Which Industries Use Liposomal Vitamin C Powder Bulk Most Often?
Liposomal Vitamin C Powder Bulk has become popular as an ingredient in many manufacturing fields because it is more bioavailable than regular ascorbic acid—up to 6–10 times better. Dietary supplements and nutraceuticals, cosmetics and personal care, functional foods and drinks, pharmaceutical companies, and animal feeding companies are the ones that use this advanced recipe the most. The phospholipid-encapsulated delivery system is valued by all sectors because it can improve cellular absorption and stay stable in a variety of formulations. This makes it an important raw material for B2B buying teams that are focused on regulatory compliance and product effectiveness.
Overview of Liposomal Vitamin C Powder and Its Bulk Market
As companies become more aware of the problems with regular vitamin C types, the market for liposomal vitamin C powder bulk has grown around the world. There are a lot of problems with traditional ascorbic acid. When doses go over 200 mg, absorption rates stop at about 15-20%, and high amounts often cause stomach problems. Because of these problems, research and development teams have turned to capsule technologies that work like the walls of living cells.
Understanding the Phospholipid Encapsulation Technology
Hongda Phytochemistry has created a unique "thin-film hydration-ultrasonic nano-shearing" linking process that makes liposomes with particles that are 100–250 nm in size. This tiny engineering lets the vitamin C molecules get around the normal ways that digestion breaks down food. Three technical factors are most important to purchasing managers when looking at bulk powder specifications: packaging efficiency above 90%; particle size distribution measured by dynamic light scattering (DLS); and polydispersity index (PDI) below 0.25. These measures have a direct link to the uniformity of the formulation and the performance of the final product. Our facility follows strict quality control procedures. Each batch is tested using HPLC to make sure the vitamin C content is between 50 and 55%. This makes sure that makers get exactly calibrated raw materials for their production plans.
Why Do B2B Buyers Choose Bulk Powder Over Liquid Forms?
When it comes to the supply chain, bulk powder forms solve important problems that liquid liposomal goods can't, and Liposomal Vitamin C Powder Bulk is a prime example. Liquid formulations usually break down in 6 to 12 months and need to be kept in the fridge, which makes foreign shipping more difficult and raises the cost of storing. Pharmaceutical-grade liposomal powder from Shaanxi Hongda Phytochemistry Co., Ltd. can be kept at room temperature for three years if it is stored correctly, which greatly lowers the risk of running out of stock. The fine powder is light yellow to white and comes in 12 kg drums. There are 27 drums on a stack, which makes it easy to ship containers from our factory in China to markets in North America, Europe, and Asia. Just-in-time production methods can be used with delivery times of 3–7 working days from the confirmation of the order. This advantage of stability lets contract makers keep to regular production plans without having to deal with the time pressures that come with using liquid ingredients that go bad quickly.
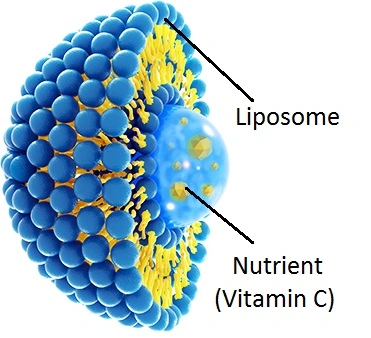
Primary Industries Using Bulk Liposomal Vitamin C Powder
As formulators discover new applications for its unique properties, the use of bulk liposomal vitamin C powder expands across multiple sectors. Understanding each industry's specific requirements helps procurement professionals align technical specifications with end-use needs.
Dietary Supplements and Nutraceuticals: The Dominant Sector
Nutraceutical manufacturers represent the largest consumer group, driven by demand for effective immune-support products. Liposomal encapsulation enables high-dose formulations (often 1,000–2,000 mg per serving) without causing osmotic diarrhea associated with standard ascorbic acid. Quality assurance teams prioritize suppliers providing comprehensive Certificates of Analysis (COAs) that include heavy metal testing, microbial counts, and residual solvent levels. Typical specifications meet USP‑NF 2022 limits, e.g., lead ≤3 ppm, arsenic ≤3 ppm, cadmium ≤1 ppm, and mercury ≤0.1 ppm. The powder’s excellent flow properties support automated capsule filling, maintaining consistent fill weights of 500–800 mg per size 0 capsule. Formulations using non‑GMO sunflower phospholipids avoid common allergen labeling, simplifying clean‑label product launches.
Cosmetics and Personal Care: Advanced Dermal Delivery
Cosmetic scientists incorporate liposomal vitamin C into serums, creams, and powder‑based beauty products because the phospholipid bilayer enhances skin penetration. The encapsulated form remains stable in dry matrices, whereas free ascorbic acid degrades rapidly upon exposure to air and light. Anti-aging lines leverage clinically observed benefits such as improved skin elasticity and reduced photodamage. Powder format enables innovative delivery systems, including “fresh‑activated” serums where consumers mix the powder with a liquid base immediately before use, ensuring maximum potency and overcoming stability challenges in finished goods. Cosmetic purchasers often start with smaller orders (50–100 kg) to test compatibility with various base formulations before scaling up.
Functional Foods and Beverages: Fortification Without Compromise
Food technologists value liposomal vitamin C powder for its ability to withstand processing, maintain shelf stability, and avoid off‑flavors. In drink mixes, the ingredient dissolves readily, creating a milky suspension that conveys a premium sensory experience. The phospholipid coating masks the sharp acidity of raw ascorbic acid, allowing higher fortification levels without altering taste profiles. For extruded or baked goods, the liposomal structure protects vitamin C from thermal degradation, preserving nutritional content. Regulatory teams appreciate the accompanying documentation—allergen statements, non‑GMO certifications, and detailed nutritional composition—which facilitates compliance with FDA labeling requirements.
Pharmaceutical Applications: Clinical‑Grade Requirements
Pharmaceutical developers sourcing oral supplementation protocols for targeted patient populations demand the highest purity and exhaustive documentation. Liposomal vitamin C is increasingly prescribed by hospital nutrition departments and integrative medicine practices for individuals with malabsorption syndromes or those requiring megadose therapy. Its enhanced bioavailability achieves therapeutic plasma concentrations with lower oral doses, improving patient adherence. Procurement departments conduct rigorous supplier audits focusing on cGMP compliance, batch record integrity, and change‑control procedures. Certifications such as ISO 22000 and FSSC 22000 demonstrate adherence to food‑safety management systems expected of pharmaceutical‑grade ingredient suppliers. Manufacturers must also support post‑market surveillance and stability monitoring by providing retained samples from each production batch.
Animal Nutrition: Emerging High‑Growth Segment
The animal health sector is a rapidly growing market for liposomal vitamin C. Premium pet supplement brands use liposomal vitamin C powder bulk to support immune function and joint health in chewable pills and powders. Poultry and swine nutritionists report improved growth performance and disease resistance in feeding trials compared to conventional vitamin C supplementation. The enhanced cellular uptake allows lower inclusion rates, delivering cost savings at scale. Animal feed buyers typically negotiate multi-ton annual contracts and favor suppliers with reliable inventory levels of liposomal vitamin C powder bulk to prevent production interruptions. The powder’s three‑year shelf life is especially advantageous in this segment, where ingredient turnover is slower than in human nutrition applications.

Comparing Bulk Liposomal Vitamin C Powder to Other Forms
When procurement teams know the technical differences between delivery forms, they can make smart buying choices that fit their production needs and help them place themselves in the market for liposomal vitamin C powder in bulk.
Powder Versus Capsule Formats: Production Flexibility
Pre-encapsulated goods are convenient, but they make it harder to change the way they are made. When companies buy finished capsules, they are limited to certain pill amounts and capsule types, which makes it harder for them to differentiate their product lines. Bulk powder lets contract makers change serving amounts based on the age group of the people they are making them for. For example, a chewable tablet for kids might have 250 mg of powder in it, while an immune support plan for adults might have 1,500 mg in each dose. The powder form also lets you make mixed goods where liposomal vitamin C is one part of a mixture of ingredients that helps with a wide range of health issues. Production managers like how powder works with current production processes, whether they are compressing tablets, filling capsules, or packing sticks. It doesn't cost nearly as much to set up as liquid liposomal processing, which needs specialized homogenization tools and cold chain transport infrastructure.
Enhanced Absorption Efficiency: The Core Value Proposition
The main reason formulators are okay with liposomal forms' higher raw material costs is that they are more bioavailable. Standard vitamin C is absorbed at a rate that can't be topped off by SVCT1 transporters in the gut epithelium. When you take more than 1,000 mg, your body gets rid of a lot of the vitamin that it doesn't receive through your kidneys. Liposomal packaging lets substances get absorbed through passive membrane fusion and lymphatic uptake routes that get around these transport mechanisms that get full quickly. Comparative pharmacokinetic studies have tested vitamin C levels in the blood 3–4 hours after administration, finding that liposomal forms have significantly higher area-under-curve values than ascorbic acid doses of the same size. This better delivery speed lets brands make strong marketing claims that are backed up by science, which is why they can charge higher prices. More and more, quality-conscious customers see liposomal delivery as a sign of advanced formulation, which affects their choice to buy in the competitive supplement market.
Shelf Life and Storage Considerations: Supply Chain Benefits
Liposomal vitamin C powder bulk, when added to liquid liposomal products that change with temperature, makes logistics more difficult, which affects landed costs and delivery route choices. Having to be kept cool limits where it can be put in stores and makes delivering online purchases harder, especially in warm places. These problems are taken care of by the stable powder structure. According to our product standards, they need to be stored in cases that keep out light and keep moisture out, but storing them at room temperature is fine too. Because of this stability profile, procurement managers can get better freight rates by moving in normal containers instead of temperature-controlled logistics. The longer shelf life also makes it easier to buy in bulk when prices are low, so buyers can get bigger savings without having to worry too much about running out of stock. Manufacturers that sell to foreign markets prefer the powder form because it makes customs clearance and regulatory reviews easier than liquid forms, which might need more stability data or preservative system reasons.
Conclusion
The market for pharmaceutical-grade liposomal vitamin C powder bulk shows a larger trend toward bioavailability-optimized nutrients that give real bodily benefits. When buying this advanced raw material for businesses, procurement professionals have to look at suppliers on a number of factors, such as their technical abilities, such as particle size control and encapsulation efficiency; their quality systems, which are shown through thorough certifications and testing protocols; their supply chain reliability, which is supported by enough production capacity and inventory management; and their commercial flexibility, which means they can handle different order volumes and customization needs. Dietary supplements, cosmetics, functional foods, medicines, and animal nutrition are the industries that use a lot of bulk. Each of these has its own technical and legal needs that affect where to get the bulk. As the market grows and production methods improve, liposomal vitamin C will probably go from being a high-end specialty ingredient to a common ingredient in many formulations. This will give early user brands the chance to become market leaders.
FAQ1. What differentiates true liposomal powder from simple vitamin C and lecithin blends?
Real liposomal products go through high-shear homogenization or microfluidization, which traps vitamin C molecules in phospholipid bilayers and makes tiny structures called vesicles. Mixing vitamin C and lecithin physically doesn't make them more bioavailable. Make sure that the sellers test the effectiveness of packaging and can give you data on particle size distribution to show that the liposomes are really made of liposomes and not just a bunch of ingredients mixed together.
2. How should manufacturers handle and store bulk liposomal vitamin C powder?
Keep at room temperature in containers with tight lids that are out of the way of light and moisture. Powder forms are more stable than liquid ones, but air can make them clump together and possibly break down the lipid structure. To avoid cross-contamination in places that handle many items, use picking tools that are only used for that purpose. First-expired-first-out (FEFO) rules should be used to rotate goods, but the three-year shelf life gives you plenty of time to get things done.
3. Can liposomal vitamin C powder be combined with other active ingredients in formulations?
Compatibility depends on the ingredients and how they were processed. Most plant products, amino acids, and other water-soluble vitamins can be mixed with the phospholipid parts. During making, keep the liposomes away from strong acids or bases for long periods of time. This could make the structure less stable. Do stability tests on finished recipes to make sure that the vitamin C that is contained stays intact for the whole shelf life of the product when stored the way it was meant to be stored.
Partner with a Trusted Liposomal Vitamin C Powder Bulk Manufacturer
HONGDA Phytochemistry has been extracting plants for more than 25 years and is now working with advanced liposomal ingredients. Our Liposomal Vitamin C Powder Bulk is pharmaceutical-grade and has been tested and certified by third parties to be more than 90% effective at packaging. It also has governmental certifications such as ISO 22000, FSSC 22000, cGMP, HALAL, and KOSHER. As a fully integrated bulk supplier of liposomal vitamin C powder, we keep 15 tons of stock on hand to quickly fill pressing requests. Typical shipping times for global markets are 3–7 business days. Procurement managers like how our MOQ structures are open enough to work with both new names and well-known contract makers. You can email our technical sales team at duke@hongdaherb.com to get product information, prices for the amount you need, and free samples for testing new recipes.
References
1. Padayatty, S.J., et al. "Vitamin C Pharmacokinetics: Implications for Oral and Intravenous Use." Annals of Internal Medicine, vol. 140, no. 7, 2004, pp. 533-537.
2. Davis, J.L., et al. "Liposomal-Encapsulated Ascorbic Acid: Influence on Vitamin C Bioavailability and Capacity to Protect Against Ischemia-Reperfusion Injury." Nutrition and Metabolic Insights, vol. 9, 2016, pp. 25-30.
3. Hickey, S. and Roberts, H. "Ascorbate: The Science of Vitamin C." Lulu Press, 2004.
4. Lykkesfeldt, J. and Tveden-Nyborg, P. "The Pharmacokinetics of Vitamin C." Nutrients, vol. 11, no. 10, 2019.
5. Carr, A.C. and Maggini, S. "Vitamin C and Immune Function." Nutrients, vol. 9, no. 11, 2017.
6. Pullar, J.M., et al. "The Roles of Vitamin C in Skin Health." Nutrients, vol. 9, no. 8, 2017.


_1745918362135.webp)






