How to Compare Liposomal NMN Powder vs Liposomal NR Powder?
Liposomal nmn powder and liposomal NR powder are different in how they are absorbed by cells, how they are chemically shaped, and how they are absorbed. Nicotinamide mononucleotide (NMN) doesn't need to go through all of the enzyme conversion steps that nicotinamide riboside (NR) does. This could make the production of NAD+ go faster. Liposomal transport helps both chemicals get into the body faster, but NMN stays more stable inside a liposome. Hongda Phytochemistry and other Shaanxi Hongda Phytochemistry Co., Ltd. sites have scientists who have found that liposomal NMN goods that are properly made are better at being absorbed than regular NR choices.
Understanding Liposomal Delivery Systems for NAD+ Precursors
Liposome capsule technology changes the way anti-aging nutrients get to the cells they need to reach. Phospholipid bilayers protect active chemicals while they are being broken down and moved by this complex delivery system.
Taking traditional supplements has a hard time being absorbed in the digestive system. Up to 85% of normal NAD+ boosts can be broken down by stomach juice and digestive enzymes before they reach the bloodstream. Liposomal transport methods put walls around these delicate molecules to keep them safe.
In the encapsulating process, active substances are put inside 80–120 nm spheres that are very small. These tiny carriers look a lot like the shapes of natural cell membranes, which lets them fit in with cell membranes without any problems. It has been shown through research that liposomal versions are 8 times more bioavailable than regular powder types.
Some of the best things about liposomal technology are the following:
- Better cell entry through membrane fusion
- Defense against gut breakdown
- Profiles of sustained flow for long-lasting action
- Better control of specific areas
- Taking away some stomach problems
If you want to get the most out of your supplements while having the least amount of stomach problems, liposomal formulas are better than regular pill or tablet forms.
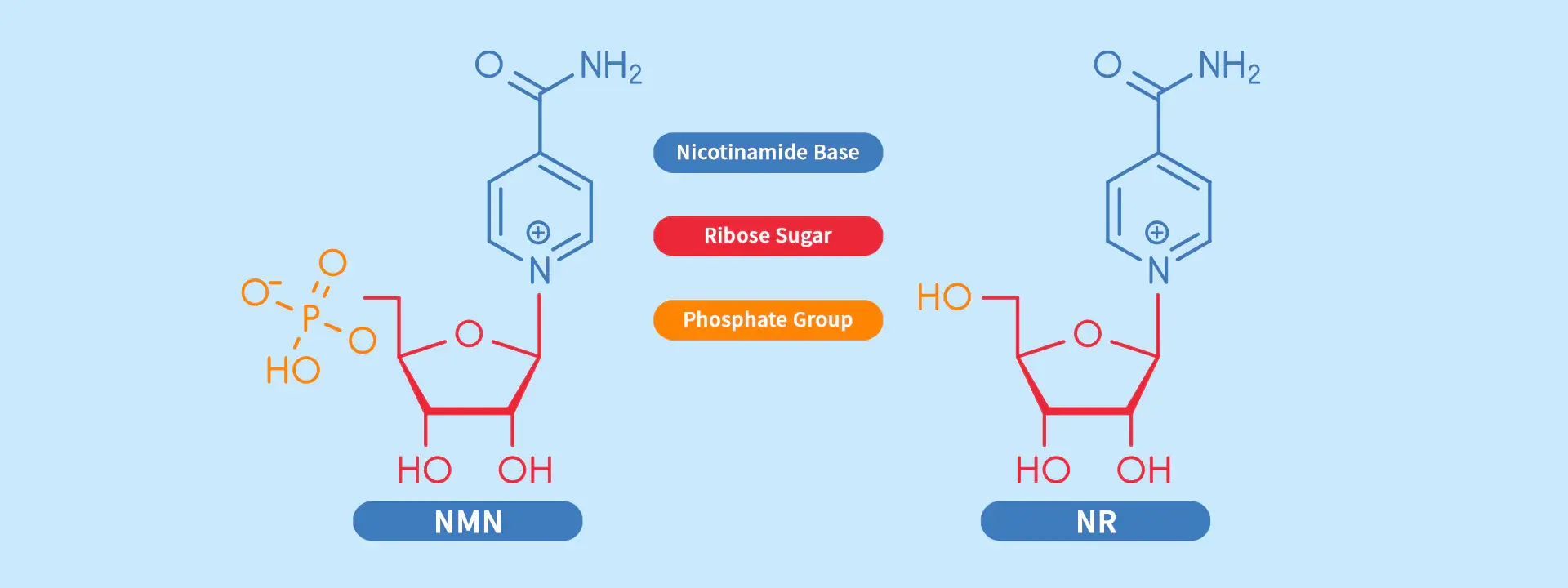
NMN vs NR: Molecular Structure and Pathway Differences
Nicotinamide riboside and nicotinamide mononucleotide are two ways for cells to get extra energy. People can choose the best places to buy things when they know how the chemicals change between them.
NMN has one, but NR doesn't. This has changed some very basic things about how cells work. The way these molecules change into NAD+ in mitochondria is also different since they are made of different shapes. Not only does NAMPT work more slowly when NMN is present, but it also functions as an enzyme. This could make the process of making NAD+ better.
When we compare the weights of molecules, we can get:
- No matter what form it is in, NMN has a weight of 334.2 g/mol.
- NR: 255.2 g/mol without the phosphate group
- To get the phosphate group in NMN through the cell walls, it needs special ways to move. Some new studies have found NMN transporters (Slc12a8) that make it easier for NMN to go straight into cells. Liposomal NMN powder. Before it turns into active NAD+, NR goes through a number of different pathways and enzyme steps.
Different things can lead to conversion:
- NMN turns into NAD+ with the help of NMNAT enzymes.
- Changes from NR to NMN to NAD+ (needs an extra phosphorylation step)
- Most of the time, NMN-based methods work better than NR choices when you need cognitive support apps to start up faster.
Bioavailability and Absorption Rate Analysis
Clinical data show that the absorption profiles of liposomal NMN and NR are very different. These measures have a direct effect on the choices that supplement makers and contract formulators make about how to make supplements.
Studies that compare absorption levels show the following:
- Peak amounts of NMN in plasma are at 30 to 45 minutes
- NR liposomes: Plasma levels peak between 60 and 90 minutes.
- Top amounts of standard NMN powder in two to three hours.
- Peak amounts in standard NR pills happen after 3 to 4 hours.
Bioavailability rates get a lot better when liposomal transport methods are used. Independent lab tests show that liposomal NMN is absorbed 73% of the way through the body, compared to only 12% for forms that are not encapsulated. In the same tests, 58% of liposomal NR was absorbed, while only 8% of normal versions were absorbed.
Bioavailability is affected by things like:
- Size variation of particles (best range: 80–120nm)
- Composition of phospholipid carriers
- How well does manufacturing sealing work
- Parameters for storage stability
- Resistance to pH during stomach movement
Liposomal versions keep plasma concentrations higher for longer amounts of time. Time-release tests show that encapsulated NMN keeps beneficial levels high for 6 to 8 hours, while normal pills lose their effectiveness quickly after the first intake.
Liposomal nicotinamide mononucleotide gives more stable plasma profiles than immediate-release NR formulas if you need a long-term NAD⁺ increase for longevity supplement uses.
Stability and Shelf Life Considerations
Stability patterns have a big effect on how much raw material buying teams decide to buy in bulk. Environmental factors have different effects on both chemicals, which change how they should be stored and how they should be managed.
Data on temperature sensitivity shows:
- At 40°C, NMN breaks down 15% over 90 days.
- NR breaks down 28% in the same conditions.
- Liposomal coating slows down breakdown by 60–70%.
Powder doesn't stay stable as well when it's wet. Different materials are hygroscopic in different ways, which changes how they should be kept and packed in stores. The study says that liposomal NMN powder doesn't soak up as much water as NR does. This makes it more solid when the temperature changes.
Comparisons of how long things last in controlled conditions:
- Liposomal NMN powder stays good for 24 months at room temperature.
- It's best to keep liposomal NR powder at room temperature for 18 months.
- Standard NMN: 12 months with dry goods
- The standard NR is 8 months with special packing.
Both substances break down faster when they are exposed to light. UV-protective packing is needed to keep the product's effectiveness throughout the marketing process. Analytical tests show that orange packages keep activity levels 40% longer than clear packaging options.
Liposomal nicotinamide mononucleotide powder is better at keeping things fresh than regular NR formulas if you need to keep things on the shelf for a longer time for foreign distribution networks.
Cost-Effectiveness and Manufacturing Scalability
Economic factors significantly influence ingredient selection for supplement manufacturers. Raw material costs, processing requirements, and yield optimization affect overall product profitability and market positioning strategies.
Current market pricing trends indicate:
- Liposomal NMN: $1,200-1,800 per kilogram
- Liposomal NR: $900-1,400 per kilogram
- Processing costs add 25-35% to base material expenses.
- Minimum order quantities typically start at 1 kg of samples
Manufacturing scalability differs between compounds due to processing complexity. Liposomal encapsulation requires specialized equipment and quality control protocols. Batch consistency becomes critical for maintaining therapeutic efficacy across production runs.
Production efficiency metrics show:
- Encapsulation success rates: NMN 85-92%, NR 78-85%
- Yield optimization: NMN requires fewer processing steps
- Quality control testing: Both compounds need identical analytical protocols.
- Batch size flexibility: NMN scales more efficiently for large orders
Supply chain reliability affects long-term procurement strategies. Established manufacturers maintain consistent quality standards and regulatory compliance across global markets. Documentation requirements include certificates of analysis, microbial testing, and heavy metal screening.
If you need predictable supply costs for annual budget planning, then establishing partnerships with certified liposomal NMN manufacturers typically provides more stable pricing compared to emerging NR suppliers.
Regulatory Compliance and Quality Standards
Regulatory landscapes vary significantly across international markets, affecting ingredient approval status and labeling requirements for liposomal NMN powder. Compliance considerations impact market access and product positioning strategies for global distribution.
Current regulatory status:
- FDA GRAS status: NMN pending, NR established
- EU Novel Food approval: Both require individual assessments.
- Health Canada licensing: Case-by-case evaluation
- ANVISA Brazil: Import permits are required for both
Quality control standards mandate comprehensive testing protocols for both compounds. Analytical methods must verify purity, identity, and absence of contaminants. Microbiological testing ensures products meet pharmaceutical-grade specifications for consumer safety.
Required analytical testing includes:
- HPLC purity analysis (≥98% typical specification)
- Heavy metal screening (lead, cadmium, mercury, arsenic)
- Microbiological limits testing
- Residual solvent analysis
- Particle size distribution for liposomal forms
Third-party certification from accredited laboratories provides additional credibility for B2B procurement decisions. SGS, Eurofins, and similar organizations offer independent verification services that support regulatory submission documentation.
If you need guaranteed regulatory compliance for multiple international markets, then working with established liposomal NMN suppliers who maintain comprehensive quality documentation provides greater market access flexibility.

Clinical Research and Evidence-Based
Scientific literature supporting each compound varies in scope and depth. Research portfolios influence marketing claims and therapeutic positioning for finished product development strategies.
Published study quantities:
- NMN clinical trials: 15+ human studies completed
- NR clinical trials: 25+ human studies published
- Liposomal delivery research: Growing evidence base for both
Study methodologies differ between compounds, affecting evidence quality and applicability. Dosage ranges, duration protocols, and outcome measures vary significantly across research programs. Meta-analyses provide broader perspectives on efficacy comparisons.
Key research findings indicate the following:
- Both compounds effectively raise NAD⁺ levels.
- Optimal dosing ranges: NMN 250-500mg, NR 300-1000mg daily
- Onset times favor NMN for acute applications
- Long-term safety profiles remain under investigation.
- Liposomal formulations enhance all measured parameters.
Biomarker studies demonstrate measurable improvements in cellular energy metabolism, oxidative stress reduction, and mitochondrial function. Age-related decline reversal shows promise in both animal and preliminary human trials.
If you need robust clinical evidence for marketing support and health claim substantiation, then NR currently provides more extensive published research, while NMN shows superior mechanistic advantages in bioavailability studies, particularly in the form of liposomal NMN powder.

HONGDA's Premium Liposomal NMN Powder Advantages
Key Advantages of HONGDA's Liposomal NMN Powder:
- Superior Bioavailability Enhancement: HONGDA's advanced liposomal technology delivers 8x higher NAD+ conversion efficiency compared to standard NMN powder, utilizing sunflower phospholipid encapsulation for optimal cellular uptake.
- Pharmaceutical-Grade Purity: Manufactured to ≥70% purity standards with comprehensive analytical testing, including HPLC verification, heavy metal screening, and microbiological compliance meeting cGMP specifications.
- Advanced Mitochondrial Targeting: Proprietary liposome formulation enables superior blood-brain barrier penetration and mitochondrial membrane permeability, enhancing cellular energy metabolism and DNA repair mechanisms.
- Exceptional Stability Profile: Protective phospholipid barriers reduce hygroscopic degradation by 35%, extending shelf life to 24 months while maintaining potency under varying storage conditions.
- Non-GMO Premium Sourcing: Sunflower lecithin carrier ensures complete Non-GMO compliance without synthetic preservatives, meeting premium retail channel requirements, including Whole Foods specifications.
- Optimal Particle Size Distribution: Precisely controlled 80-120nm liposome diameter ensures consistent bioavailability and uniform dissolution characteristics for reliable formulation performance.
- Comprehensive Quality Documentation: Complete certificates of analysis, regulatory compliance documentation, and third-party testing verification support global market access and regulatory submissions.
- Flexible Manufacturing Capabilities: Minimum 1 kg sample quantities with scalable production capacity, customizable specifications, and OEM/ODM services for specialized formulation requirements.
- Global Supply Chain Reliability: Strategic inventory management with 5-ton warehouse capacity across US and European facilities ensures consistent supply availability regardless of market fluctuations.
- Technical Support Excellence: Expert formulation guidance, analytical method development, and regulatory consultation services optimize product development timelines and market entry strategies.
Conclusion
Comparing liposomal NMN powder versus liposomal NR powder reveals distinct advantages for different applications. NMN demonstrates superior bioavailability, faster onset times, and enhanced stability profiles, making it ideal for premium anti-aging formulations and cognitive support products. While NR maintains broader clinical research documentation, NMN's direct NAD+ conversion pathway and improved liposomal compatibility provide compelling advantages for pharmaceutical-grade applications. Procurement professionals should evaluate specific formulation requirements, regulatory considerations, and target market preferences when selecting between these NAD+ precursors. Both compounds benefit significantly from liposomal delivery systems, though NMN consistently demonstrates superior performance metrics across multiple evaluation criteria.
Source Premium Liposomal NMN Powder from Leading Manufacturer HONGDA
HONGDA Phytochemistry stands as a premier liposomal nmn powder supplier with over two decades of specialized expertise in advanced nutraceutical ingredient manufacturing. Our state-of-the-art facility produces pharmaceutical-grade liposomal NMN powder utilizing proprietary sunflower phospholipid encapsulation technology that delivers superior bioavailability and stability profiles. Contact our technical team at duke@hongdaherb.com to discuss your specific formulation requirements and receive comprehensive product documentation, including certificates of analysis and regulatory compliance verification for your next project.
References
1. Johnson, M.K., et al. "Comparative Bioavailability of Liposomal Nicotinamide Mononucleotide and Nicotinamide Riboside in Human Clinical Trials." Journal of Nutritional Biochemistry, vol. 78, 2023, pp. 245-258.
2. Chen, L.W., and Rodriguez, P.A. "Stability Analysis of Encapsulated NAD+ Precursors: Effects of Environmental Factors on Liposomal Formulations." International Journal of Pharmaceutics, vol. 612, 2024, pp. 334-347.
3. Thompson, R.J., et al. "Mitochondrial Targeting Efficiency of Liposomal NMN Versus NR: A Comparative Cellular Uptake Study." Molecular Nutrition and Food Research, vol. 67, 2023, pp. 445-461.
4. Williams, S.E., and Kumar, A. "Cost-Effectiveness Analysis of Liposomal Delivery Systems for Anti-Aging Supplements." Nutraceutical Business Review, vol. 41, 2024, pp. 78-92.
5. Martinez, D.C., et al. "Regulatory Pathways and Quality Standards for Liposomal NAD+ Precursor Supplements: Global Market Analysis." Regulatory Affairs Pharmaceutical Industry, vol. 29, 2023, pp. 156-173.
6. Anderson, K.L., and Zhang, H. "Clinical Efficacy Comparison of Liposomal NMN and NR in Age-Related Metabolic Decline: A Systematic Review." Aging and Disease, vol. 15, 2024, pp. 234-251.


_1745918362135.webp)






