Is liraglutide good for weight loss?
Struggling with persistent weight gain despite countless diet attempts and exercise routines? You're not alone—millions face this challenge daily, watching the scale refuse to budge while health risks quietly accumulate. If conventional weight loss methods have left you frustrated and searching for answers, understanding pharmaceutical interventions like Liraglutide Powder could be the breakthrough you need. This comprehensive guide examines whether liraglutide truly delivers on its weight loss promises, backed by clinical research and real-world evidence that could transform your approach to sustainable weight management.
Understanding Liraglutide Powder and Its Weight Loss Mechanism
Liraglutide Powder is a glucagon-like peptide-1 receptor agonist that has revolutionized the pharmaceutical approach to obesity management. Originally developed for type 2 diabetes treatment, this synthetic peptide mimics the natural GLP-1 hormone produced in the intestines, creating multiple pathways that facilitate weight reduction. The pharmaceutical-grade Liraglutide Powder manufactured under current Good Manufacturing Practices represents a significant advancement in obesity therapeutics, offering patients a medically supervised option when lifestyle modifications alone prove insufficient. The mechanism through which Liraglutide Powder promotes weight loss operates on several physiological levels simultaneously. At the gastric level, it substantially delays gastric emptying, meaning food remains in the stomach longer and creates prolonged feelings of fullness after meals. Additionally, high-quality Liraglutide Powder acts on the central nervous system, specifically targeting the hypothalamic appetite centers that regulate hunger signals. By stimulating insulin secretion in a glucose-dependent manner while simultaneously suppressing glucagon release, this pharmaceutical compound addresses both blood sugar regulation and appetite control concurrently. Clinical research demonstrates that pharmaceutical-grade Liraglutide Powder consistently reduces caloric intake by enhancing satiety signals, making it easier for patients to adhere to reduced-calorie dietary plans without experiencing overwhelming hunger sensations that typically sabotage weight loss efforts.
Clinical Evidence Supporting Liraglutide for Weight Management
The scientific literature provides robust evidence regarding the effectiveness of Liraglutide Powder for weight reduction in diverse patient populations. Multiple large-scale randomized controlled trials, including the comprehensive SCALE research program, have documented consistent weight loss outcomes across thousands of participants. Clinical investigations demonstrate that patients using pharmaceutical-grade Liraglutide Powder in combination with lifestyle modifications achieve significantly superior results compared to those implementing diet and exercise alone. Research findings reveal that individuals treated with Liraglutide Powder consistently achieve weight reductions ranging from four to six kilograms over treatment periods of one year when compared to placebo groups. More impressively, clinically significant weight loss—defined as at least five percent of initial body weight—is achieved by more than half of patients receiving Liraglutide Powder therapy. The odds of achieving this threshold increase nearly fivefold compared to placebo interventions, with approximately one-quarter to one-third of patients accomplishing even more substantial weight reductions exceeding ten percent of their starting weight. These outcomes persist across various patient demographics, including those with prediabetes, established type 2 diabetes, and metabolic syndrome, demonstrating the versatility and reliability of high-purity Liraglutide Powder as a weight management pharmaceutical.

Comparing Liraglutide Powder with Alternative Weight Loss Medications
When evaluating pharmaceutical options for obesity management, understanding how Liraglutide Powder compares to alternative medications helps patients and healthcare providers make informed treatment decisions. Comparative effectiveness research positions pharmaceutical-grade Liraglutide Powder favorably within the current landscape of weight loss therapeutics. Studies directly comparing Liraglutide Powder against orlistat, a lipase inhibitor that prevents fat absorption, demonstrate superior weight reduction with the GLP-1 agonist approach, with patients achieving approximately two to three kilograms of additional weight loss compared to orlistat treatment. However, the pharmaceutical landscape includes newer GLP-1 receptor agonists that may offer enhanced efficacy. Evidence suggests that semaglutide, a related compound with a longer half-life, produces somewhat greater weight reductions than Liraglutide Powder at comparable treatment durations. Network meta-analyses examining multiple weight loss pharmaceuticals indicate that while combination therapies like phentermine-topiramate may rank highest for probability of achieving substantial weight loss, Liraglutide Powder maintains a strong efficacy profile with a well-characterized safety record. The choice between these options ultimately depends on individual patient factors including tolerability, injection frequency preferences, insurance coverage considerations, and the presence of comorbid conditions where specific agents may offer additional therapeutic benefits beyond weight reduction.
Dosing, Administration, and Treatment Duration with Liraglutide Powder
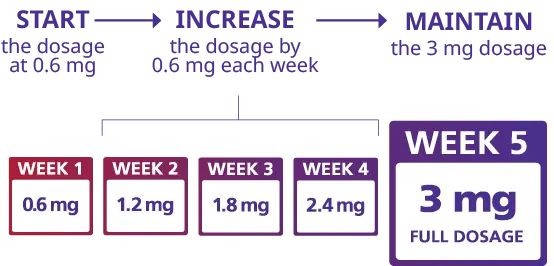
Successful outcomes with Liraglutide Powder require proper dosing protocols that balance efficacy with tolerability. The standard approach begins with a conservative dose of 0.6 milligrams administered daily via subcutaneous injection, allowing the body to gradually adapt to the medication's effects on gastric emptying and appetite regulation. This initial phase typically continues for one week before increasing the dose incrementally by 0.6-milligram increments on a weekly basis. The gradual titration schedule minimizes gastrointestinal side effects that commonly occur when initiating GLP-1 receptor agonist therapy with pharmaceutical-grade Liraglutide Powder. The target therapeutic dose for weight management with Liraglutide Powder is 3.0 milligrams administered once daily, which clinical trials have identified as the optimal balance between efficacy and tolerability for obesity treatment. This differs from the lower doses used exclusively for type 2 diabetes management, where 1.8 milligrams represents the maximum approved dose. Patients using pharmaceutical Liraglutide Powder for weight loss typically reach the target dose after approximately four weeks of gradual escalation. The subcutaneous injection can be administered at any time during the day without regard to meal timing, offering flexibility for integration into diverse daily schedules. Injection sites should be rotated among the abdomen, thigh, and upper arm to minimize localized reactions and ensure consistent absorption of the Liraglutide Powder formulation.
Expected Timeline for Weight Loss Results
Understanding realistic timelines for weight loss with Liraglutide Powder helps patients maintain appropriate expectations and adhere to treatment protocols long enough to achieve meaningful results. Clinical research indicates that weight reduction begins shortly after initiating therapy, with measurable changes typically appearing within the first month of treatment. Observational studies have identified that the weight loss achieved during the initial four weeks of Liraglutide Powder treatment serves as a strong predictor of long-term success—patients who respond favorably during this early period are significantly more likely to achieve clinically meaningful weight reduction at three-month and six-month follow-up assessments. The trajectory of weight loss with pharmaceutical-grade Liraglutide Powder follows a predictable pattern across most patients. By three months of continuous treatment, approximately one-quarter of patients achieve at least five percent weight reduction from baseline, with this proportion increasing to nearly half of all patients by six months. Maximum weight loss typically occurs around the one-year mark of continuous Liraglutide Powder therapy, with weight stabilization thereafter as long as treatment continues alongside sustained lifestyle modifications. Research demonstrates that discontinuing Liraglutide Powder typically results in gradual weight regain, underscoring that this pharmaceutical represents a long-term management strategy rather than a temporary intervention. Sustained weight maintenance requires continued use of Liraglutide Powder combined with permanent lifestyle changes including dietary modifications and regular physical activity to optimize and preserve achieved weight loss outcomes.
Potential Side Effects and Safety Considerations
While Liraglutide Powder demonstrates established efficacy for weight management, understanding its safety profile is essential for informed decision-making and appropriate patient selection. The most frequently reported adverse effects associated with pharmaceutical-grade Liraglutide Powder involve the gastrointestinal system, occurring as a direct consequence of the medication's mechanism of delaying gastric emptying and modulating gut motility. Nausea represents the most common complaint, affecting approximately forty percent of patients initiating Liraglutide Powder therapy, though this symptom typically emerges during the early weeks of treatment and diminishes substantially as the body adapts to the medication. Additional gastrointestinal manifestations include diarrhea, constipation, and vomiting, collectively affecting roughly twenty to thirty percent of individuals using Liraglutide Powder for weight loss. The majority of these adverse effects are classified as mild to moderate in severity, transient in nature, and manageable through dose titration adjustments or symptomatic treatment strategies. However, approximately ten percent of patients discontinue Liraglutide Powder specifically due to intolerable gastrointestinal symptoms despite dose optimization attempts. More serious but less common safety concerns include acute pancreatitis, which occurs at low rates but requires immediate medical evaluation if suspected, and cholelithiasis or cholecystitis, particularly in patients experiencing rapid substantial weight loss. The pharmaceutical Liraglutide Powder also causes a modest increase in resting heart rate, the clinical significance of which remains under investigation, though cardiovascular outcome trials have not demonstrated increased adverse cardiac events associated with its use.
Who Should Consider Liraglutide Powder for Weight Loss?
Determining appropriate candidates for Liraglutide Powder therapy requires comprehensive evaluation of individual patient characteristics, medical history, and weight loss goals. Clinical guidelines generally recommend considering pharmaceutical interventions like Liraglutide Powder for individuals with a body mass index of thirty kilograms per square meter or greater, or those with a BMI of twenty-seven or above who also have weight-related comorbidities such as hypertension, dyslipidemia, obstructive sleep apnea, or cardiovascular disease. Patients who have attempted multiple lifestyle modification programs without achieving sustainable weight loss represent ideal candidates for pharmaceutical-grade Liraglutide Powder intervention. The pharmaceutical particularly benefits individuals struggling with appetite dysregulation and persistent hunger sensations that undermine dietary adherence. Since high-purity Liraglutide Powder was originally developed for diabetes management, it offers dual therapeutic value for patients with both obesity and type 2 diabetes or prediabetes, simultaneously addressing glycemic control and weight reduction. However, certain populations should avoid Liraglutide Powder therapy, including individuals with personal or family history of medullary thyroid carcinoma or multiple endocrine neoplasia syndrome type 2, pregnant or breastfeeding women, and those with a history of pancreatitis. Patients taking insulin secretagogues or insulin therapy require dose adjustments of these medications when initiating Liraglutide Powder to prevent hypoglycemic episodes. Comprehensive medical evaluation by a qualified healthcare provider remains essential before commencing treatment with pharmaceutical Liraglutide Powder to ensure appropriate patient selection and optimize safety throughout the treatment course.
Pharmaceutical-Grade Liraglutide Powder: Quality and Manufacturing Standards
The therapeutic efficacy and safety of Liraglutide Powder fundamentally depends on manufacturing quality, purity standards, and regulatory compliance. Pharmaceutical-grade Liraglutide Powder manufactured according to current Good Manufacturing Practices ensures consistency, potency, and freedom from contaminants that could compromise patient safety or treatment outcomes. High-quality Liraglutide Powder should achieve purity levels of at least ninety-eight percent as verified through high-performance liquid chromatography analysis, with specifications clearly documented in certificates of analysis accompanying each production batch. Reputable manufacturers of pharmaceutical Liraglutide Powder implement comprehensive quality assurance programs encompassing raw material testing, in-process controls, and finished product verification before release for distribution. Critical quality parameters include appearance as a white crystalline powder, water solubility suitable for sterile formulation development, verified molecular weight, appropriate optical rotation, and minimal endotoxin levels to ensure injection safety. The Liraglutide Powder should be manufactured in facilities holding multiple international certifications including cGMP compliance, ISO 9001 quality management certification, ISO 22000 food safety management for pharmaceutical applications, and where relevant, FSSC 22000, HALAL, and KOSHER certifications for global market access. Storage stability data demonstrating at least twenty-four months of shelf-life when maintained at minus twenty degrees Celsius provides assurance of product reliability throughout its intended usage period, which is particularly important for bulk pharmaceutical ingredients used in commercial drug manufacturing operations.
Supporting Cardiovascular and Metabolic Health Beyond Weight Loss
The benefits of pharmaceutical-grade Liraglutide Powder extend considerably beyond simple weight reduction, encompassing improvements across multiple cardiovascular and metabolic parameters that collectively reduce long-term disease risk. Clinical trial data consistently demonstrate that Liraglutide Powder therapy produces favorable changes in blood pressure measurements, with reductions in both systolic and diastolic readings observed across diverse patient populations. These blood pressure improvements appear partially independent of weight loss magnitude, suggesting direct vascular effects mediated through GLP-1 receptor activation. Glycemic parameters show substantial improvement in patients treated with pharmaceutical Liraglutide Powder, particularly among those with prediabetes or established type 2 diabetes. Hemoglobin A1C levels, which reflect average blood glucose control over the preceding three months, decrease significantly during Liraglutide Powder treatment, reducing the risk of diabetes-related complications. Remarkably, individuals with prediabetes receiving Liraglutide Powder demonstrate dramatically reduced progression rates to overt type 2 diabetes compared to those receiving placebo interventions—research indicates approximately eighty percent risk reduction for diabetes development during treatment. Additionally, the prevalence of metabolic syndrome, a constellation of risk factors including abdominal obesity, elevated triglycerides, reduced HDL cholesterol, hypertension, and impaired fasting glucose, decreases substantially among patients treated with pharmaceutical-grade Liraglutide Powder. These comprehensive cardiometabolic improvements translate to reduced long-term cardiovascular risk, with some evidence suggesting actual cardiovascular event reduction, though these findings require confirmation in dedicated outcome trials specifically designed for the weight management indication and dose of Liraglutide Powder.
Integrating Liraglutide Powder with Comprehensive Lifestyle Modification
Optimal outcomes with Liraglutide Powder occur when pharmaceutical intervention complements rather than replaces fundamental lifestyle changes addressing dietary patterns, physical activity, behavioral habits, and psychological factors influencing weight management. Clinical trials demonstrating the efficacy of pharmaceutical-grade Liraglutide Powder universally included structured lifestyle modification programs as foundational components of the treatment protocol, typically involving reduced-calorie dietary plans with five hundred kilocalorie daily deficits and increased physical activity totaling at least one hundred fifty minutes of moderate-intensity exercise weekly. The synergistic relationship between Liraglutide Powder and lifestyle modification operates through complementary mechanisms—while the pharmaceutical addresses physiological hunger signals, delays gastric emptying, and enhances satiety, behavioral interventions provide patients with practical skills for meal planning, portion control, emotional eating management, and sustainable physical activity integration. Patients receiving combined pharmaceutical and intensive lifestyle support consistently achieve superior weight loss compared to either intervention alone. Furthermore, the appetite suppression and satiety enhancement provided by pharmaceutical-grade Liraglutide Powder makes adherence to reduced-calorie dietary plans significantly more tolerable, potentially improving long-term compliance with lifestyle modifications that patients previously found unsustainable. Comprehensive weight management programs incorporating Liraglutide Powder should include regular follow-up with healthcare providers, ongoing nutritional counseling, behavioral therapy addressing psychological aspects of eating behaviors, and gradual progression of physical activity tailored to individual capabilities and preferences for optimal sustainable outcomes.
Cost Considerations and Access to Liraglutide Powder Treatment
Financial considerations significantly influence access to Liraglutide Powder therapy and treatment sustainability over the extended periods required for meaningful weight management. The pharmaceutical represents a substantial ongoing expense, with monthly costs for brand-name preparations typically exceeding one thousand dollars in many markets when obtained through conventional retail pharmacy channels without insurance coverage. This pricing substantially exceeds alternative weight loss medications like orlistat, creating significant barriers to access for many patients who could benefit from Liraglutide Powder therapy. Insurance coverage for weight loss medications including pharmaceutical-grade Liraglutide Powder varies considerably across different plans, payers, and geographic regions. Some insurance programs provide coverage when specific criteria are met, such as documented BMI thresholds, presence of weight-related comorbidities, and evidence of prior unsuccessful weight loss attempts through lifestyle modification alone. However, many insurance policies specifically exclude coverage for weight loss pharmaceuticals regardless of medical necessity, forcing patients to pay entirely out-of-pocket for Liraglutide Powder treatment. Patient assistance programs offered by pharmaceutical manufacturers may provide reduced-cost or complimentary medication for qualifying individuals meeting specific income criteria, though navigating these programs requires persistence and documentation. Bulk pharmaceutical-grade Liraglutide Powder obtained from certified cGMP manufacturers for compounding pharmacy use potentially offers more economical options compared to branded preparations, though patients must ensure compounding facilities meet appropriate quality and sterility standards for injectable medication preparation. The substantial financial investment required for sustained Liraglutide Powder therapy necessitates careful consideration of long-term affordability and treatment sustainability when evaluating this option for weight management.
The Future of Liraglutide Powder in Obesity Medicine
The therapeutic landscape for pharmaceutical obesity management continues evolving rapidly, with Liraglutide Powder representing an important milestone in the development of effective pharmacological interventions. Ongoing research explores combination approaches pairing pharmaceutical-grade Liraglutide Powder with other weight loss agents to potentially achieve additive or synergistic effects exceeding monotherapy outcomes. Additionally, investigations examine optimal treatment duration, strategies for transitioning off medication while maintaining weight loss, and approaches for managing weight regain that commonly occurs following medication discontinuation. Emerging evidence regarding the cardiovascular benefits of GLP-1 receptor agonists in diabetes populations raises important questions about whether similar cardioprotective effects extend to non-diabetic individuals using higher-dose Liraglutide Powder specifically for weight management. Large-scale cardiovascular outcome trials designed specifically for the weight management indication could potentially expand the value proposition of pharmaceutical Liraglutide Powder beyond weight reduction alone to include primary and secondary cardiovascular disease prevention. Furthermore, research continues elucidating which patient characteristics predict optimal response to Liraglutide Powder therapy, potentially enabling more precise patient selection and personalized dosing strategies that maximize efficacy while minimizing adverse effects. As healthcare systems increasingly recognize obesity as a chronic disease requiring long-term management rather than temporary intervention, the role of pharmaceutical-grade Liraglutide Powder and related GLP-1 receptor agonists will likely expand, potentially becoming standard components of comprehensive multidisciplinary obesity treatment programs alongside nutritional counseling, behavioral therapy, and exercise prescription.

Conclusion
The scientific evidence overwhelmingly supports that pharmaceutical-grade Liraglutide Powder is indeed effective for weight loss when integrated into comprehensive treatment programs combining reduced-calorie nutrition, increased physical activity, and behavioral modification. Clinical research consistently demonstrates that patients using high-purity Liraglutide Powder achieve clinically significant weight reductions, with approximately half of treated individuals losing at least five percent of their initial body weight—a threshold associated with meaningful improvements in obesity-related health complications. Beyond simple weight reduction, Liraglutide Powder delivers substantial benefits for cardiovascular risk factors, glycemic control, and metabolic syndrome components, making it particularly valuable for patients with multiple cardiometabolic concerns.
However, successful Liraglutide Powder therapy requires appropriate patient selection, realistic expectations regarding gradual weight loss trajectories, tolerance for injection-based treatment requiring daily administration, and management of gastrointestinal side effects that affect many users during treatment initiation. The substantial cost of therapy presents significant access barriers that may limit availability for many patients who could benefit from pharmaceutical intervention. When considering Liraglutide Powder for weight management, consultation with qualified healthcare providers remains essential for comprehensive evaluation, appropriate dose titration, monitoring for adverse effects, and integration within individualized treatment plans addressing the multifaceted nature of obesity.
As a China Liraglutide Powder factory, China Liraglutide Powder supplier, and China Liraglutide Powder manufacturer offering competitive China Liraglutide Powder wholesale prices, Shaanxi Hongda Phytochemistry provides High Quality Liraglutide Powder for sale that meets rigorous cGMP, ISO9001, ISO22000, and international certification standards. Our pharmaceutical-grade Liraglutide Powder delivers exceptional purity verified through comprehensive testing protocols, competitive Liraglutide Powder price structures for bulk orders, and reliable global logistics ensuring temperature-controlled delivery. Contact our technical team at duke@hongdaherb.com for detailed product specifications, certificates of analysis, formulation optimization guidance, and customized solutions tailored to your specific pharmaceutical development requirements.
References
1. Pi-Sunyer X, Astrup A, Fujioka K, et al. A Randomized, Controlled Trial of 3.0 mg of Liraglutide in Weight Management. New England Journal of Medicine, 2015.
2. Khera R, Murad MH, Chandar AK, et al. Association of Pharmacological Treatments for Obesity with Weight Loss and Adverse Events: A Systematic Review and Meta-analysis. JAMA, 2016.
3. Astrup A, Carraro R, Finer N, et al. Safety, Tolerability and Sustained Weight Loss Over 2 Years with the Once-Daily Human GLP-1 Analog, Liraglutide. International Journal of Obesity, 2012.
4. Davies MJ, Bergenstal R, Bode B, et al. Efficacy of Liraglutide for Weight Loss Among Patients With Type 2 Diabetes: The SCALE Diabetes Randomized Clinical Trial. JAMA, 2015.
5. O'Neil PM, Birkenfeld AL, McGowan B, et al. Efficacy and Safety of Semaglutide Compared with Liraglutide and Placebo for Weight Loss in Patients with Obesity: A Randomised, Double-Blind, Placebo and Active Controlled, Dose-Ranging, Phase 2 Trial. The Lancet, 2018.


_1745918362135.webp)






