Why OEM Tirzepatide Powder Is Preferred by Global Pharma Companies?
OEM Tirzepatide Powder is becoming more and more popular as a way for pharmaceutical businesses around the world to get this innovative dual GIP/GLP-1 receptor agonist. This choice comes from the need for constant quality assurance, regulatory compliance, and low costs in the metabolic therapeutics market, which is very competitive. Tirzepatide powder, which is 98% pure and has a molecular weight of 4813.00 (CAS NO: 2023788-19-2), is an important active pharmaceutical ingredient (API) for treating diabetes and weight loss. Shaanxi Hongda Phytochemistry Co., Ltd. is a reliable company that has been providing high-quality peptide raw materials to pharmaceutical companies for more than twenty years. These materials help pharmaceutical companies meet strict international standards while keeping prices low.
Understanding Tirzepatide Powder and Its Role in Modern Therapeutics
What Makes Tirzepatide a Unique Therapeutic Agent?
Bulk tirzepatide powder is the first and only drug in the world that stimulates both the glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptors at the same time. It's different from other metabolic disease drugs on the market because it works on two different receptors at the same time. The man-made peptide is made up of an exact chain of amino acids that works like natural incretin hormones. This helps the body better control blood sugar levels and makes you feel full by working on brain areas that control appetite.
Clinical Evidence Supporting Tirzepatide Efficacy
Recently conducted clinical studies have shown that the treatments work, with patients losing more than 10 kilograms of weight and having better metabolic measurements. The activation of two receptors increases the release of glucose-dependent insulin from beta cells in the pancreas while at the same time decreasing the release of glucagon that is too high. When compared to standard treatments, this coordinated system controls blood sugar better.
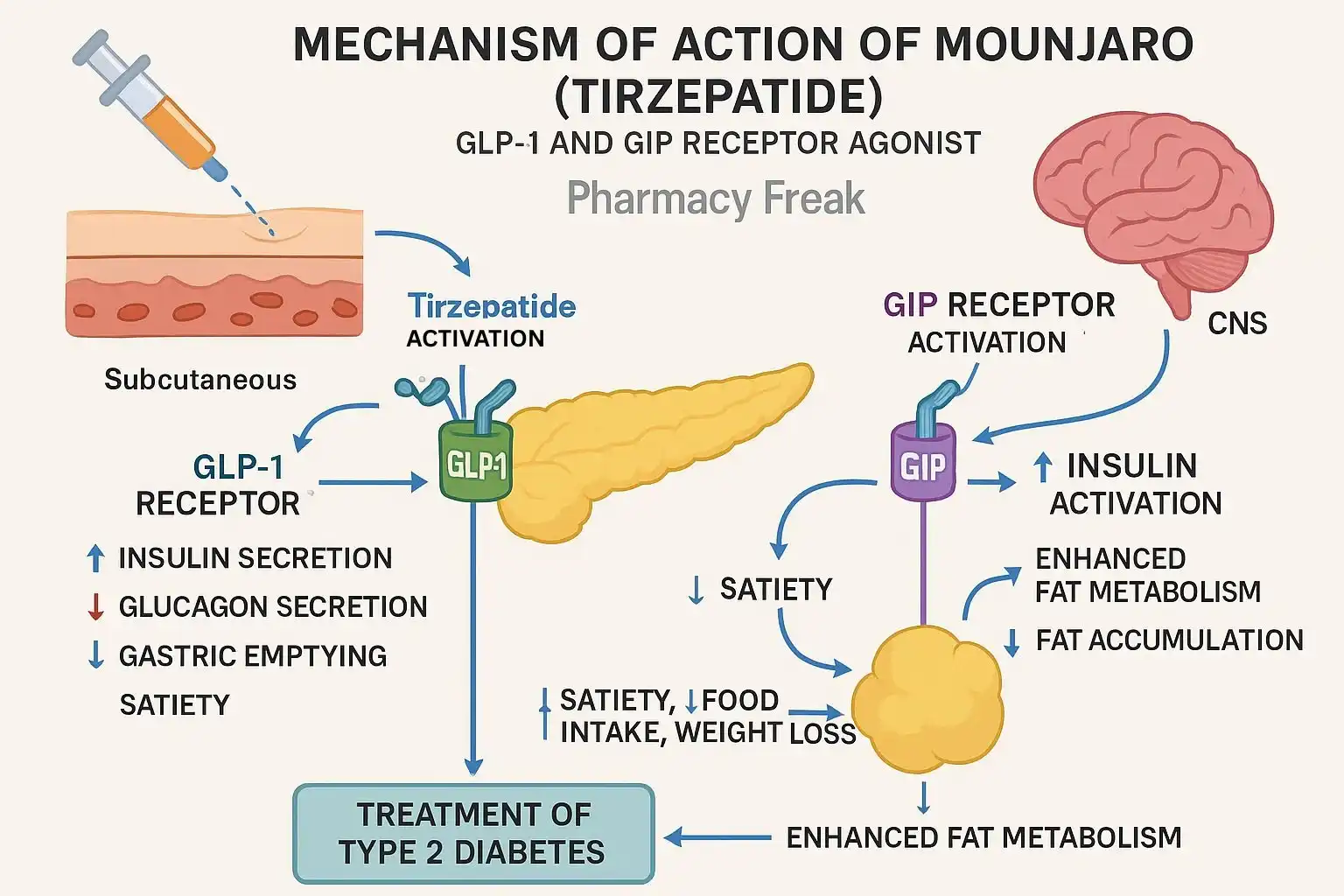
Product Specifications That Matter for Procurement
When procurement workers look for this API, they need to know about some important technical details. A good source of tirzepatide powder bulk is a white to off-white powder that dissolves easily in water with a pH between 6.0 and 9.0. The HPLC test should show that the purity is at least 99%, and any linked chemicals should be kept below 1.0% of the total impurities. To make sure that injectable versions are safe, bacterial endotoxin amounts must stay below 5 EU/mg. These requirements have a direct effect on how well the end product works as a medicine and how safe it is for patients.
Challenges Faced by Pharma Companies in Procuring Tirzepatide Powder
Navigating Complex Regulatory Requirements
Pharmaceutical companies are under more and more pressure to follow changing rules in a number of different countries. The FDA, EMA, and WHO all have different rules about peptide APIs, especially when it comes to how they should be made and how they should keep quality records. Companies that buy Tirzepatide Powder without the right cGMP certification risk being turned down by regulators, having their products take longer to reach the market, and possibly being sued. Checking the certificates of suppliers takes a lot of time and needs to be done carefully by buying teams.
Supply Chain Vulnerabilities and Scalability Concerns
Tirzepatide's peptide synthesis process includes complex chemical steps that need specific tools and technical know-how. A lot of suppliers can't go from making small amounts for study to making a lot of them for sale without losing quality. More and more often, problems in the supply chain happen because of things like a lack of raw materials, delays in shipping, and political unrest that affect delivery times. When pharmaceutical companies release new formulations or grow their market position, they need reliable partners who keep up with multiple buying strategies and keep enough supplies on hand as a buffer.
Quality Inconsistency and Supplier Reliability
Suppliers who don't meet standards pose big problems beyond not following the rules. Changes in purity levels, leftover solvent content, or amino acid ratios from batch to batch can hurt the performance of a product and require expensive reformulation efforts. Whether it's bacterial, chemical, or particle contamination, it's very bad for industry when this happens. Procurement teams can't check that claimed specs are met without full analytical testing methods that include HPLC, LC-MS, and mass spectrometry proof. It is even harder when providers don't have clear paperwork like Certificates of Analysis (CoA), Material Safety Data Sheets (MSDS), and Technical Data Sheets (TDS).

Why Do Global Pharma Companies Prefer OEM Tirzepatide Powder: Core Advantages?
Customised Manufacturing Solutions for Diverse Market Needs
When pharmaceutical firms work with original equipment manufacturers (OEMs), they can change the production factors to meet the needs of their target market and government regulations. Different areas may need different types of packaging, different languages for paperwork, or different quality guarantees. We can handle batches as small as 1 kg for clinical trials and as large as 1,000 kg or more for commercial production. We can also scale up or down quickly to match the stages of a product's lifecycle. Our 12 kg/drum packaging with 27 drums/tray configuration makes operations more efficient while keeping the quality of the product intact during foreign shipping.
Cost Efficiency Through Strategic Partnership Models
Working directly with OEM makers gets rid of the markups that come from going through middlemen and raises the cost of buying. If you buy in bulk and your order is more than 100 kg, you can get big discounts. Long-term supply deals that last between 2 and 5 years offer better price models that make your budget more predictable. Contract production lowers the costs of keeping in-house peptide synthesis capabilities. This lets pharmaceutical companies focus their resources on their main areas of expertise, such as formula development and clinical research. Regional pricing optimisation makes sure that prices are competitive in markets in North America, Asia, Vietnam, and Switzerland.
Reduced Procurement Risk and Enhanced Compliance
When you work with a well-known OEM maker, you can move some supply chain risks while still keeping an eye on quality. Our building has many different types of certificates, such as cGMP, Kosher, Halal, BRC, Organic, FDA register, ISO9001, ISO22000, FSSC22000, and ORGANIC (EU and NOP) compliance. These certificates show that you follow quality management procedures that are organised and meet foreign standards. Regulatory support teams help with making the Drug Master File (DMF) and Certificate of Suitability (CEP) forms, which speed up the clearance process. Because we follow ICH stability standards, we can be sure that goods will stay pure and effective for 24 months.
In addition to certifications, we offer professional help for the whole process of making a product, including Tirzepatide Powder. With over 20 professor-level researchers and testing staff equipped with cutting-edge analysis tools, our team can help with formulation, stability data evaluation, and problem-solving. With this partnership method, relationships with suppliers go from being commercial to being strategic partnerships that help both parties succeed.

Comparing OEM Tirzepatide Powder with Alternative Solutions in the Market
Tirzepatide Versus Traditional Diabetes Treatments
When compared to well-known treatments like insulin shots or single-receptor GLP-1 agonists like semaglutide, Tirzepatide Peptide Powder Bulk does better in more than one area of clinical testing. The dual-receptor process leads to more noticeable weight loss, which is important because fat and type 2 diabetes often happen together. Patients like the once-weekly subcutaneous injection programme because it makes it easier for them to stick to their drug plans than taking medications every day. Because it can be used in many different ways, Tirzepatide is a complete answer for both controlling blood sugar and maintaining digestive health.
OEM-Sourced Material Quality Advantages
Buying through well-known OEM routes ensures access to materials made under controlled conditions that are safe for use in medicine. Our unique peptide synthesis technology makes sure that the sequencing of amino acids is correct and that process-related flaws are kept to a minimum. A multi-stage purity study shows that the HPLC results are higher than 99.5% and that related chemicals are kept below 0.1%, which is much higher than the standard requirements. Verification by mass spectrometry, study of residual solvents using gas chromatography, and endotoxin tests below 0.1 EU/mg all help to make sure the quality is high. This level of analytical rigour backs up regulatory applications and gives people faith in the performance of the end product.
Evaluating Supplier Credentials and Capabilities
To be successful at buying, you need to do more than just compare prices when evaluating suppliers. Make sure that the qualifications of the production facility meet the needs of the market you want to reach. For example, European distribution needs different credentials than North American or Asian distribution. Check the providers' production ability to make sure they can meet your current needs and grow with you in the future without affecting delivery times. With lead times of 3–7 working days for normal orders, our yearly capacity of more than 10 metric tonnes shows that we are ready to go. Look at the transportation infrastructure, especially the cold-chain features that are needed to keep peptides stable during foreign shipping. Ask current pharmacy clients for references and look over audit records from outside inspection firms. These steps help you find trustworthy partners who can help you reach your long-term business goals.
Practical Guide for B2B Procurement of OEM Tirzepatide Powder
Identifying Trustworthy Sourcing Platforms and Verification Methods
Pharmaceutical procurement teams should give more weight to sellers whose manufacturing qualifications can be checked and whose quality paperwork is clear. Working directly with plant reps gives you more information than working with a third-party dealer. The hongdaherbs.com website for our business has a lot of detailed information, standard sheets, and certification downloads that help people make smart decisions. Before placing a big order, ask for samples along with the most recent batch COA data so that you can do your own independent verification tests. Set up ways for technical teams to talk to each other so that they can answer questions about composition and provide regulatory support paperwork.
Understanding Pricing Structures and Negotiation Strategies
The price of tirzepatide powder depends on the cost of raw materials, the difficulty of making it, the money spent on quality control, and changes in market demand. Instead of giving vague bundled prices, transparent providers give thorough quotes that break down the costs of each component. By committing to a certain amount of goods, you can get better prices. For example, sales of 100 kg or more get big savings. Long-term supply deals are good for both parties because they keep prices stable and make sure that capacity is allocated correctly. When negotiating, don't just focus on per-kilogram prices. Instead, talk about the total cost of ownership, which should include things like consistent quality, legal support, and reliable delivery. Building relationships where people work together often gets better results than dealing in a hostile way.
Managing International Logistics and Regulatory Documentation
Customs rules, import permits, and temperature-controlled shipping must all be carefully followed in global pharmaceutical supply lines. Our packaging options use sealed packages with desiccant protection to keep wetness from getting inside while they're being shipped. Cold-chain logistics and constant temperature tracking make sure that peptides stay stable in all kinds of weather. We offer full documentation packages that include business invoices, packing lists, certificates of origin, and regulatory compliance certificates that are specific to the needs of the country where the goods are going. Our established transportation networks in Europe, North America, Asia, Vietnam, and Switzerland allow us to deliver items from door to door in 7 to 15 days. Our 3,000-square-metre warehouse keeps a lot of goods on hand, so there are never any backorders that mess up production plans.
It's easy to get through customs when the paperwork matches the rules in the country. Our skilled export team works with freight forwarders and customs agents to make the complicated import process go smoothly. Tracking along the whole chain of custody lets you see a package from the time it leaves the plant until it arrives at its final destination. This lets you plan ahead for any delays that might happen. With this kind of logistical know-how, foreign purchasing goes from being hard to being a competitive benefit.

Conclusion
FDA-approved OEM Tirzepatide powder is preferred by the pharmaceutical business because it meets quality standards, follows rules, and saves money. This dual GIP/GLP-1 receptor agonist is a big step forward in treating metabolic diseases, but it needs production partners who understand both how complicated science is and how the global pharmaceutical market works. Shaanxi Hongda Phytochemistry Co., Ltd. has been making peptides for more than 20 years and has many certifications, a production capacity that can be scaled up or down, and service models that focus on the needs of each customer. Our dedication to purity levels above 99%, thorough analytical testing, and on-time delivery makes us a reliable partner for pharmaceutical companies, original equipment makers (OEMs), and raw material dealers who want to gain a competitive edge in this rapidly growing therapeutic area.
FAQ1. What distinguishes OEM tirzepatide from commercially available finished products?
OEM Tirzepatide powder is the active pharmaceutical ingredient (API) because it is very pure and comes in its raw form, not in a finished dose form. This lets drug companies make delivery methods, dosage levels, and combination treatments that are specifically designed for groups of patients or to meet legal needs. Before the raw powder is turned into the pre-filled syringe pens that patients can buy, it goes through more steps of processing, such as reconstitution, clean filtration, and aseptic filling.
2. How can I verify the quality standards of an OEM supplier?
Ask for complete analytical records from recognised labs, such as HPLC chromatograms, mass spectrometry data, and certificates of analysis. Check that the seller has the right licences, like cGMP, ISO9001, and FDA registration, by searching an outside database. Do site surveys or hire a third-party inspection service to check the conditions of the factory. Before signing big contracts for purchases, sending samples to independent labs for testing gives you clear proof of quality.
3. What minimum order quantities should I expect when sourcing tirzepatide powder?
Different suppliers have different minimum order numbers, but they are usually between 1 kg and 10 kg or more for business manufacturing. Our production methods are flexible enough to handle orders ranging from small clinical trial runs to business quantities of more than 1,000 kg. When you make a volume promise, you can get better prices and make sure that all batch amounts meet the same high-quality standards.
Partner with a Proven Tirzepatide Powder Manufacturer
Shaanxi Hongda Phytochemistry Co., Ltd. can help you meet your pharmaceutical manufacturing goals by providing you with high-quality OEM Tirzepatide powder. Our manufacturing site is 20,000 square meters and has several modern production lines inside 100,000-level purification workshops. It is a national high-tech business and has many patent technologies. We give your business the stability and compliance it needs with a purity promise of 99% or more, full standards like cGMP and ISO22000, and factory-direct prices that make you more competitive. For every batch, our expert team gives full regulatory support with CoA, MSDS, and TDS paperwork. You can email our procurement experts at duke@hongdaherb.com to ask for samples, talk about your unique needs, or get personalised quotes. Discover the dependability that pharmaceutical businesses around the world rely on when they need to get this important treatment ingredient.
References
1. Wilson JM, Nikooienejad A, Robins DA, Roell WC, Riesmeyer JS, Haupt A, Dellva MA, Barve A. "The dual glucose-dependent insulinotropic peptide and glucagon-like peptide-1 receptor agonist, tirzepatide, improves lipoprotein biomarkers associated with insulin resistance and cardiovascular risk in patients with type 2 diabetes." Diabetes, Obesity and Metabolism, 2022; 24(8): 1451-1459.
2. Jastreboff AM, Aronne LJ, Ahmad NN, Wharton S, Connery L, Alves B, Kiyosue A, Zhang S, Liu B, Bunck MC, Stefanski A. "Tirzepatide Once Weekly for the Treatment of Obesity." New England Journal of Medicine, 2022; 387(3): 205-216.
3. Frías JP, Davies MJ, Rosenstock J, Pérez Manghi FC, Fernández Landó L, Bergman BK, Liu B, Cui X, Brown KS, Grandy S, Urva S. "Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes." New England Journal of Medicine, 2021; 385(6): 503-515.
4. Coskun T, Sloop KW, Loghin C, Alsina-Fernandez J, Urva S, Bokvist KB, Cui X, Briere DA, Cabrera O, Roell WC, Kuchibhotla U. "LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus: From discovery to clinical proof of concept." Molecular Metabolism, 2018; 18: 3-14.
5. Thomas MK, Nikooienejad A, Bray R, Cui X, Wilson J, Duffin K, Milicevic Z, Haupt A, Robins DA. "Dual GIP and GLP-1 Receptor Agonist Tirzepatide Improves Beta-cell Function and Insulin Sensitivity in Type 2 Diabetes." Journal of Clinical Endocrinology & Metabolism, 2021; 106(2): 388-396.
6. Rosenstock J, Wysham C, Frías JP, Kaneko S, Lee CJ, Fernández Landó L, Mao H, Cui X, Karanikas CA, Thieu VT. "Efficacy and safety of a novel dual GIP and GLP-1 receptor agonist tirzepatide in patients with type 2 diabetes (SURPASS-1): a double-blind, randomised, phase 3 trial." The Lancet, 2021; 398(10295): 143-155.


_1745918362135.webp)






