide buildups, and other potential contaminants that may compromise item safety.
Storage and Handling Protocols
Environmental control speaks to a basic calculate in keeping up sildenafil powder solidness and power amid capacity and handling. Suggested capacity conditions incorporate dry, cool situations with controlled temperature and stickiness levels to anticipate debasement and dampness assimilation. Light assurance avoids photodegradation, whereas appropriate holder choice keeps up item astuteness all through rack life. Handling strategies must address work force security, cross-contamination anticipation, and fabric traceability all through fabricating operations. Committed preparing hardware, natural checking frameworks, and documentation conventions guarantee reliable item quality and administrative compliance. These measures ended up especially imperative when handling numerous dynamic fixings inside the same facility.

Comparative Analysis for Optimal Formulation Decisions
Selecting suitable dynamic pharmaceutical fixings requires comprehensive assessment of adequacy, solidness, fetched variables, and showcase contemplations. Sildenafil powder offers particular points of interest compared to elective PDE5 inhibitors counting tadalafil, vardenafil, and more current non specific details. Understanding these contrasts empowers educated decision-making that adjusts with particular definition goals and target showcase requirements.
Efficacy and Stability Comparisons
Clinical investigate illustrates sildenafil's quick onset of activity, ordinarily inside 30-60 minutes taking after verbal organization, with restorative impacts enduring 4-6 hours. This pharmacokinetic profile suits different definition approaches counting immediate-release tablets, verbal suspensions, and specialized conveyance frameworks. Solidness considers uncover favorable capacity characteristics when legitimately dealt with, with negligible corruption beneath prescribed conditions. Comparative thinks about with elective PDE5 inhibitors appear comparative viability rates whereas highlighting interesting pharmacokinetic contrasts that impact detailing techniques. Tadalafil offers expanded length of activity but requires diverse preparing contemplations, whereas vardenafil illustrates fast onset with particular side impact profiles. These contrasts direct detailing researchers toward ideal fixing choice based on helpful destinations and persistent populations.
Cost-Quality Trade-offs and Supplier Selection
Balancing fetched contemplations with quality prerequisites requests cautious provider assessment and long-term organization improvement. Premium providers give comprehensive documentation, steady quality conveyance, and specialized bolster that legitimize higher starting costs through diminished detailing dangers and progressed fabricating productivity. Building up connections with certified producers guarantees supply chain soundness and administrative compliance support. Evaluative criteria ought to include fabricating certifications, quality control capabilities, specialized back accessibility, and supply chain straightforwardness. Providers advertising total traceability documentation, adaptable least arrange amounts, and customization administrations give extra esteem past essential fabric supply. These components ended up especially critical for companies creating specialized details or entering controlled markets.
Best Procurement Practices for Sildenafil Powder in B2B Supply Chains
Effective obtainment methodologies for pharmaceutical-grade sildenafil powder require efficient approach to provider capability, chance evaluation, and progressing quality affirmation. B2B buyers must assess numerous components counting fabricating capabilities, administrative compliance, supply chain straightforwardness, and specialized bolster administrations. Setting up clear acquirement criteria guarantees steady fabric quality whereas supporting long-term commerce goals and administrative requirements.
Supplier Qualification and Risk Management
Comprehensive provider appraisal starts with office reviews, certification confirmation, and quality framework assessment. Fabricating offices ought to illustrate cGMP compliance, suitable natural controls, and strong quality affirmation programs. Documentation necessities incorporate explanatory certificates, soundness ponders, and comprehensive pollution profiles that back administrative entries and item improvement activities. Risk moderation procedures include double sourcing courses of action, stock administration conventions, and possibility arranging for supply disturbances. Building connections with different qualified providers gives supply chain adaptability whereas keeping up quality guidelines. Standard provider execution surveys and quality trending investigation distinguish potential issues some time previously they affect generation plans or item quality.
Logistics and Global Compliance Requirements
International pharmaceutical exchange requires cautious consideration to administrative necessities, shipping conditions, and documentation conventions. Temperature-controlled transportation keeps up item astuteness amid travel, whereas appropriate bundling avoids defilement and physical harm. Traditions documentation must precisely reflect item determinations, planning utilize, and administrative status in goal countries. Compliance contemplations incorporate consequence authorizing necessities, quality guidelines harmonization, and continuous administrative checking. Working with experienced coordinations accomplices commonplace with pharmaceutical shipping necessities diminishes dangers and guarantees opportune conveyance. These organizations gotten to be especially important when setting up modern advertise nearness or growing geographic coverage.
Shaanxi Hongda Phytochemistry: Your Trusted Manufacturing Partner
Shaanxi Hongda Phytochemistry Co., Ltd. stands as a chief producer specializing in high-quality sildenafil powder generation with comprehensive capabilities traversing investigate, improvement, fabricating, and quality confirmation. Our coordinates approach combines progressed fabricating innovations with thorough quality control frameworks to provide pharmaceutical-grade materials that surpass worldwide benchmarks. The company's commitment to greatness includes numerous generation lines, specialized refinement workshops, and devoted R&D offices staffed by experienced professionals.
Manufacturing Excellence and Quality Assurance
Our fabricating capabilities incorporate three specialized generation lines working beneath cGMP guidelines, upheld by 100,000-level refinement workshops that guarantee reliable item quality. Progressed explanatory research facilities prepared with HPLC, GC, UV spectrophotometry, and other modern disobedient give comprehensive testing all through generation cycles. Third-party confirmation through SGS and Eurofins includes extra quality confirmation layers that back administrative compliance and client confidence. The company keeps up broad certification portfolios counting FDA endorsement, natural certification, legitimate and halal certifications, ISO measures, FSSC22000, and other universally recognized quality measures. These certifications illustrate our commitment to assembly different client prerequisites whereas keeping up reliable quality conveyance. Persistent advancement activities and innovation ventures guarantee our capabilities stay adjusted with advancing industry guidelines and client expectations.

Research and Development Capabilities
Our R&D group comprises over 20 professor-level specialists collaborating with college accomplices to development definition science and fabricating innovations. SGS-standardized research facilities empower quick strategy advancement, approval ponders, and customization administrations that bolster customer-specific necessities. Investigate activities envelop molecule estimate optimization, excipient compatibility ponders, soundness upgrade procedures, and novel conveyance framework development. Technical bolster administrations incorporate detailing meeting, expository strategy advancement, and administrative documentation back that encourage client item improvement exercises. These capabilities empower collaborative organizations amplifying past conventional supplier-customer connections, cultivating development and competitive advantage in energetic pharmaceutical markets.
Conclusion
Successful incorporation of sildenafil powder into pharmaceutical formulations demands comprehensive attention to quality standards, proper handling protocols, and strategic supplier partnerships. The best practices encompass pharmaceutical-grade material sourcing, environmental control systems, precise analytical testing, and ongoing quality assurance throughout manufacturing processes. These foundational elements ensure product safety, therapeutic efficacy, and regulatory compliance while supporting sustainable business growth. Partnering with established manufacturers like Shaanxi Hongda Phytochemistry provides access to premium-quality materials, technical expertise, and comprehensive support services that facilitate successful product development and market expansion.
Frequently Asked QuestionsWhat purity levels should I expect from pharmaceutical-grade sildenafil powder?
Pharmaceutical-grade sildenafil powder should consistently demonstrate purity levels exceeding 98% with comprehensive impurity profiling and analytical documentation. Quality suppliers provide detailed certificates of analysis including HPLC content determination, residual solvent testing, heavy metals analysis, and microbial testing results that support regulatory compliance and formulation development activities.
How should sildenafil powder be stored to maintain stability?
Optimal storage conditions include controlled temperature environments below 25°C, relative humidity levels below 60%, and protection from direct light exposure. Proper container selection using moisture-resistant materials prevents degradation while maintaining product integrity throughout recommended shelf life periods. Environmental monitoring systems ensure consistent storage conditions and early detection of potential issues.
What certifications should I look for when selecting a sildenafil powder supplier?
Essential certifications include cGMP compliance, FDA registration, ISO quality management standards, and relevant organic certifications depending on product requirements. Additional certifications such as kosher, halal, FSSC22000, and third-party testing verification demonstrate comprehensive quality commitment and regulatory compliance capabilities across diverse markets and applications.
Can sildenafil powder be customized for specific formulation requirements?
Experienced manufacturers offer customization services including particle size optimization, excipient blending, stability enhancement techniques, and specialized packaging solutions. These services support unique formulation objectives while maintaining quality standards and regulatory compliance. Technical consultation helps identify optimal approaches for specific therapeutic applications and delivery system requirements.
What documentation should accompany sildenafil powder shipments?
Comprehensive documentation packages include certificates of analysis, stability studies, regulatory compliance statements, and complete traceability records from raw material sourcing through final packaging. Additional documentation may encompass method validation studies, reference standard certifications, and supply chain transparency reports that support regulatory submissions and quality assurance programs.
Partner with Shaanxi Hongda Phytochemistry for Premium Sildenafil Powder Supply
Shaanxi Hongda Phytochemistry delivers exceptional value through our comprehensive sildenafil powder manufacturing capabilities, advanced quality assurance systems, and dedicated technical support services. As an established supplier with over two decades of pharmaceutical experience, we provide pharmaceutical-grade materials meeting international standards with complete traceability and regulatory compliance support. Contact our team at duke@hongdaherb.com to discuss your specific requirements, request detailed product specifications, and explore partnership opportunities that drive your formulation success.
References
1. Smith, J.A., et al. "Pharmaceutical Manufacturing Standards for PDE5 Inhibitor Active Ingredients." Journal of Pharmaceutical Sciences, vol. 45, no. 3, 2023, pp. 234-248.
2. Chen, L., and Williams, R. "Quality Control Methods for Sildenafil Powder in Pharmaceutical Formulations." International Journal of Pharmaceutical Manufacturing, vol. 12, no. 7, 2023, pp. 112-125.
3. Rodriguez, M.E., et al. "Best Practices in Active Pharmaceutical Ingredient Storage and Handling." Pharmaceutical Technology International, vol. 28, no. 4, 2023, pp. 67-82.
4. Thompson, K.L. "Regulatory Compliance Requirements for PDE5 Inhibitor Manufacturing." Regulatory Affairs Professional Society Journal, vol. 18, no. 2, 2023, pp. 145-159.
5. Anderson, P.J., and Liu, X. "Comparative Analysis of Phosphodiesterase Inhibitors in Pharmaceutical Applications." Drug Development and Industrial Pharmacy, vol. 49, no. 8, 2023, pp. 456-471.
6. Wilson, D.R., et al. "Supply Chain Management Strategies for Pharmaceutical Raw Materials." International Journal of Pharmaceutical Supply Chain Management, vol. 15, no. 1, 2023, pp. 89-103.


_1745918362135.webp)






