What Are the Main Constituents in High-Quality Liposomal Nad Powder?
A good liposomal NAD powder has three main parts: phospholipid bilayers mostly made up of phosphatidylcholine from non-GMO sunflower lecithin; pharmaceutical-grade Nicotinamide Adenine Dinucleotide (NAD⁺) with purity levels above 98%; and special ingredients that keep the powder stable. At Hongda Phytochemistry, we know that getting the right ingredients is key. We know how these parts work together to give you better solubility, which is usually 8 to 10 times higher than regular NAD+ forms. The phospholipid covering protects the NAD+ molecule from being broken down in the stomach and makes it easier for cells to take it in directly through membrane fusion processes. In today's competitive nutraceutical market, expensive formulas can be told apart from lesser ones by quality control measures like encapsulation efficiency above 92%, particle size distribution between 100 and 300 nm, and full third-party proof.
Understanding Liposomal NAD Powder: Composition and Core Constituents
Knowing the different layers that make up liposomal NAD powder products is essential to knowing how well they work. When procurement experts look at suppliers, they have to look at three linked groups of factors that affect how well the product works and how profitable it is.
The Active Pharmaceutical Ingredient: NAD⁺ Purity and Stability
Nicotinamide adenine dinucleotide is an important coenzyme in cellular metabolism. It takes part in more than 500 enzyme processes that are needed to make energy, fix DNA, and keep mitochondria working. Shaanxi Hongda Phytochemistry Co., Ltd. makes liposomal NAD powder that has been tested and found to be pure (NAD+ values of ≥98%) using High-Performance Liquid Chromatography (HPLC). This level of purity meets medicinal standards and gets rid of any impurities that could affect stability or treatment results. Our light yellow powder stays the same colour after going through strict sensory testing, which makes sure it doesn't have any smells, changes, or obvious alien impurities that could mean it's getting old or wasn't made right.
Oral administration methods are very hard to make work because NAD+ molecules need to stay together. When unprotected NAD⁺ comes into contact with stomach acid, it breaks down quickly and goes through a lot of first-pass hepatic processing. This means that in most pill forms, the bioavailability rate is less than 5%. Because of this problem, we need more improved liposomal encapsulation technologies that can keep the chemical structure of NAD+ while it is being digested.
Phospholipid Bilayers: The Delivery System Foundation
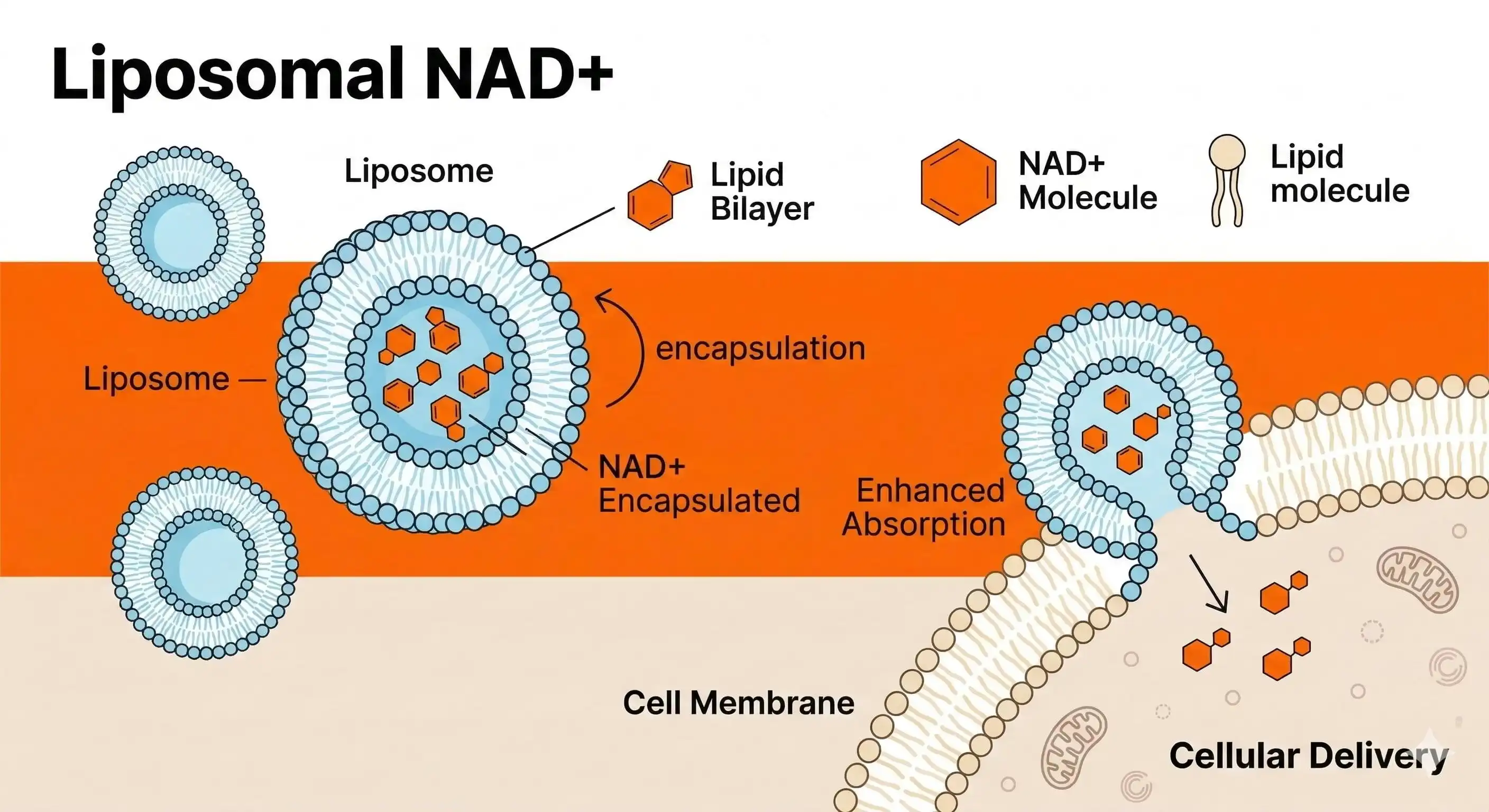
Phospholipids give liposomal mixtures their structure and help make spherical spheres that look like the membranes of human cells. High-quality formulas use phosphatidylcholine that is extracted from sunflower lecithin, which is better for clean labelling than standard options that come from soy. High-pressure homogenised microparticle encapsulation technology is used in Hongda's production process to insert NAD⁺ in natural phospholipid bilayers at the nanoscale level. This makes a "biological Trojan horse" that keeps the active ingredient safe from being broken down by enzymes while also making transmembrane transfer easier.
The phospholipid makeup has a direct effect on packaging efficiency, which is a very important quality indicator. Premium liposomal NAD powder regularly has absorption rates above 92%. This means that most of the NAD+ molecules are protected by lipid structures and not just floating around as free powder. With every shipment, we include a report on the particle size distribution that shows Z-average readings between 100 and 300 nm and a Polydispersity Index (PDI) number below 0.3. This shows that the liposome populations are uniform, which is important for steady cell uptake.
Stabilising Excipients and Processing Innovations
In addition to the NAD⁺-phospholipid core, stabilising excipients make the mixture last longer and keep its structure even when stored in a variety of ways. Our one-of-a-kind low-temperature vacuum drying method turns liquid liposomal solutions into stable powders that keep the active ingredients inside at the microscopic level. This new way of processing solves a major problem in the industry: liquid liposomal products usually need to be kept cool and go bad within a few weeks, which makes it hard to ship them around the world.
The powder that was made is very stable and will stay effective for 24 months at room temperature if it is kept away from light, moisture, and air. The loss on drying values of 8.48%, which is much lower than the standard of 10%, show that the moisture is being managed well, stopping hydrolysis processes that break down NAD+ and phospholipid structures. This stability profile gets rid of the need for a cold chain, which lowers the costs of shipping for foreign business-to-business clients while still making sure of the integrity of the product when it gets to factories in different countries.
Why Constituent Quality Matters in Liposomal NAD Powder Procurement?
The quality of the constituents is the most important factor in determining both the treatment effectiveness and the economic success of finished goods. When choosing a supplier, procurement professionals should put verifiable quality measures ahead of cost alone, because problems with constituents directly lead to lower bioavailability and worse results for end users in the liposomal NAD powder market.
Bioavailability Enhancement Through Optimised Formulation
The process by which liposomal carriers and intestinal cell membranes fuse together makes it possible for better absorption. Our liposome capsule method keeps NAD+ from being broken down by stomach acid. This way, the active ingredient goes straight to the bloodstream, where it is 8 to 10 times more bioavailable than regular NAD+ powder. This big change is due to the synergy between the parts: high-purity NAD+ mixed with perfectly sized phospholipid vesicles makes it possible for cells to take nutrients directly without using normal absorption routes.
Certificates of Analysis (CoAs) are an objective way to check these quality standards. Hongda includes a lot of information with every batch, such as HPLC purity proof, particle size analysis using dynamic light scattering, measures of encapsulation efficiency, and heavy metal screening. Our dedication to pharmaceutical-grade quality that meets strict international regulatory standards is shown by the fact that our arsenic levels of 0.01 mg/kg and lead content of 0.02 mg/kg are both much lower than the 2.0 mg/kg standards.
Safety and Compliance in Regulated Markets
The safety of liposomal products depends on how well their constituents are kept together. Phospholipids that aren't up to par may still have solvents, oxidised lipid leftovers, or allergenic proteins that cause bad responses. When we buy non-GMO sunflower lecithin, we don't have to worry about soy allergens, and the "clean label" labelling is useful in places where people are paying more attention to where ingredients come from.
GMP-compliant microbiological testing makes sure that every batch meets strict cleanliness requirements. Our FSSC 22000, ISO 22000, and cGMP certifications show that we use structured quality management to find raw materials, make products, and test the finished goods. These qualifications are especially important for B2B clients that do business in controlled markets like the US and EU. These markets have strict rules about paperwork that needs to be kept all the way through the supply chain, from buying ingredients to registering finished goods.
Economic Implications of Quality Variance
The total cost of ownership estimate shows that premium parts are more cost-effective, even though they have higher unit prices. More NAD+ gets into the systemic circulation per dose when encapsulation efficiency is higher. This lets product makers reach effective levels with smaller amounts of active ingredient. The 24-month shelf life gets rid of the costs of product obsolescence that come with formulations that don't last as long. This makes managing cash flow easier and lowers the cost of getting rid of trash.
Our minimum order number of 1 kg covers test production runs and product development stages. This lets R&D teams check how well the formulation works before committing to bulk purchases. This gives you more options, which means you have less financial risk during the product creation cycle, and you can still get the pharmaceutical-grade ingredients you need for useful efficacy tests.
Comparative Analysis: Liposomal NAD Powder vs Other NAD Supplement Forms
Understanding the different recipe options lets you make a smart choice that fits the needs of your product's marketing and target market. There are a lot of different ways to give NAD⁺ in the nutraceutical world, and each one has its own specific performance and ingredient issues concerning liposomal NAD⁺ powder and alternatives.
Liposomal Powder vs Traditional Capsules and Tablets
Normal NAD+ pills and tablets have a solid active ingredient mixed with common fillers and coatings, such as cellulose binders, magnesium stearate flow agents, and protective coatings. The bioavailability of these products is very low because stomach acid breaks down NAD⁺ molecules very quickly before they can be absorbed, and liver first-pass metabolism further lowers the amount that gets into the systemic circulation. Bioavailability rates for oral NAD+ products that are not enclosed are usually less than 5%, according to clinical reports.
Liposomal technology gets around these problems by enclosing the substance in a safe shell. Our fast transmembrane transport method lets liposome carriers work like human cell membranes, getting into cells without hurting them and adding to the NAD+ stores inside them directly. This delivery benefit means that therapeutic effects can be achieved at much lower amounts. This opens the door for cost-effective product formulations that keep market prices low while still providing better results for consumers.
The powder shape gives manufacturers more options for how to make things. Liposomal NAD powder can be added to a variety of delivery methods, such as stick packs, effervescent tablets, functional beverage mixes, and custom capsule blends with ingredients that work well together. Free-flowing and stable at room temperature make it easier to use automatic filler equipment. This makes production simpler than with liquid liposomal suspensions, which need to be handled carefully and kept in the fridge.
NAD⁺ vs Precursor Compounds: NMN and NR
Nicotinamide mononucleotide (NMN) and nicotinamide riboside (NR) are two NAD⁺ sources that can be bought on the market as other ways to raise NAD⁺ levels inside cells. Before these molecules can be used by the cells' metabolism, they need to be changed by enzymes. Because precursors are stable and have a small molecular size, they are easily absorbed by the body. However, the multistep conversion process adds factors that change how much NAD+ is available in the end.
Adding NAD+ directly through liposomes skips the conversion steps and gives cells instant access to substrates for processes. This difference is especially important for uses that need to help with short-term energy, recovery after exercise, or brain enhancement, where speed of action is important. Our recipe helps fix DNA by directly activating PARP enzymes. This speeds up the repair of the skin layer and the maintenance of cells without relying on metabolic conversion rates that vary from person to person.
The complexity of the constituents is very different between these choices. For precursor formulations, there is only one active ingredient, and the production needs are pretty simple. Liposomal NAD⁺ is a complex way to give drugs that needs special technology for encapsulation, quality control analytics, and manufacturing know-how. Hongda has put money into modern production facilities and SGS-certified labs that work with universities. This lets us keep the quality standards of the ingredients high enough to support the higher prices these formulations command in the market.
Selecting Trusted Manufacturers and Suppliers of High-Quality Liposomal NAD Powder
Choosing the right supplier has a big effect on the stability of product quality, following the rules, and the long-term success of a business. Systematic evaluation frameworks that look at manufacturing skills in more than just price comparisons are helpful for procurement workers seeking liposomal NAD powder.
Essential Manufacturing Credentials and Infrastructure
Certification packages give factory-quality systems concrete proof that they work. There are many certifications that Hongda Phytochemistry has, such as cGMP, FSSC 22000, ISO 9001, ISO 22000, HALAL, KOSHER, and ORGANIC certifications for EU and NOP norms. These qualifications prove that the company uses a structured approach to quality management that includes checking the quality of raw materials, keeping an eye on the production process, testing finished products, and keeping records that are needed for regulatory entries in global markets.

Supply dependability is directly affected by the capacity of the physical infrastructure. Our current production facilities are about 20,000 square metres and have more than 10 specialised production lines and workshops for purification at the 100,000-level. This scale can support production of more than 3,000 tonnes per year, so there will always be enough to meet market demand. Over 150 trained workshop workers run the plant by following standard operating procedures that have been developed over 20 years of manufacturing experience.
Laboratory skills set advanced makers apart from commodity sellers. Our SGS-certified lab has more than 20 research and development staff at the professor level who use high-tech analysis tools like atomic absorption units, gas chromatographs, spectrophotometers, HPLC systems, and biochemical testing instruments, including the analysis of liposomal NAD powder. This analytical power allows for thorough checking of each production batch's ingredients. Extra tests are also done by authorised third-party agencies like SGS and Eurofins to check for pesticide residues, make sure nutritional labels are correct, and meet customer-specific analytical needs.

Conclusion
The therapeutic effectiveness and economic feasibility of liposomal NAD powder formulations depend on the quality of their constituents. Professionals in purchasing must judge sellers based on quality indicators that can be checked, such as NAD+ purity levels above 98%, phospholipid encapsulation efficiency levels above 92%, and full certification files that show they follow GMP and international quality standards. According to research, properly designed liposomal delivery methods improve bioavailability by 8 to 10 times compared to regular NAD+ supplements. This is why these goods are priced so high. Picking manufacturing partners with strong analytical tools, methods for tracking raw materials, and expert support resources is important for successful product development and long-term supply chain stability, which is needed for growth in the competitive nutraceutical market.
FAQ
1. What role does NAD+ play in cellular health and metabolism?
Nicotinamide adenine dinucleotide is an important coenzyme that is needed for more than 500 enzyme processes that control how cells make energy, fix DNA, and work the mitochondria. NAD+ is an important part of the electron transport chain, which is where cells make ATP, which is the basic energy currency that all living things need to work. Low amounts of NAD⁺ are linked to ageing and metabolic problems, which makes supplementation methods more important for uses that aim to extend life.
2. How does encapsulation efficiency affect product quality?
Encapsulation efficiency measures how much NAD+ is successfully trapped within liposomal structures compared to being an open powder that is not protected. Higher encapsulation rates—ideally above 92%—protect active ingredients from stomach acid and improve their ability to be taken up by cells. Formulations with lower efficiency lose expensive active ingredients and don't always work as well as they should, so this standard is very important when evaluating suppliers.
3. Are there long-term safety considerations for NAD+ supplementation?
NAD+ is an endogenous molecule that is found naturally in all parts of the body. It has a good safety rating that is backed up by a lot of studies. Tolerability ratings are very good for high-quality formulations made according to GMP guidelines, with purity checks and full heavy metal screens. Our methods for checking microbes make sure that every batch meets pharmaceutical-grade safety standards that are good for long-term supplementation plans.
Partner with HONGDA for Premium Liposomal NAD Powder Supply
Pharmaceutical-grade liposomal nad powder is made by Shaanxi Hongda Phytochemistry Co., Ltd., which has been in business for 24 years and has a lot of quality certifications, such as cGMP, FSSC 22000, and ISO standards. Our advanced high-pressure homogenised microparticle encapsulation technology achieves an encapsulation rate of more than 92%, and thorough HPLC research confirms that the NAD+ is pure at ≥98%. We keep a steady supply of inventory to support sales of at least 1 kg by buying in bulk in 20 kg drums. All of our products have 24-month shelf stability, which takes the guesswork out of cold-chain operations. We are a reliable provider of liposomal nad powder to nutraceutical companies, dietary supplement makers, and functional beverage formulators around the world. We offer full technical support, custom formulation services, and all regulatory paperwork. You can email our procurement team at duke@hongdaherb.com to ask for Certificates of Analysis, talk about OEM customisation options, or set up facility checks that show our dedication to quality excellence and help you succeed with your product development.
References
1. Amjad S, Nisar S, Bhat AA, Shah AR, Frenneaux MP, fakhro K, Haris M, Reddy R, Patay Z, Baur J, Bagga P. "Role of NAD+ in regulating cellular and metabolic signaling pathways." Molecular Metabolism, 2021.
2. Covarrubias AJ, Perrone R, Grozio A, Verdin E. "NAD+ metabolism and its roles in cellular processes during ageing." Nature Reviews Molecular Cell Biology, 2021.
3. Liu L, Su X, Quinn WJ, Hui S, Krukenberg K, Frederick DW, Redpath P, Zhan L, Chellappa K, White E, Migaud M, Mitchison TJ, Baur JA, Rabinowitz JD. "Quantitative analysis of NAD synthesis-breakdown fluxes." Cell Metabolism, 2018.
4. Poddar SK, Sifat AE, Haque S, Nahid NA, Chowdhury S, Mehedi I. "Nicotinamide Mononucleotide: Exploration of Diverse Therapeutic Applications of a Potential Molecule." Biomolecules, 2019.
5. Shade C. "Liposomes as Advanced Delivery Systems for Nutraceuticals." Integrative Medicine: A Clinician's Journal, 2016.
6. Xie N, Zhang L, Gao W, Huang C, Huber PE, Zhou X, Li C, Shen G, Zou B. "NAD+ metabolism: pathophysiologic mechanisms and therapeutic potential." Signal Transduction and Targeted Therapy, 2020.


_1745918362135.webp)






