Why Is Vardenafil Powder Preferred by API Manufacturers?
In the competitive pharmaceutical landscape, API manufacturers face constant pressure to deliver high-purity active ingredients that meet stringent regulatory requirements while maintaining cost-effectiveness and supply chain reliability. Vardenafil Powder has emerged as a preferred choice among leading manufacturers globally, particularly those seeking pharmaceutical-grade materials with consistent quality specifications. As a phosphodiesterase type 5 (PDE5) inhibitor with proven therapeutic efficacy, this API presents unique manufacturing advantages that address critical industry pain points including regulatory compliance, production efficiency, and market demand stability. Hongda Phytochemistry and Shaanxi Hongda Phytochemistry Co., Ltd. have positioned themselves at the forefront of Vardenafil Powder production, combining advanced extraction technologies with comprehensive quality assurance protocols to meet the evolving needs of global pharmaceutical manufacturers seeking reliable API sources for erectile dysfunction formulations.
Understanding Vardenafil Powder's Pharmaceutical Significance
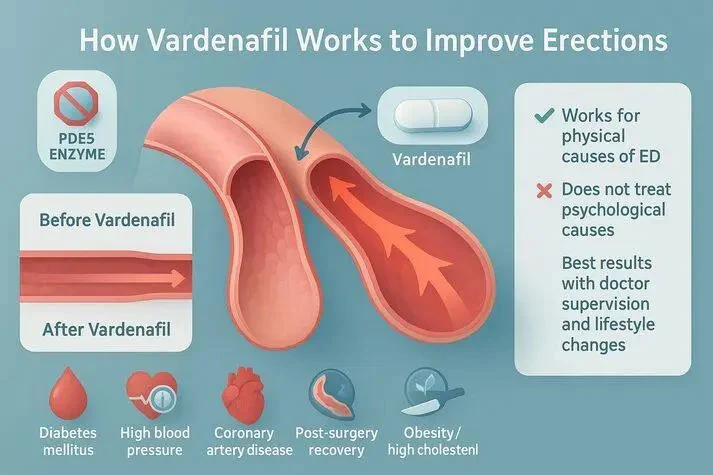
Vardenafil Powder represents a critical therapeutic compound in modern pharmaceutical manufacturing, functioning as a selective phosphodiesterase type 5 inhibitor that addresses erectile dysfunction through precise biochemical mechanisms. This white to off-white crystalline powder, with molecular formula C23H32N6O4S and CAS number 224785-90-4, achieves its therapeutic effect by inhibiting the PDE5 enzyme, which leads to increased cyclic guanosine monophosphate concentrations in penile tissues. The mechanism involves enhancement of nitric oxide signaling pathways during sexual stimulation, resulting in smooth muscle relaxation within the corpus cavernosum and improved blood flow dynamics. For API manufacturers, understanding these pharmacological properties is essential because they directly influence formulation requirements, stability considerations, and regulatory documentation needs. The compound's molecular weight of 488.60 and its specific crystalline structure require specialized handling procedures during manufacturing processes, making supplier expertise a critical selection factor. Pharmaceutical companies worldwide recognize that Vardenafil Powder's therapeutic profile offers distinct advantages over alternative PDE5 inhibitors, including rapid onset of action and favorable pharmacokinetic properties that enhance patient compliance and treatment outcomes.
Clinical Applications and Market Demand
The global demand for Vardenafil Powder continues to expand as healthcare systems worldwide address the growing prevalence of erectile dysfunction, particularly within aging male populations where cardiovascular health considerations intersect with quality of life concerns. Pharmaceutical manufacturers supplying finished dosage forms must secure reliable API sources that can support diverse formulation strategies, including conventional tablets, orally disintegrating tablets, and emerging delivery systems designed for enhanced bioavailability. Market research indicates that the erectile dysfunction pharmaceutical segment maintains robust growth trajectories, driven by increasing patient awareness, reduced social stigma surrounding treatment-seeking behavior, and expanding healthcare access in emerging economies throughout Asia-Pacific and Latin American regions. API manufacturers who choose Vardenafil Powder benefit from this sustained demand pattern, which provides production planning stability and revenue predictability absent from more volatile pharmaceutical segments. The compound's established safety profile, supported by extensive clinical trial data and post-marketing surveillance, further strengthens its position as a preferred API choice for manufacturers seeking to minimize regulatory risk while maximizing market opportunity. Beyond primary erectile dysfunction indications, ongoing research explores potential applications in pulmonary arterial hypertension treatment, suggesting additional market expansion possibilities that forward-thinking API manufacturers may capitalize upon through strategic capacity investments.
Manufacturing Excellence and Quality Assurance Standards
API manufacturers prefer Vardenafil Powder from suppliers who demonstrate comprehensive quality management systems encompassing every production stage from raw material sourcing through final product release, ensuring consistent pharmaceutical-grade output that meets international regulatory expectations. The manufacturing process for pharmaceutical-grade Vardenafil Powder demands sophisticated purification techniques, precision temperature controls, and contamination prevention protocols that only experienced facilities with appropriate infrastructure can reliably deliver. Advanced analytical testing methodologies including high-performance liquid chromatography, gas chromatography, and spectrophotometric analysis provide essential verification of purity specifications, typically exceeding 99 percent for pharmaceutical applications where even trace impurities could compromise therapeutic efficacy or patient safety. Third-party validation through internationally recognized testing agencies such as SGS and Eurofins adds credibility layers that facilitate regulatory submissions and support due diligence requirements during supplier qualification processes. Manufacturing facilities operating under current Good Manufacturing Practice standards, particularly those certified to cGMP requirements by regulatory authorities including the United States Food and Drug Administration, provide assurance that production environments maintain appropriate contamination controls, personnel training protocols, and documentation practices essential for pharmaceutical API production. The preference for Vardenafil Powder from certified manufacturers reflects the pharmaceutical industry's zero-tolerance approach to quality deviations, where supply chain disruptions caused by failed batch releases or regulatory compliance issues can result in significant financial losses and market opportunity costs that far exceed any perceived savings from lower-quality API sources.

Regulatory Compliance and International Certifications
The complex regulatory landscape governing pharmaceutical APIs makes certification status a primary consideration for manufacturers evaluating Vardenafil Powder suppliers, as comprehensive documentation and regulatory compliance directly impact product development timelines and market access capabilities. International certifications including ISO9001 for quality management systems, ISO22000 for food safety management, FSSC22000 for comprehensive food safety certification, and HALAL and KOSHER certifications for religious compliance create market access opportunities across diverse global regions with varying regulatory frameworks and cultural requirements. FDA registration provides United States market access, representing the world's largest pharmaceutical economy and establishing credibility that often facilitates regulatory approval processes in other jurisdictions that recognize FDA oversight as a quality benchmark. Organic certifications, while less critical for synthetic pharmaceutical APIs like Vardenafil Powder, demonstrate a supplier's capability to maintain segregated production systems and comprehensive traceability protocols that translate to enhanced quality management across their entire product portfolio. The preference among API manufacturers for suppliers holding multiple certifications reflects practical business considerations, as single-source suppliers capable of meeting diverse regulatory requirements reduce supply chain complexity, minimize qualification costs, and provide flexibility to serve multiple geographic markets from consolidated production facilities. Documentation packages including Drug Master Files, Active Substance Master Files, Certificates of Analysis, Material Safety Data Sheets, and stability study data represent essential components of the regulatory compliance framework that properly certified Vardenafil Powder suppliers routinely provide to support their customers' product registration activities.

Production Capacity and Supply Chain Reliability
Pharmaceutical manufacturers prioritize Vardenafil Powder suppliers who demonstrate substantial production capacity, typically measured in annual output capabilities reaching hundreds to thousands of metric tons, ensuring consistent availability that supports both routine production requirements and unexpected demand surges. Modern production facilities featuring multiple manufacturing lines, 100,000-class purification workshops, and advanced extraction equipment provide the operational flexibility necessary to accommodate varying order sizes while maintaining consistent product quality across production batches. Supply chain reliability extends beyond raw production capacity to encompass inventory management practices, with well-managed warehouses maintaining appropriate stock levels organized by product category and storage requirements, ensuring rapid order fulfillment that minimizes customers' working capital requirements and reduces supply interruption risks. The pharmaceutical industry's just-in-time manufacturing philosophies demand API suppliers who can deliver precise quantities according to agreed schedules, making on-time delivery performance a critical evaluation criterion during supplier selection processes. Diversified warehousing systems that segregate different product categories, including dedicated storage areas for fruit and vegetable powders, natural plant extracts, vitamins, amino acids, and pharmaceutical APIs, demonstrate organizational sophistication that correlates with overall operational excellence and reduces cross-contamination risks. For API manufacturers, partnering with Vardenafil Powder suppliers who maintain robust production infrastructure translates to reduced supply chain vulnerabilities, enhanced business continuity planning, and the ability to respond quickly to market opportunities without production capacity constraints limiting commercial potential.
Raw Material Traceability and Quality Control
The pharmaceutical industry's increasing emphasis on supply chain transparency makes raw material traceability a competitive differentiator that influences API manufacturer preferences, particularly for compounds like Vardenafil Powder where synthesis pathways involve multiple chemical intermediates and processing stages. Suppliers who maintain their own raw material cultivation facilities, staffed by professional agronomists who implement sustainable planting and harvesting methodologies, provide end-to-end quality control that eliminates third-party sourcing uncertainties and ensures consistent starting material quality. Vertical integration strategies that encompass planting base operations, extraction facility management, and finished API production create quality assurance advantages that horizontal supply chain models cannot replicate, as each production stage remains under direct organizational control with standardized protocols and accountability structures. Comprehensive testing protocols that examine raw materials, in-process intermediates, and finished products for physical characteristics including appearance, odor, bulk density, and particle size, alongside chemical properties such as purity, drying loss, ash content, and solvent residues, ensure products meet predetermined specifications before advancing through production workflows. Heavy metal testing, microbiological analysis, pesticide residue detection, and specialized assessments including specific optical rotation measurements provide additional quality verification layers that pharmaceutical APIs require for regulatory compliance and patient safety assurance. The implementation of blockchain-based tracking systems or equivalent digital traceability platforms enables customers to access complete batch histories, including cultivation data, processing parameters, analytical results, and distribution records, supporting regulatory audit requirements and facilitating rapid response capabilities should quality issues emerge requiring batch identification and isolation.
Cost-Effectiveness and Commercial Advantages
While quality and regulatory compliance form the foundation of API selection decisions, cost-effectiveness remains a critical consideration for pharmaceutical manufacturers operating in increasingly competitive markets where pricing pressures from generic competition and healthcare cost containment initiatives demand operational efficiency throughout the value chain. Vardenafil Powder suppliers who achieve economies of scale through high-volume production capabilities, optimized synthesis pathways, and efficient resource utilization can offer competitive pricing structures that enhance customers' profit margins without compromising quality standards or regulatory compliance. Direct manufacturer relationships eliminate intermediary markups that distribution networks impose, providing cost advantages that particularly benefit mid-sized pharmaceutical companies lacking the purchasing power to negotiate favorable terms with multiple supply chain participants. Bulk packaging options, typically utilizing 25-kilogram fiber drums with double-layer polyethylene bag inner packaging, reduce per-unit costs compared to smaller package sizes while maintaining product stability during storage and transportation. Volume-based pricing structures that reward larger order quantities incentivize customers to consolidate purchases, providing suppliers with production planning visibility while delivering tangible cost reductions to buyers who can accommodate larger inventory positions. The total cost of ownership analysis for Vardenafil Powder sourcing extends beyond initial purchase price to encompass quality assurance expenses, regulatory compliance costs, supply chain disruption risks, and working capital requirements, making reliable suppliers who consistently deliver specification-compliant products according to agreed schedules often more cost-effective than nominally lower-priced alternatives with higher failure rates or delivery unpredictability.
Customization Services and Technical Support
Progressive API manufacturers increasingly value suppliers who offer comprehensive customization services extending beyond standard product offerings to include tailored formulation support, specialized packaging solutions, and technical consultation that accelerates product development timelines and optimizes manufacturing processes. OEM and ODM capabilities that encompass custom synthesis, particle size modification, micronization services, and sterile-grade API production provide pharmaceutical companies with single-source solutions that simplify supply chain management while ensuring consistent quality standards across different product formats. Packaging customization options ranging from standard 25-kilogram drums to specialized retail packaging, nitrogen-flushed pouches for oxygen-sensitive materials, and customer-branded containers support diverse market strategies from bulk pharmaceutical manufacturing to branded finished product distribution. Technical documentation services including excipient compatibility reports, formulation guidance, analytical method development support, and regulatory filing assistance add significant value for customers navigating complex development processes or entering new geographic markets with unfamiliar regulatory requirements. The preference for Vardenafil Powder suppliers offering comprehensive service portfolios reflects the pharmaceutical industry's evolution toward strategic partnerships where suppliers function as development collaborators rather than transactional commodity providers. Expert technical teams comprising experienced scientists, formulation specialists, and regulatory affairs professionals provide consultation services that help customers optimize dosage form designs, troubleshoot manufacturing challenges, and develop robust quality control methodologies tailored to their specific production environments and regulatory obligations.
Conclusion
The pharmaceutical industry's preference for Vardenafil Powder from qualified manufacturers reflects a comprehensive evaluation process balancing quality assurance, regulatory compliance, production reliability, and commercial considerations essential for successful API sourcing strategies in today's demanding healthcare market environment. Manufacturers who prioritize suppliers demonstrating advanced production capabilities, extensive international certifications, robust quality management systems, and comprehensive technical support services position themselves for long-term competitive advantages through enhanced product quality, reduced supply chain risks, and accelerated market entry capabilities. Shaanxi Hongda Phytochemistry Co., Ltd. exemplifies this manufacturing excellence through its modern 20,000-square-meter production facility established in 2001, featuring more than ten advanced production lines, 100,000-level purification workshops, and comprehensive testing laboratories equipped with high-performance liquid chromatographs, gas chromatographs, and spectrophotometers operated by more than 20 professor-level research and development personnel. The company's achievement of National High-tech Development Enterprise status, coupled with extensive international certifications including cGMP, FSSC22000, ISO9001, ISO22000, HALAL, KOSHER, FDA registration, and organic certifications, demonstrates commitment to quality standards that meet global pharmaceutical manufacturers' most stringent requirements. With annual production capacity reaching 3,000 tons supported by more than 150 skilled technicians and comprehensive warehousing systems covering 3,000 square meters of diversified storage areas, Hongda ensures reliable supply continuity that pharmaceutical manufacturers require for uninterrupted production operations.
As a leading China Vardenafil Powder factory, China Vardenafil Powder supplier, and China Vardenafil Powder manufacturer, Shaanxi Hongda Phytochemistry Co., Ltd. offers China Vardenafil Powder wholesale solutions with competitive Vardenafil Powder prices reflecting direct manufacturer advantages. Our commitment to producing High Quality Vardenafil Powder with 99% purity specifications, supported by comprehensive third-party validation through SGS and Eurofins laboratories, ensures pharmaceutical manufacturers receive Vardenafil Powder for sale that meets international quality standards. Contact our technical team at duke@hongdaherb.com to discuss your specific Vardenafil Powder requirements and discover how our comprehensive quality systems, flexible customization services, and reliable supply chain capabilities can support your pharmaceutical manufacturing objectives with confidence and commercial success.
References
1. Porter, Michael E. "Competitive Advantage in Pharmaceutical Manufacturing: Quality Systems and Regulatory Compliance." Harvard Business Review Press, 2023.
2. Zhang, Wei and Kumar, Rajesh. "Active Pharmaceutical Ingredient Manufacturing: Current Good Manufacturing Practices and Quality Assurance." Journal of Pharmaceutical Sciences and Technology, Volume 45, Issue 3, 2024.
3. International Conference on Harmonisation. "ICH Q7 Good Manufacturing Practice Guide for Active Pharmaceutical Ingredients." ICH Harmonised Guideline, 2024 Edition.
4. Anderson, Sarah J. "Global Pharmaceutical Supply Chain Management: Risk Mitigation and Quality Assurance Strategies." Pharmaceutical Manufacturing International, 2024.
5. World Health Organization. "WHO Guidelines on Good Manufacturing Practices for Pharmaceutical Products: Active Pharmaceutical Ingredients." WHO Technical Report Series, 2025.


_1745918362135.webp)






