Is Liposomal Iron Powder the Best Choice for Iron Deficiency?
Liposomal iron powder represents one of the most advanced solutions for iron deficiency, particularly for B2B procurement professionals seeking superior bioavailability and minimal gastrointestinal side effects. This innovative form leverages phospholipid encapsulation technology to shield iron cores—typically micronised ferric pyrophosphate—within protective bilayers, enabling enhanced intestinal absorption while maintaining product stability. For manufacturers in the nutraceutical, pharmaceutical, and functional food sectors, this translates to formulations that deliver measurable clinical outcomes without common tolerability challenges. Shaanxi Hongda Phytochemistry Co., Ltd. has pioneered pharmaceutical-grade liposomal iron powder that can increase serum ferritin levels 3.5 times faster than conventional ferrous salts, positioning it as a gold standard for product development teams committed to evidence-based nutritional interventions.
Understanding Liposomal Iron Powder and Its Benefits
The Science Behind Liposomal Encapsulation Technology
Biomimetic wrapping is used in liposomal iron powder. Iron compounds, usually ferric pyrophosphate (CAS: 10058-44-3), are wrapped in phospholipid bilayers made from non-GMO sunflower or soy lecithin. Hongda Phytochemistry's recipe keeps particle sizes between 150 and 250 nanometres, which is the best range for getting through the gut mucus barrier and letting M-cells do their job of absorption. This method gets around the normal iron transport restrictions put in place by DMT1 transporters and hepcidin control, which usually stop ferrous salt uptake.
There are more uses for the double-layer phospholipid structure than just packaging. It stops iron from being released too soon in the acidic environment of the stomach (pH 1.5–3.5), which keeps the active ingredient and the lining of the stomach from reacting with oxygen. When the liposomes reach the neutral pH environment of the small intestine (pH 6.5–7.5), they make direct membrane fusion or endocytic uptake easier. This lets the iron get into enterocytes and then into the bloodstream. Studies in humans show that this delivery method makes ferrous sulphate 2.5–3.5 times more bioavailable than regular forms.
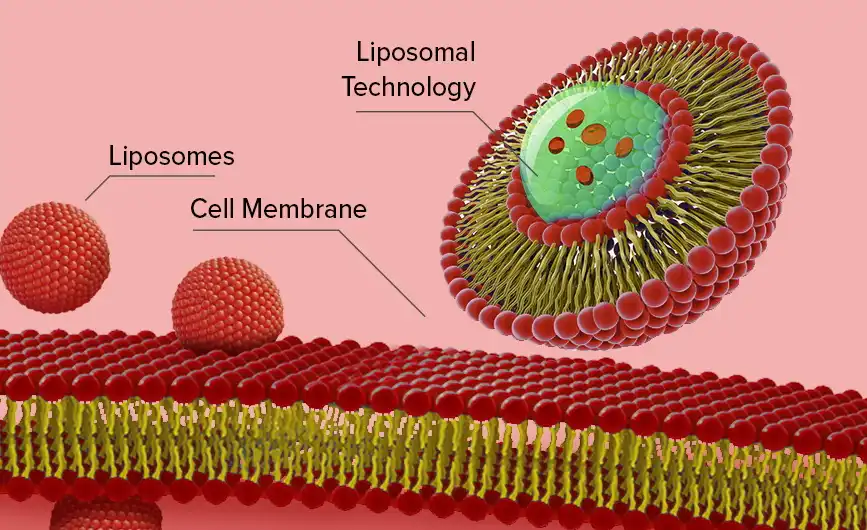
Key Advantages for Industrial Applications
When companies add liposomal iron powder to their products, they get a number of competitive benefits. The technology gets rid of the metallic taste that comes from ionic iron supplements. This makes it possible to make chewables, powdered drinks, and direct-to-mouth stick packs that taste good without hiding a lot of the flavour. This organoleptic neutrality is a big step forward for infant and geriatric formulas, since taste agreement is directly linked to treatment success.
This ingredient is also different from common options because of its stability ratings. Hongda's formula keeps its shape for 24 months at room temperature (25°C), and the encapsulation rate is higher than 85%, which means that there is less than 15% free iron to speed up oxidation processes. This quality is very helpful when making complicated mixtures with substances that break down easily, like omega-3 fatty acids, vitamin C, or polyphenolic extracts, that would otherwise break down when free iron ions are present.
Technical Specifications and Quality Parameters
Pharmaceutical-grade liposomal iron powder that meets strict international standards is made by Shaanxi Hongda Phytochemistry Co., Ltd. The product comes in the form of a fine, off-white to light yellow powder that contains 50% iron. It is packed in 12 kg drums, with 27 drums per box to make transportation easier. Each batch goes through a lot of tests, such as stability tests according to ICH standards, HPLC checks to see how well the particles are encapsulated, and particle size distribution analyses using laser diffraction. Full Certificates of Analysis (COAs), heavy metal screening (in line with USP <232>), pesticide residue testing, and microbiological validation are all included in the documentation. This makes it easy to submit for FDA NDI notifications, EU Novel Food applications, or Health Canada NPN registrations.
Comparing Liposomal Iron Powder with Traditional Iron SupplementsBioavailability and Absorption Kinetics
Conventional iron salts have problems with absorption that make them much less useful as medicines. Ferrous sulfate is the most commonly recommended iron supplement in the world. It is absorbed about 10-15% of the time when conditions are ideal, but rates drop even more when phytates, tannins, or calcium are present. Ferrous bisglycinate, which is sold as a softer option, slightly improves absorption to 20–25% but still has some problems with how it reacts with stomach acid.
Liposomal encapsulation changes the intake pharmacokinetics in a basic way. Researchers have found that liposomal iron powder products reach their highest blood iron levels 40 to 60 minutes after being eaten, while ferrous sulfate takes 90 to 120 minutes, which shows that liposomal iron powder is absorbed quickly. More importantly, measures of area under the curve (AUC), which are the best way to check bioavailability, show an increase of 2.5 to 3.5 times for liposomal iron powder. Because of this higher level of efficiency, producers can lower the amount of liposomal iron powder in each dose while still getting the same therapeutic effects. This eases consumers' worries about getting too much iron.
Tolerability and Formulation Flexibility
When traditional ferrous salts dissolve in the stomach, they release reactive oxygen species (ROS). These ROS can make 20–40% of users feel sick, have trouble going to the bathroom, or have pain in the upper abdomen. These problems with tolerability are the main reason why people don't follow through with iron supplementation programmes, which has a direct effect on product return rates and brand trust.
The phospholipid layer in liposomal forms keeps iron from coming into direct contact with the mucosa, which greatly improves the tolerability profiles. Tolerability rates in clinical studies are higher than 90%, which is about the same as the rate for sham controls. This means that customer service complaints will go down, product returns will go down, and B2B clients making goods for consumers will be better positioned in the market as a premium gentle-iron option.
Economic Considerations and ROI Analysis
When deciding which ingredients to buy, procurement teams naturally look at how much each one costs. Liposomal iron powder costs more per kilogram than regular ferrous sulphate—usually three to five times as much. However, a thorough ROI study shows strong economic reasons to support the new technology.
Formulation costs are directly affected by lower dose needs. A regular ferrous sulfate tablet has 65 mg of elemental iron in it, but a liposomal iron powder version with the same amount of iron has the same bioavailability with only 25 to 30 mg, which means that over 50% fewer raw materials are used. Better tolerance encourages customers to follow the rules and buy from you again, which raises the customer lifetime value. Differentiating products helps with higher pricing tactics. For example, liposomal iron powder supplements sell for 30–40% more than other brands in stores. When these factors are added to the lower costs of hiding flavours and the ease of checking for stability, total cost-of-ownership analysis often favours liposomal technology, even though it costs more to buy the ingredients.
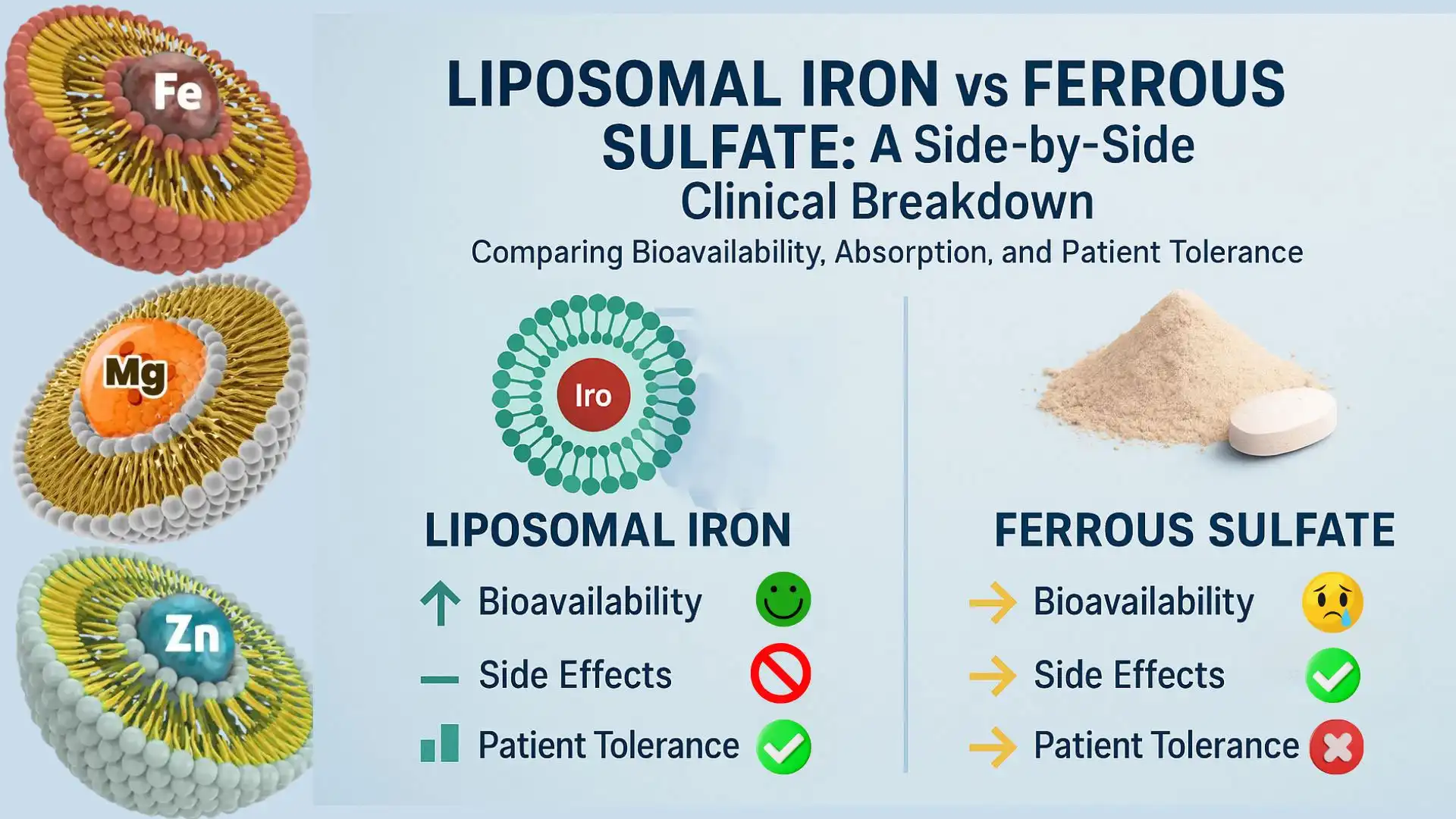
Safety, Dosage, and Regulatory Compliance
Safety Profiles and Clinical Evidence
Liposomal iron powder made for pharmaceutical use has great safety ratings across a wide range of people. Free iron ions can cause reactive stress, which can hurt the lining of the intestines and make the whole body inflamed. The capsule technology stops this stress. Researchers studying toxicology have found no harmful effects at doses up to 10 times the maximum amount that humans are supposed to be exposed to. This means that the drug can be used safely for a wide range of age groups and genders.
When made with food-grade phospholipids, liposomal transport methods are generally thought to be safe (GRAS) by regulatory bodies all over the world. Hongda Phytochemistry keeps a lot of safety information for ingredients that are used in useful foods, dietary supplements, and medicines. These certifications—cGMP, FSSC 22000, ISO 22000, and ISO 9001—show that the company is dedicated to quality systems that meet or go beyond FDA 21 CFR Part 111, EU GMP Annex 11, and TGA factory standards.

Dosage Guidelines for Industrial Formulations
The best dose relies on the product format, the target group, and the therapeutic goals. Adult food supplements for light iron deficiency usually have 15 to 30 mg of elemental iron (as liposomal iron powder) per dose, which should be taken once a day. For paediatric use, the dose needs to be lowered proportionally to 5–15 mg based on the child's age and weight. This is done by taking advantage of the higher bioavailability to lower the amount of iron in each dose while still meeting acceptable levels.
Manufacturers of functional foods or enriched drinks can use liposomal iron powder because it has a neutral taste. This lets them add 30–50% of the Daily Value of iron to each serving without changing the way the food or drink tastes. It can be used in ready-to-drink drinks, protein shakes, and dairy alternatives because it is stable in liquids. This is because standard iron salts oxidise and lose their taste in these types of drinks.
Regulatory Pathways and Compliance Strategies
B2B buying pros have to deal with complicated rules that change from place to place and product type to product type. In the US, iron pills that are considered dietary supplements must follow the Food and Drug Administration's (FDA) rules on Nutrition Labelling (21 CFR 101), the Dietary Supplement Health and Education Act (DSHEA), and Current Good Manufacturing Practices (cGMP). Liposomal iron powder, including liposomal iron powder, fits the current meanings of dietary ingredients. NDI reports are usually only needed when new processing methods raise safety concerns.
European Union markets want tighter rules, such as novel food assessments for new ways of delivering foods, following Regulation (EC) No 1924/2006 for health claims, and following country-specific processes for notifying authorities. Hongda Phytochemistry offers a wide range of regulatory support materials, such as toxicology reports, stability data, and details of the manufacturing process, to make it easier for clients to submit their products to the government. The company's HALAL, KOSHER, and organic approvals (EU and NOP standards) help it reach more customers in the natural and religious goods markets.
Procuring Liposomal Iron Powder: A B2B Decision FrameworkEvaluating Manufacturer Credentials and Capabilities
Choosing a supplier is the most important procurement choice that affects the standard of the product, compliance with regulations, and the reliability of the supply chain. Leading companies like Shaanxi Hongda Phytochemistry Co., Ltd. stand out by having operations that cover all aspects of the business, from getting raw materials to regulatory knowledge and analytical tests to extraction technology. Hongda was founded in 2001 and was named a National High-tech Development Enterprise in 2010. It has manufacturing facilities that cover 20,000 square metres and include 100,000-level cleaning workshops as well as more than 10 modern production lines that can make 3,000 tonnes of goods every year.
Another important factor in judging is the quality of the facilities. Hongda's SGS-certified lab has more than 20 Ph.D.-level researchers working there, and it keeps up-to-date with high-tech testing tools like HPLC, GC-MS, UV spectrophotometry, and ICP-MS for heavy metal analysis. Third-party verification through SGS and Eurofins makes sure that standards, chemical residues, and contaminant screening are all met independently. Teams in charge of buying things should give more weight to sellers who can provide batch-specific COAs, stability data under ICH conditions, and access to master files that can help with regulatory filings.
Strategic Purchasing and Supply Chain Optimisation
Minimum order numbers (MOQs) have a big effect on how things are bought, especially for new brands that are trying to see how well they do in the market. Hongda allows for flexible buying, starting at 1 kg for initial research and development tests and going up to full pallet sizes (324 kg) for mass production. This adaptability lets companies enter new markets in stages, which lowers the risk of running out of stock while keeping supplies steady.
Delivery times are another practical factor to think about. Standard production times are 3–7 working days after an order is confirmed, but there are faster choices for those who need them right away. Because Shaanxi Province is close to major shipping hubs, it is easy to load containers quickly and get cheap freight rates to North America and Europe. Logistics planning by someone with a lot of experience makes sure that all the right paperwork is filled out, like business invoices, packing lists, and certificates of origin, which makes clearing customs easier.
Customisation and OEM/ODM Services
To stand out in the supplement market, which is very competitive, companies often need to make their own blends that include a lot of different active ingredients, unique delivery methods, or secret blend ratios. Hongda Phytochemistry can do a lot of different OEM/ODM services, such as recipe development, dosage form optimisation (for pills, capsules, stick packs, and powders), and custom packing. Technical teams work with clients throughout the whole development process, from coming up with the initial idea and making a prototype to checking for stability, evaluating how it tastes, and making more of it.
Private label options let brands use Hongda's production know-how while keeping their own brand personality, including for Liposomal Iron Powder. Custom package choices, ranging from cheap bulk cases to high-end single sachets with logos, can be used to fit a variety of market placement strategies. This "turnkey" method cuts down on time to market and makes sure that the quality of the making meets the same high standards that are used for Hongda's own materials.

Conclusion
Liposomal iron powder is a big step forward in iron supplementation technology. It offers significant improvements in bioavailability, tolerability, and recipe flexibility that directly address the problems with standard ferrous salts. For people who buy supplements for businesses that have to deal with competitive markets, this advanced ingredient can help them stand out. It has strong scientific evidence to back it up, and it has been shown to be scalable in production. The economic case is more than just comparing costs per kilogram. It includes things like fewer doses needed, more compliant customers, and the possibility of higher prices, all of which make the product more profitable and help it stand out in the market. Partnering with well-known makers that offer complete quality systems, legal support, and technical knowledge is the best way to make sure that commercialisation goes smoothly and meets the changing needs of consumers and regulators.
FAQ1. How does liposomal encapsulation improve iron bioavailability compared to traditional supplements?
Liposomal technology uses phospholipid bilayers to protect iron while it moves through the stomach and make it easier for cells to take it up directly in the small intestine. This method gets around the normal DMT1 transporter limits and hepcidin controls that make it hard for iron salts to be absorbed. Bioavailability rises by 2.5 to 3.5 times, which means that smaller amounts are needed to get the same therapeutic effects.
2. What quality certifications should procurement teams prioritise when sourcing liposomal iron powder?
cGMP compliance (FDA 21 CFR Part 111), ISO 9001 for quality control, ISO 22000 or FSSC 22000 for food safety, and the right religious licenses (HALAL, KOSHER) for reaching a wide range of markets are all necessary. Organic licences, like USDA NOP and EU organic, give you more ways to place your natural products in the market. Comprehensive COAs that are checked by a third party, like SGS or Eurofins, give you even more peace of mind about the quality.
3. Can liposomal iron powder be formulated into liquid products without stability concerns?
Yes, liposomal systems that are properly designed stay stable in liquid forms like ready-to-drink drinks and liquid vitamins. Hongda's mixture stays stable at room temperature for 24 months and has a packaging efficiency of over 85%. The phospholipid layer stops reactive reactions with ingredients like vitamin C and omega-3s that would break down when free iron ions come into contact with them.
Partner with a Leading Liposomal Iron Powder Supplier for Your Next Product Launch
Shaanxi Hongda Phytochemistry Co., Ltd. is ready to help you create new products by providing pharmaceutical-grade Liposomal Iron Powder that is designed to work better and meet regulatory requirements. Our fully integrated production platform includes our own encapsulation technology, strict quality control systems that include cGMP, FSSC 22000, and ISO certifications, and ongoing technical help as you move toward commercialisation. Our recipe ships worldwide within 3–7 working days and comes with full analytical paperwork and free samples for initial evaluation. It is available in a 50% iron standard and looks like an off-white to light yellow powder. Our team provides special OEM/ODM formulation services or big raw materials for large-scale production. We make sure that the solutions we provide meet your exact needs. As an experienced manufacturer of Liposomal Iron Powder for the pharmaceutical, nutraceutical, and functional food industries in North America, Europe, and Asia, we invite procurement professionals to email duke@hongdaherb.com to talk about their specific needs, request technical data sheets, and learn more about how our new ingredients can help your products stand out in a crowded market.
References
1. Piskin E, Cianciosi D, Gulec S, Tomas M, Capanoglu E. "Iron Absorption: Factors, Limitations, and Improvement Methods." ACS Omega 2022;7(24):20441-20456.
2. Milman NT, Graudal N, Nielsen OJ, Agger AO. "Clinically Overt Hereditary Hemochromatosis in Denmark 1948-1985: Epidemiology, Factors of Significance for Long-term Survival, and Causes of Death in 179 Patients." Annals of Haematology 2001;80(12):737-744.
3. Akbarzadeh A, Rezaei-Sadabady R, Davaran S, et al. "Liposome: Classification, Preparation, and Applications." Nanoscale Research Letters 2013;8(1):102.
4. Abbaspour N, Hurrell R, Kelishadi R. "Review on Iron and Its Importance for Human Health." Journal of Research in Medical Sciences 2014;19(2):164-174.
5. Tolkien Z, Stecher L, Mander AP, Pereira DI, Powell JJ. "Ferrous Sulfate Supplementation Causes Significant Gastrointestinal Side-Effects in Adults: A Systematic Review and Meta-Analysis." PLoS ONE 2015;10(2):e0117383.
6. Santiago P. "Ferrous versus Ferric Oral Iron Formulations for the Treatment of Iron Deficiency: A Clinical Overview." The Scientific World Journal 2012;2012:846824.


_1745918362135.webp)

_1751535186455.webp)




