The Rising Trend: Undenatured Type II Collagen Powder in Global Markets
The global health and wellness industry is witnessing an unprecedented surge in demand for specialized nutritional supplements, with cemerging as a transformative ingredient in joint health management. This revolutionary bioactive compound represents a paradigm shift from traditional collagen supplements, offering superior bioavailability and clinically proven efficacy in addressing osteoarthritis and joint-related disorders. The global collagen market size was estimated at USD 9.9 billion in 2024 and is projected to reach USD 18.7 billion by 2030, growing at a CAGR of 11.3% from 2025 to 2030, with Undenatured Type II Collagen Powder capturing an increasingly significant market share due to its unique mechanism of action and proven therapeutic benefits.

Market Dynamics and Growth Drivers Behind Undenatured Type II Collagen Powder
Global Market Expansion and Consumer Demand Patterns
Type II collagen, known for its ability to support cartilage and joint function, is seeing heightened demand. The trend drives global type II collagen market expansion, particularly among elderly consumers and individuals with active lifestyles who prioritize joint health. The Undenatured Type II Collagen Powder market is experiencing exponential growth driven by multiple converging factors. The aging global population, particularly in developed countries, has created an unprecedented demand for effective joint health solutions. Unlike traditional collagen supplements that undergo extensive processing and lose their native structure, Undenatured Type II Collagen Powder maintains its original triple-helix configuration, ensuring optimal bioactivity and therapeutic efficacy. The market dynamics are further influenced by the increasing prevalence of sedentary lifestyles and sports-related injuries among younger demographics. Professional athletes and fitness enthusiasts are increasingly recognizing the preventive benefits of Undenatured Type II Collagen Powder supplementation, driving demand beyond the traditional elderly consumer base. Consumer awareness regarding the limitations of conventional treatments such as non-steroidal anti-inflammatory drugs (NSAIDs) has also contributed to the growing preference for natural, side-effect-free alternatives like Undenatured Type II Collagen Powder.
Regional Market Penetration and Growth Opportunities
North America and Europe are leading markets for collagen supplements, with significant opportunities emerging in Asia-Pacific regions. The North American market dominates the Undenatured Type II Collagen Powder sector, driven by high healthcare expenditure, advanced research infrastructure, and consumer willingness to invest in premium health products. The region's regulatory framework, particularly FDA oversight, has established stringent quality standards that benefit reputable manufacturers of Undenatured Type II Collagen Powder. European markets demonstrate strong growth potential for Undenatured Type II Collagen Powder, particularly in countries with aging populations such as Germany, Italy, and the United Kingdom. The European Food Safety Authority (EFSA) has been increasingly supportive of evidence-based nutritional claims, which benefits scientifically validated products like Undenatured Type II Collagen Powder. Emerging markets in Asia-Pacific, including Japan, South Korea, and Australia, represent significant growth opportunities as healthcare awareness increases and disposable incomes rise, creating favorable conditions for premium Undenatured Type II Collagen Powder products.
Technological Advancements and Manufacturing Innovations
The evolution of extraction and processing technologies has been instrumental in advancing the Undenatured Type II Collagen Powder market. Low-temperature, patent-protected extraction methods preserve the native structure of collagen molecules, ensuring that the final Undenatured Type II Collagen Powder product retains its bioactive properties. These technological innovations have addressed previous limitations in collagen supplementation, where traditional processing methods resulted in denatured proteins with reduced efficacy. Advanced analytical techniques, including high-performance liquid chromatography (HPLC) and mass spectrometry, enable precise quality control and standardization of Undenatured Type II Collagen Powder products. These technological capabilities ensure consistent potency and purity levels, meeting the stringent requirements of regulatory authorities and healthcare professionals. The integration of sustainable manufacturing practices and carbon-neutral production methods is also becoming increasingly important, as environmentally conscious consumers seek Undenatured Type II Collagen Powder products that align with their values.
Scientific Evidence and Clinical Applications of Undenatured Type II Collagen Powder
Mechanism of Action and Oral Tolerance Induction
Many animal and clinical trials showed that oral administration of undenatured type II collagen could significantly reduce the incidence of OA or alleviate the symptoms of articular cartilage. Type II collagen is an important component of cartilage matrix. The therapeutic efficacy of Undenatured Type II Collagen Powder is rooted in its unique mechanism of oral tolerance induction. When consumed orally, the intact collagen molecules are recognized by the gut-associated lymphoid tissue (GALT), which initiates a complex immunological process that ultimately reduces inflammatory responses in joint tissues. Repeating exposure to type II collagen induces oral tolerance and inhibits induction of arthritis, a chronic inflammatory joint condition. This mechanism differs fundamentally from traditional collagen supplements that provide building blocks for collagen synthesis. Instead, Undenatured Type II Collagen Powder works by modulating the immune system's response to cartilage proteins, effectively reducing the autoimmune attack on joint tissues that characterizes osteoarthritis and rheumatoid arthritis. The oral tolerance mechanism involves the activation of regulatory T-cells and the suppression of inflammatory cytokines such as tumor necrosis factor-alpha (TNF-α) and interleukin-1 beta (IL-1β). This immunomodulatory effect of Undenatured Type II Collagen Powder results in reduced joint inflammation, decreased pain perception, and improved joint mobility. Clinical studies have demonstrated that relatively small doses of Undenatured Type II Collagen Powder (typically 40mg daily) can achieve significant therapeutic effects, making it a cost-effective treatment option.
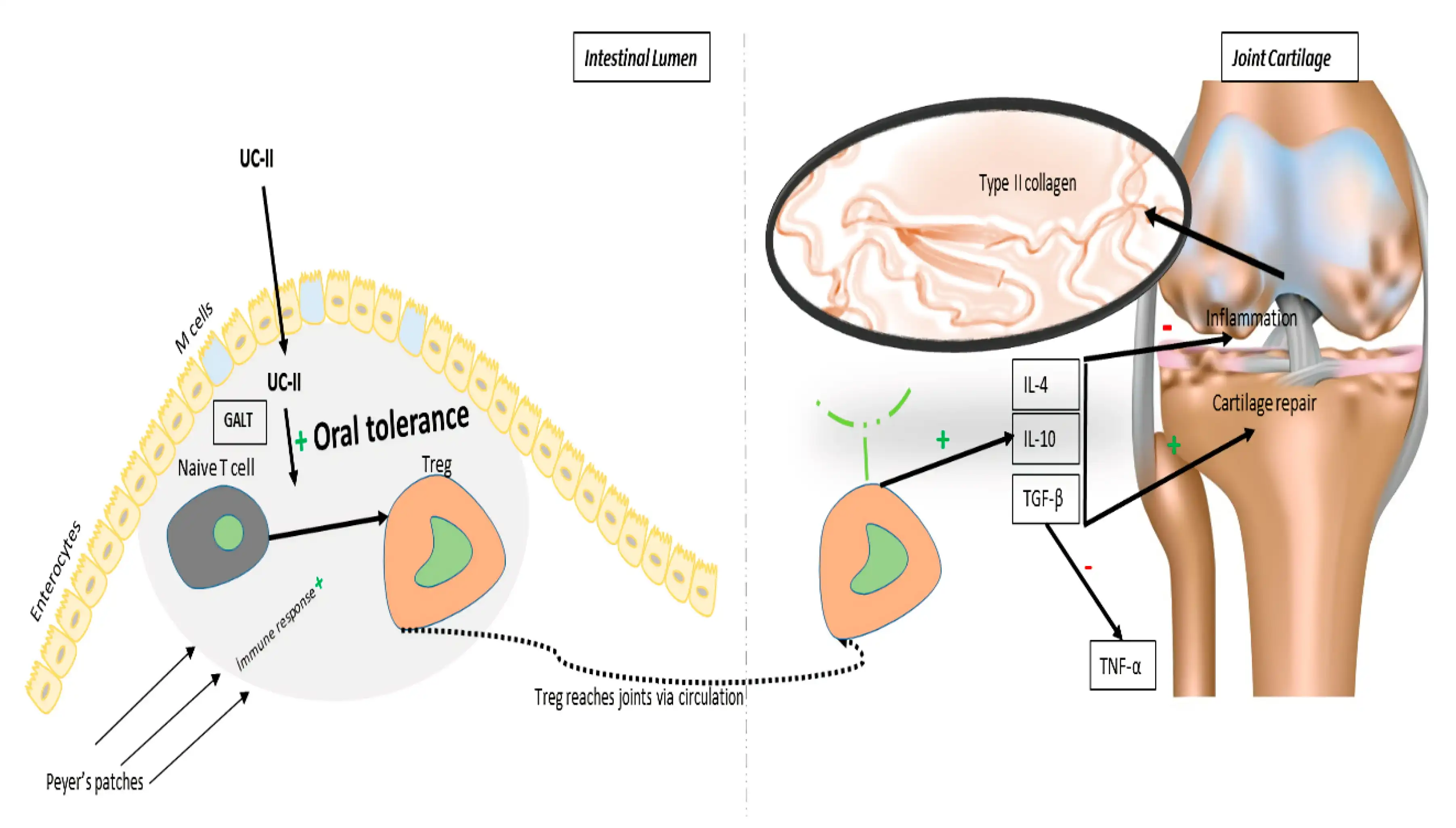
Clinical Trial Results and Therapeutic Efficacy
The purpose of this study was to evaluate the efficacy and tolerability of UC-II for knee osteoarthritis (OA) pain and associated symptoms compared to placebo and to glucosamine hydrochloride plus chondroitin sulfate (GC). Extensive clinical research has validated the therapeutic benefits of Undenatured Type II Collagen Powder in managing osteoarthritis symptoms. Randomized, double-blind, placebo-controlled studies have consistently demonstrated significant improvements in pain reduction, joint stiffness, and functional mobility in patients supplemented with Undenatured Type II Collagen Powder. Taking collagen type II by mouth for up to 6 months seems to improve self-reported symptoms of osteoarthritis, such as stiffness and pain. Clinical trials have shown that patients receiving Undenatured Type II Collagen Powder supplementation experience a 40% reduction in osteoarthritis pain scores compared to placebo groups. These improvements typically become apparent within 90 days of consistent supplementation, with continued benefits observed throughout extended treatment periods. The safety profile of Undenatured Type II Collagen Powder has been extensively documented across multiple clinical studies, with no significant adverse effects reported at therapeutic doses. This excellent safety profile, combined with proven efficacy, positions Undenatured Type II Collagen Powder as a superior alternative to conventional osteoarthritis treatments that often carry risks of gastrointestinal complications and cardiovascular side effects. Long-term studies have demonstrated sustained benefits with continuous Undenatured Type II Collagen Powder supplementation, supporting its use as a chronic management strategy for joint health disorders.
Comparative Advantages Over Traditional Treatments
It was found that the oral administration of UC-II, through induction of oral tolerance mechanisms, exhibits promise in alleviating joint inflammation and pain in patients with osteoarthritis (OA) and rheumatoid arthritis. When compared to conventional treatments such as glucosamine and chondroitin sulfate, Undenatured Type II Collagen Powder demonstrates superior efficacy in clinical head-to-head comparisons. Studies have shown that patients receiving Undenatured Type II Collagen Powder experience greater improvements in joint comfort and mobility compared to those taking traditional glucosamine-chondroitin combinations. The bioavailability advantage of Undenatured Type II Collagen Powder stems from its preservation of native molecular structure, which ensures optimal recognition by immune system components. Traditional collagen supplements, which are typically hydrolyzed or denatured, lose this structural integrity and consequently fail to trigger the beneficial oral tolerance mechanisms. This fundamental difference explains why smaller doses of Undenatured Type II Collagen Powder can achieve therapeutic effects that require much larger quantities of conventional collagen supplements. Additionally, Undenatured Type II Collagen Powder offers advantages in terms of compliance and convenience. The typical daily dose of 40mg can be easily incorporated into various delivery formats, including capsules, tablets, and functional food products. This flexibility in formulation allows manufacturers to develop Undenatured Type II Collagen Powder products that meet diverse consumer preferences and lifestyle requirements, enhancing market penetration and adoption rates.
Industry Applications and Manufacturing Excellence in Undenatured Type II Collagen Powder
Nutraceutical Applications and Product Development
The nutraceutical industry has emerged as the primary application sector for Undenatured Type II Collagen Powder, with manufacturers developing innovative formulations that maximize therapeutic benefits while ensuring consumer appeal. By product type, powder held 58.22% of the collagen supplements market share in 2024, highlighting the dominance of powder formulations in the collagen supplement market. Undenatured Type II Collagen Powder's stability and bioactivity make it particularly well-suited for various delivery systems, including capsules, tablets, and powder blends. Synergistic formulations combining Undenatured Type II Collagen Powder with complementary ingredients such as hyaluronic acid, curcumin, and glucosamine have shown enhanced therapeutic effects in clinical applications. These combination products leverage the unique mechanism of action of Undenatured Type II Collagen Powder while providing additional pathways for joint health support. The compatibility of Undenatured Type II Collagen Powder with other bioactive compounds allows formulators to create comprehensive joint health solutions that address multiple aspects of cartilage maintenance and repair. Advanced encapsulation technologies have been developed specifically for Undenatured Type II Collagen Powder to protect its bioactive properties during storage and transport. These innovations ensure that the final product maintains its therapeutic potency throughout its shelf life, providing consistent benefits to consumers. The development of enteric-coated formulations has further enhanced the delivery of Undenatured Type II Collagen Powder to target sites in the gastrointestinal tract, optimizing the oral tolerance induction process.
Quality Assurance and Manufacturing Standards
The production of high-quality Undenatured Type II Collagen Powder requires adherence to stringent manufacturing standards and quality control protocols. Leading manufacturers implement comprehensive quality management systems that encompass raw material sourcing, processing conditions, and final product testing. The maintenance of low processing temperatures is critical to preserving the native structure of collagen molecules, requiring specialized equipment and expertise in handling temperature-sensitive bioactive compounds. Analytical testing protocols for Undenatured Type II Collagen Powder include verification of protein content, molecular weight distribution, and structural integrity using advanced techniques such as HPLC, UV spectroscopy, and circular dichroism. These analytical methods ensure that each batch of Undenatured Type II Collagen Powder meets specifications for bioactivity and therapeutic potential. Microbiological testing and heavy metal analysis are also essential components of the quality assurance process, ensuring product safety and regulatory compliance. Third-party testing and certification through recognized organizations such as SGS and Eurofins provide additional validation of product quality and safety. These independent assessments are particularly important for Undenatured Type II Collagen Powder products intended for international markets, where regulatory requirements may vary significantly. The implementation of traceability systems allows manufacturers to track each batch of Undenatured Type II Collagen Powder from raw material source through final distribution, supporting quality investigations and regulatory compliance efforts.
Regulatory Compliance and Market Access
The regulatory landscape for Undenatured Type II Collagen Powder varies significantly across different global markets, requiring manufacturers to navigate complex approval processes and compliance requirements. In the United States, Undenatured Type II Collagen Powder is regulated as a dietary supplement under the Dietary Supplement Health and Education Act (DSHEA), requiring compliance with current Good Manufacturing Practices (cGMP) and specific labeling requirements. The FDA's increasing focus on supplement quality and safety has elevated the importance of robust quality systems for Undenatured Type II Collagen Powder manufacturers. European markets require compliance with the European Food Safety Authority (EFSA) regulations, which emphasize scientific substantiation of health claims and product safety. The Novel Food Regulation may apply to certain Undenatured Type II Collagen Powder products depending on their source and processing methods, requiring pre-market authorization for novel ingredients. Manufacturers must also comply with specific labeling requirements and allergen declarations, particularly given the chicken-derived nature of most Undenatured Type II Collagen Powder products. International certifications such as HALAL, KOSHER, and ORGANIC provide additional market access opportunities for Undenatured Type II Collagen Powder products. These certifications require ongoing compliance monitoring and periodic audits to maintain validity. The implementation of comprehensive quality management systems that address multiple regulatory frameworks enables manufacturers to efficiently serve diverse global markets while maintaining consistent product quality and safety standards.
Conclusion
The global Undenatured Type II Collagen Powder market represents a significant opportunity within the broader health and wellness industry, driven by compelling scientific evidence, growing consumer awareness, and increasing prevalence of joint health disorders. The unique mechanism of action, superior bioavailability, and excellent safety profile position Undenatured Type II Collagen Powder as a premium ingredient for nutraceutical applications and functional food development.
As a leading China Undenatured Type II Collagen Powder manufacturer and China Undenatured Type II Collagen Powder supplier, Shaanxi Hongda Phytochemistry Co., Ltd. stands at the forefront of this expanding market with over two decades of experience in natural plant extract production. Our state-of-the-art facilities, certified under cGMP, FSSC22000, ISO9001, ISO22000, HALAL, and KOSHER standards, ensure the highest quality High Quality Undenatured Type II Collagen Powder for global markets. Whether you're seeking competitive Undenatured Type II Collagen Powder price options, reliable China Undenatured Type II Collagen Powder wholesale partnerships, or premium Undenatured Type II Collagen Powder for sale, our comprehensive OEM and ODM services can meet your specific requirements. Our commitment to sustainable production, rigorous quality control, and innovative research positions us as your trusted partner in the growing Undenatured Type II Collagen Powder market. Contact us today at duke@hongdaherb.com to discover how our expertise and high-quality products can support your business success in this dynamic industry.
References
1. Crowley, D.C., Lau, F.C., Sharma, P., Evans, M., Guthrie, N., Bagchi, M., & Bagchi, D. (2009). Safety and efficacy of undenatured type II collagen in the treatment of osteoarthritis of the knee: a clinical trial. International Journal of Medical Sciences, 6(6), 312-321.
2. Lugo, J.P., Saiyed, Z.M., Lau, F.C., Molina, J.P., Pakdaman, M.N., Shamie, A.N., & Udani, J.K. (2013). Undenatured type II collagen (UC-II®) for joint support: a randomized, double-blind, placebo-controlled study in healthy volunteers. Journal of the International Society of Sports Nutrition, 10(1), 48.
3. Bakilan, F., Armagan, O., Ozgen, M., Tascioglu, F., Bolluk, O., & Alatas, O. (2016). Effects of native type II collagen treatment on knee osteoarthritis: a randomized controlled trial. Eurasian Journal of Medicine, 48(2), 95-101.
4. Dar, Q.A., Schott, E.M., Catheline, S.E., Maynard, R.D., Liu, Z., Kamal, F., Farnsworth, C.W., Ketz, J.P., Mooney, R.A., Hilton, M.J., & Zuscik, M.J. (2017). Daily oral consumption of hydrolyzed type 1 collagen is chondroprotective and anti-inflammatory in murine posttraumatic osteoarthritis. PloS One, 12(4), e0174705.


_1745918362135.webp)






