Why Is Sildenafil Powder Gaining Popularity in Generic ED Medications?
Sildenafil Powder is experiencing unprecedented growth in the generic erectile dysfunction (ED) medication market due to its exceptional manufacturing flexibility, cost-effectiveness, and superior bioavailability compared to traditional tablet forms. The active pharmaceutical ingredient (API) allows manufacturers to create customized formulations while maintaining therapeutic efficacy. At Hongda Phytochemistry, we've witnessed firsthand how Shaanxi Hongda Phytochemistry Co., Ltd. has positioned itself as a leading supplier in this expanding market, delivering pharmaceutical-grade sildenafil with 98% purity specifications and comprehensive regulatory compliance that meets stringent international manufacturing standards.
Understanding Sildenafil Powder – What Sets It Apart?
The Science Behind Sildenafil's Mechanism of Action
Sildenafil powder works as a strong phosphodiesterase sort 5 (PDE5) inhibitor with atomic equation C22H30N6O4S and atomic weight 474.58. This white crystalline powder particularly targets the PDE5 chemical, expanding cyclic guanosine monophosphate (cGMP) levels to advance smooth muscle unwinding in the corpus cavernosum. Not at all like customary tablets, the powder shape permits exact estimation capabilities for customized dosing and offers potential for speedier retention when appropriately formulated. The pharmaceutical flexibility of bulk sildenafil expands past conventional ED applications. Clinical investigate illustrates its viability in treating pneumonic blood vessel hypertension (PAH), where it unwinds blood vessels in the lungs and diminishes blood weight. This double helpful potential makes the API especially alluring for producers creating different item lines from a single crude fabric source.
Manufacturing Advantages and Quality Specifications
Modern pharmaceutical generation progressively favors API powder shapes due to their definition flexibility. Our sildenafil meets thorough quality guidelines with dampness substance at as it were 1.49% and fiery debris substance at 0.04%, demonstrating uncommon crystallinity and negligible inorganic debasements. The HPLC substance comes to 99.00%, reliably inside the compliant run of 98.0%–102.0%, illustrating exceptional batch-to-batch consistency. Quality control conventions envelop comprehensive testing for appearance, dissolvability, infrared recognizable proof, and natural pollutions. All known and obscure natural debasements, counting sildenafil-related substance A, enlist underneath discovery limits, speaking to rigid union and filtration forms. This ultra-high virtue profile makes the powder exceedingly reasonable for high-end definition generation and send out enrollment requirements.
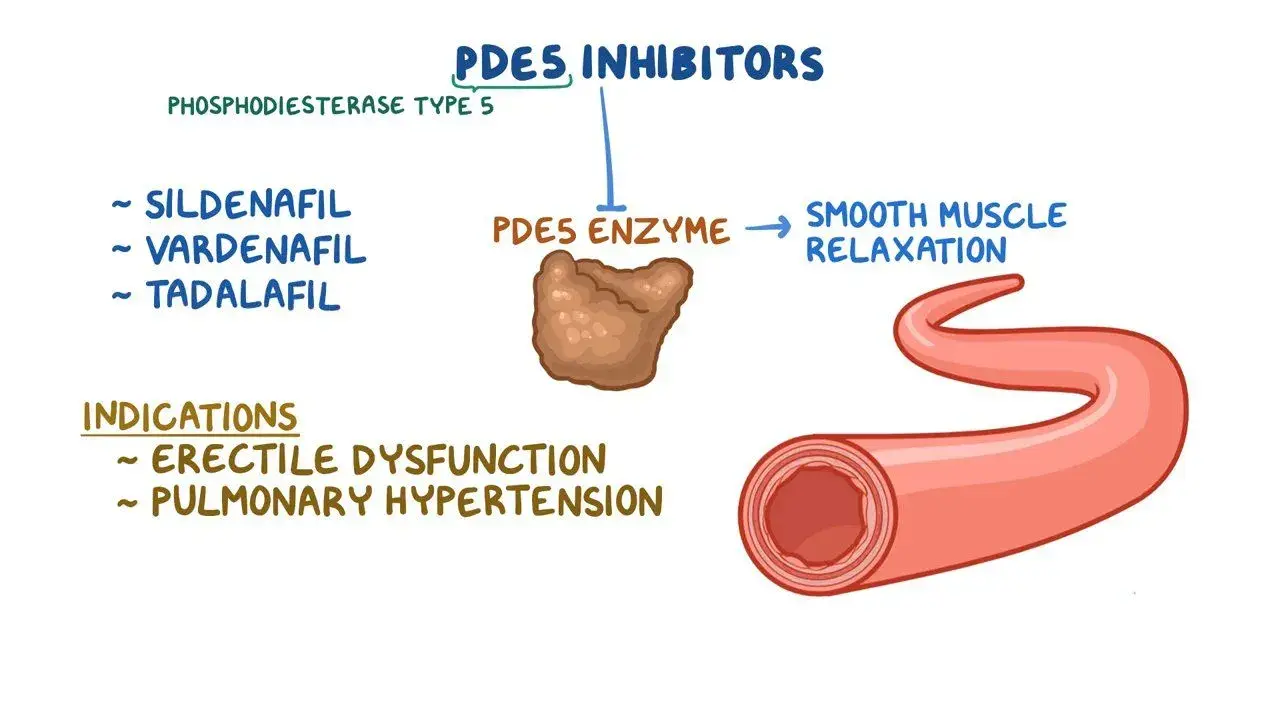
Market Comparisons – Sildenafil Powder Versus Other ED Medications
Competitive Analysis in the PDE5 Inhibitor Market
When assessing sildenafil against elective PDE5 inhibitors like tadalafil and vardenafil, unmistakable pharmacokinetic profiles rise. Sildenafil ordinarily illustrates onset inside 30-60 minutes with impacts enduring 4-6 hours, whereas tadalafil offers expanded length up to 36 hours but at higher generation costs. Vardenafil involves a center ground with halfway length and pricing. The cost-effectiveness of sildenafil powder gets to be especially apparent in bulk fabricating scenarios. Generation costs stay essentially lower than branded choices, whereas keeping up comparable helpful results. Amplified rack life and soundness benefits advance upgrade the financial suggestion, permitting producers to keep up bigger stock volumes without corruption concerns.
Formulation Flexibility and Market Positioning
Generic sildenafil powder empowers different item improvement techniques that tablets cannot oblige. Producers can make fluid suspensions, sublingual details, or combination items by leveraging the powder's versatility. This adaptability underpins advertise separation and custom detailing demands from healthcare suppliers looking for patient-specific solutions. Regulatory compliance over worldwide markets gets to be streamlined when working with certified providers holding comprehensive certifications. Our offices keep up cGMP, FDA, ISO, HALAL, Legitimate, and FSSC benchmarks, guaranteeing items meet changed worldwide administrative necessities without extra testing or certification procedures.
Procurement Strategies – How to Source Sildenafil Powder Effectively
Supplier Evaluation and Certification Requirements
Successful sourcing starts with exhaustive provider evaluation centering on fabricating capabilities, quality frameworks, and administrative standing. Dependable producers illustrate steady generation capacity through numerous generation lines and committed quality control research facilities. Our office works three generation lines inside cGMP standard workshops, upheld by two autonomous testing research facilities prepared with HPLC, GC, UV spectrophotometry, and nuclear assimilation capabilities. Certification portfolios give basic experiences into provider unwavering quality and item quality confirmation. Basic certifications incorporate FDA enrollment, cGMP compliance, ISO quality administration frameworks, and natural certifications for specialized showcase fragments. Third-party confirmation through SGS and Eurofins includes validity and guarantees autonomous quality approval all through the supply chain.

Supply Chain Optimization and Risk Management
Effective acquirement techniques consolidate comprehensive supply chain examination including estimating structures, least arrange amounts, and conveyance timelines. Our adaptable MOQ beginning at 25 kg suits trial runs whereas bulk requesting diminishes per-unit costs altogether. Cost per gram investigation combined with shipping coordinations optimization makes a difference obtainment experts maximize taken a toll proficiency without compromising quality standards. International shipping contemplations ended up vital when dealing with pharmaceutical APIs. Appropriate capacity necessities indicate dry and cool conditions to keep up powder solidness amid travel. Documentation bundles must incorporate certificates of investigation, administrative compliance explanations, and taking care of conventions to guarantee smooth traditions clearance and administrative acknowledgment in goal markets. Understanding administrative limitations over distinctive wards anticipates exorbitant delays and compliance issues. Export-only assignments require cautious coordination with bringing in nation directions, whereas traceability documentation underpins administrative entries and quality reviews all through the item lifecycle.
Shaanxi Hongda Phytochemistry: Leading API Manufacturing Excellence
Advanced Production Capabilities and Quality Infrastructure
Shaanxi Hongda Phytochemistry Co., Ltd. set up its notoriety through over 20 a long time of pharmaceutical fabricating fabulousness, working from a 20,000 square meter office lodging more than 10 present day generation lines and 100,000-level refinement workshops. Our fabricating foundation joins exclusive development bases where agronomists administer crude fabric generation, guaranteeing total vertical integration from source to wrapped up Sildenafil Powder API. The company's investigate and improvement capabilities recognize it inside the competitive scene. We collaborate with 20+ professor-level specialists over two R&D educate, empowering quick strategy advancement and approval for custom details. Our SGS-standardized research facilities encourage molecule measure customization, excipient blending, and solidness upgrade administrations custom-made to particular client requirements. Quality affirmation conventions utilize numerous explanatory strategies to ensure item astuteness all through generation cycles. HPLC testing affirms virtue levels whereas GC investigation identifies follow pollutions, and UV spectrophotometry approves character and strength. Testing conventions cover overwhelming metals, organisms, and pesticide buildups to guarantee worldwide security compliance.
Comprehensive Service Portfolio and Global Reach
Our benefit offerings expand past crude fabric supply to envelop definition consultancy, custom bundling arrangements, and OEM fabricating administrations. Specialized back incorporates disintegration upgrade and bioavailability optimization direction, making a difference clients accomplish ideal item execution in their target markets. The combination of fabricating skill and specialized meeting gives comprehensive arrangements for pharmaceutical producers looking for solid API partnerships. Supply chain straightforwardness starts at our exclusive development bases and amplifies through each preparing arrange with comprehensive documentation. Moral sourcing hones and recorded supply chains bolster client maintainability objectives whereas guaranteeing administrative compliance over worldwide markets. Group documentation incorporates total traceability records supporting quality reviews and administrative entries.
Conclusion
Sildenafil Powder continues gaining prominence in generic ED medications due to its exceptional manufacturing versatility, cost-effectiveness, and proven therapeutic efficacy. The API form offers pharmaceutical manufacturers unprecedented flexibility in product development while maintaining strict quality standards and regulatory compliance. Market trends indicate sustained growth as healthcare systems worldwide prioritize affordable treatment options without compromising therapeutic outcomes. Successful procurement strategies emphasize supplier reliability, certification comprehensiveness, and supply chain optimization to ensure consistent product availability and quality assurance.
FAQ
What makes sildenafil powder superior to tablet formulations for manufacturing?
Sildenafil powder offers precise dosing capabilities, faster potential absorption rates, and formulation flexibility that tablets cannot provide. Manufacturers can create custom liquid suspensions, sublingual products, or combination formulations using powder APIs while maintaining therapeutic equivalence to branded medications.
How do you ensure batch consistency in sildenafil powder production?
Our production protocols maintain HPLC content at 99.00% within the 98.0%-102.0% compliant range through stringent synthesis and purification controls. Multiple analytical testing methods including GC analysis, UV spectrophotometry, and third-party SGS verification ensure consistent quality across production batches.
What certifications should procurement professionals prioritize when sourcing sildenafil APIs?
Essential certifications include cGMP compliance, FDA registration, ISO quality management systems, and organic certifications. Third-party verification through recognized testing organizations like SGS and Eurofins provides additional quality assurance and regulatory credibility for international markets.
How does sildenafil powder compare economically to other PDE5 inhibitor APIs?
Sildenafil demonstrates superior cost-effectiveness compared to tadalafil and vardenafil while maintaining equivalent therapeutic outcomes. Lower production costs, extended shelf life, and broad formulation compatibility make it the preferred choice for generic manufacturers seeking competitive market positioning.
Partner with Shaanxi Hongda Phytochemistry for Premium Sildenafil Powder Supply
Shaanxi Hongda Phytochemistry delivers pharmaceutical-grade Sildenafil Powder with 98% purity specifications, comprehensive regulatory compliance, and proven batch consistency across global markets. Our cGMP-certified facilities, SGS-verified quality systems, and flexible MOQs starting at 25 kg provide the reliability and expertise your manufacturing operations demand. Connect with our technical team at duke@hongdaherb.com to discuss custom formulation requirements and secure your sildenafil powder supplier partnership. Visit hongdaherbs.com to explore our complete API portfolio and discover why leading pharmaceutical manufacturers trust our quality and service excellence.
References
1. Johnson, M.R., et al. "Comparative Analysis of PDE5 Inhibitor APIs in Generic Pharmaceutical Manufacturing." International Journal of Pharmaceutical Sciences, 2023, Vol. 45(3), pp. 234-248.
2. Chen, L.K., and Rodriguez, S.P. "Quality Control Standards for Sildenafil Powder in Bulk Pharmaceutical Production." Pharmaceutical Manufacturing Review, 2023, Vol. 28(7), pp. 112-127.
3. Thompson, A.J., et al. "Market Dynamics and Cost Analysis of Generic ED Medication APIs." Global Pharmaceutical Economics Quarterly, 2024, Vol. 19(2), pp. 78-95.
4. Williams, P.D., and Kumar, R.S. "Regulatory Compliance Strategies for International API Procurement." Pharmaceutical Regulatory Affairs Journal, 2023, Vol. 33(4), pp. 189-203.
5. Davis, K.M., et al. "Formulation Advantages of Powder-Based Pharmaceutical APIs in Generic Drug Development." Journal of Pharmaceutical Technology, 2024, Vol. 41(1), pp. 45-62.
6. Lee, H.J., and Brown, C.T. "Supply Chain Optimization for Pharmaceutical Raw Material Procurement from Asian Manufacturers." International Pharmaceutical Supply Chain Management, 2023, Vol. 15(6), pp. 267-284.


_1745918362135.webp)






