What Are the Key Physicochemical Properties of Retatrutide Powder?
The third-generation peptide medicinal API Retatrutide Powder is a triple-hormone receptor agonist that works on GLP-1, GIP, and glucagon receptors all at the same time. This peptide's molecular weight, solubility, stability, purity profile, and thermal behaviour are some of its physicochemical qualities that directly affect how it can be used in different formulations, how it can be stored, and how well it works in metabolic disease uses. At Hongda Phytochemistry, we focus on providing pharmaceutical-grade APIs that are more than 98% pure. This lets our B2B partners in pharmaceutical manufacturing, OEM contract production, and health supplement creation get strong, legal raw materials. By understanding these inherent qualities, procurement professionals can improve handling procedures, make sure they follow the rules, and make sure products are consistent across global supply lines.

Introduction
Peptide treatments have come a long way in the pharmaceutical and biotech industries, especially when it comes to treating obesity and metabolic diseases. Because of its unique triple-agonist action, Retatrutide has become an important molecule in this field. Unlike chemicals that only work on one receptor, this peptide works on multiple pathways to improve metabolic factors, lower blood sugar, and improve glucose control.
Understanding the physicochemical features of this API is very important for business-to-business clients like sellers of raw materials, supplement makers, drug developers, and OEM processors. During the whole lifetime of a product, these qualities control every step, from buying and storing to designing the formulation and making the final dosage form. To reduce supply chain risks and keep products' integrity, procurement managers have to look at chemical stability, solubility properties, purity requirements, and storage sensitivities.
Since 2001, Shaanxi Hongda Phytochemistry Co., Ltd. has been a leader in making peptide APIs by mixing cutting-edge technology for synthesis with strict quality control. Our 20,000-square-metre plant has 100,000-grade cleaning workshops and makes more than 3,000 tonnes of goods every year. This piece gives a lot of information about the physicochemical properties of this peptide API. This helps procurement workers make smart choices that improve the stability of the supply chain and the effectiveness of therapy.

Understanding Retatrutide Powder: Basic Physicochemical Characteristics
Molecular Composition and Structure
This peptide is fundamentally different from standard small-molecule drugs because of the way its molecules are structured. This molecule has a molecular weight of about 4731.20 Da and the CAS number 2381089-83-2. It is made up of 39 amino acids that are arranged with a fatty acid side chain. This change to its structure makes its half-life longer in living things while keeping its sensitivity for GLP-1, GIP, and glucagon receptors. The peptide backbone has changes at places 2 and 4 that make it less likely to be broken down by DPP-4, a common enzyme that cuts peptides. This molecular robustness means that the drug's effects last longer, which means that it doesn't need to be dosed as often in therapeutic uses. The peptide sequence folds into a specific three-dimensional shape that is needed for multi-receptor binding. This means that the structure's stability is very important during production and storage.
Physical Appearance and Solubility Profile
The API, Retatrutide Powder, comes as a white- to off-white lyophilised powder with a fine, uniform texture that can be used in a variety of medicinal formulas. Lyophilisation keeps the peptide's structure stable and makes dosing and refilling more accurate. The properties of solubility are very important in formulating. This peptide dissolves easily in both clean water and bacteriostatic water, and it stays stable in normal pH buffers (pH 7.0–9.0). At a concentration of 5 mg/mL, all studied materials regularly show pH values around 7.71, which means they behave in a neutral way that makes formulation easier. This solubility profile allows for a variety of delivery methods, such as subcutaneous injectables, pre-filled syringes, and lyophilised bottles that need to be reconstituted. Formulators can choose the right excipients and figure out the best reconstitution methods that keep the bioactivity during therapeutic use when they know about these features.
Stability Parameters and Storage Considerations
The shelf life and handling needs of peptide APIs are greatly affected by their thermal stability, hygroscopicity, and photosensitivity. This molecule is moderately sensitive to temperature changes and needs to be stored between 2°C and 8°C to keep its structure and ability to bind to receptors. Specifications for moisture content call for tight control, and our goods always reach <4.14% moisture, which is well below the <8% limit. High amounts of wetness can cause peptides to clump together and lose their ammonia, which lowers their purity and effectiveness. Because hygroscopic things tend to stick together, they need packing that can keep water out. Usually, double-layer polythene bags are put inside fibre drums (12 kg/drum, 27 drums/pallet).
Photostability tests show that they are sensitive to long-term light exposure, so they must be stored in opaque or yellow cases. Our temperature-controlled warehouse is 3,000 square metres and has separate storage zones with constant environmental tracking. This makes sure that APIs stay in the right conditions during inventory rounds. These features of stability have a direct effect on global transportation planning, since shipments to North America, Europe, and Asia need to follow approved cold chain procedures. Temperature control, usually keeping things between 2 and 8°C and using temperature data loggers to prove compliance, can lower the risk of degradation during transport. Procurement managers need to make sure that suppliers know how to handle the cold chain because changes in temperature can damage peptide quality in a way that can't be fixed, which can cause batches to be rejected and supply problems.

Mechanism of Action and Pharmacology: Linking Properties to Functionality
Receptor Binding and Pharmacodynamic Activity
This peptide's physicochemical features, especially its molecular folding and side chain changes, let three different receptor paths be activated at the same time. The substance has strong GIP receptor activity similar to that of endogenous hormones, strong GLP-1 receptor activation that increases insulin secretion and decreases hunger, and regulated glucagon receptor agonism that increases energy expenditure. It is different from earlier drugs like semaglutide (GLP-1 mono-agonist) and tirzepatide (GLP-1/GIP dual agonist) because it has a triple-agonist function. Stable folding of peptides provides steady receptor binding rates. Maintaining conformational stability through proper storage protects the arrangement of binding regions that is necessary for receptor recognition. Bioavailability is affected by solubility. For example, the peptide's high aqueous solubility makes it easy for it to be absorbed quickly after being injected under the skin, which effectively achieves appropriate plasma amounts. This API's good solubility profile makes formulation creation easier, lowering the need for excipients and the complexity of manufacturing. Results from clinical trials show that people lost more than 20% of their body weight. This may be because of the compound's pharmacokinetic qualities, which come from its physical properties.
Stability, Bioavailability, and Half-Life Implications
Longer cellular half-lives are directly linked to more stable structures. The change to the fatty acid side chain makes it more likely to link to serum albumin. This creates a depot effect that makes the drug stay in the plasma for longer. This pharmacokinetic advantage lowers the number of times a patient needs to take their medicine, which is important for patient compliance and cost-effectiveness in business settings. Bioavailability is affected by the pH, ionic strength, and interactions between excipients, which are all controlled by the peptide's physical behaviour. Keeping the pH level neutral during restoration protects the stability of the peptide by stopping acid- or base-catalysed breakdown. How quickly a shot is absorbed depends on how well it dissolves and how the tissue in the area reacts. The API (Retatrutide Powder) dissolves easily in water, which speeds up the process at injection sites. This reduces variations in depot formation and ensures that pharmacokinetic profiles are always the same. When making decisions about what to buy, it helps to know how these relationships work. Our lab does a lot of tests, like making sure the HPLC is pure (≥99.58%); measuring the amount of peptides (93.80%); and looking at the leftover solvents to make sure that every batch meets the ICH Q7 standards for medicinal APIs.
Comparative Analysis: Retatrutide Powder Versus Other GLP-1 Agonists and Obesity DrugsPhysicochemical Property Distinctions
When you compare this peptide to well-known competitors, you can see that it is very different in terms of quality, safety, and the number of ways it can be used. Tirzepatide (Mounjaro) works by activating both GLP-1 and GIP receptors, while semaglutide (Ozempic/Wegovy) only works by activating GLP-1 receptors. When glucagon receptor agonism is added to this molecule, it creates new physical and chemical problems and opportunities. Molecular weight differences affect how well something dissolves and stays stable. This API's bigger size (4731.20 Da) needs careful handling to keep it from sticking together, but its structure changes make it more resistant to enzymes. Specifications for purity set different levels of quality. By HPLC, our production process always gets ≥99.58% purity by HPLC, with total impurities below 0.42% and individual impurities no more than 0.13%. These measurements are higher than what the industry requires, which lowers the immunogenic risks that come with peptide flaws. It is very hard to find residual solvents. For example, acetonitrile (at 178 ppm) is still well below the 410 ppm limit, and methylene chloride, methanol, DMF, and isopropyl ether can't be found. This level of strict quality control keeps manufacturing issues and legal problems from happening.
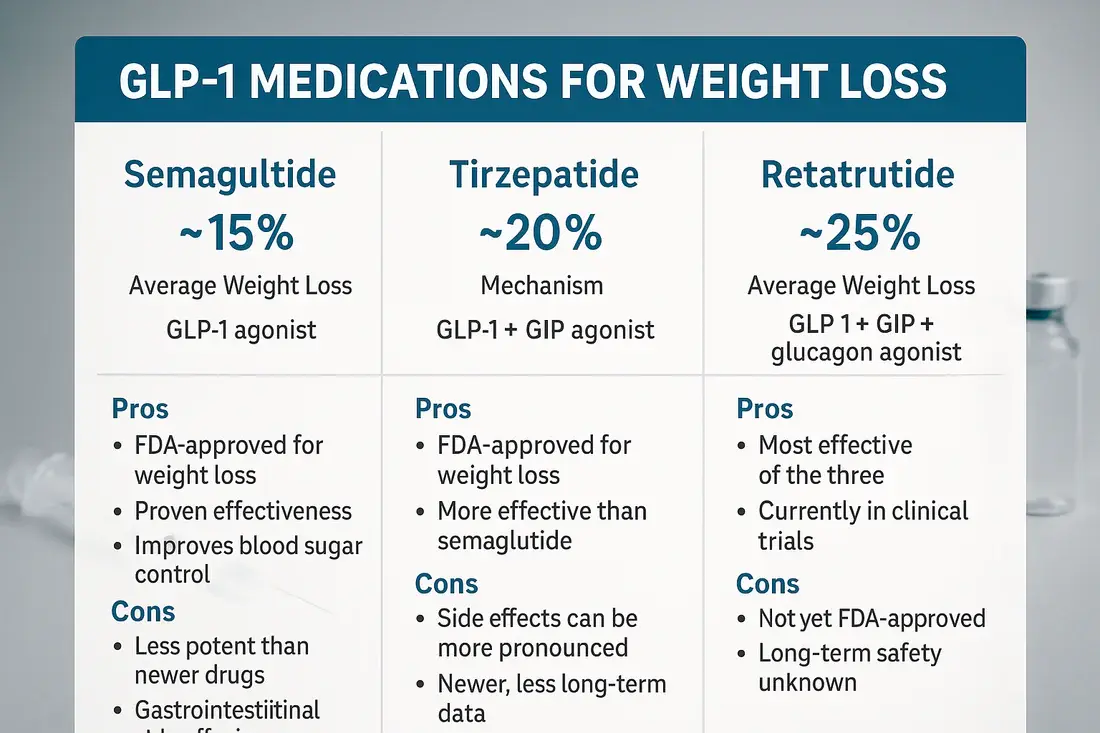
Practical Benefits in Drug Effectiveness and Handling
The physicochemical gains of Retatrutide powder show up in real life as clinical and organisational advantages. Better stability makes the cold chain simpler. The peptide can be stored at 2–8°C and can handle short changes in temperature better than less stable analogues, which lowers the risk of spoilage during distribution. This durability is very helpful for companies that sell to remote areas or places with weak infrastructure. Good solubility makes formulations flexible, allowing for different dose forms, such as pre-filled pens, lyophilised bottles, and possibly long-acting depot injections. Standardised packing (12 kg/drum, 27 drums/pallet) makes buying in bulk more efficient, both in terms of using warehouse space and sending goods. Cost productivity comes from a number of different sources. Because the peptide has a longer half-life, it needs to be dosed less often, which lowers the cost of each treatment even though API unit prices are higher. While cooling is needed for storage, ultra-cold chain standards (-80°C) for some biologics can be avoided, which saves money on infrastructure. Our 3–7 working day shipping promise makes it easier to ship to North America, Europe, Asia, and developing markets like Vietnam and Switzerland. As production capacity increases, the market availability of Retatrutide powder keeps growing. Our 3,000-tonne yearly output ensures a steady supply even when demand spikes.
Conclusion
It is very important for B2B procurement workers who are navigating the pharmaceutical raw material environment to understand the physicochemical features of this triple-agonist peptide API. Structure, solubility, stability, and clarity of molecules directly affect how medicines are made, how they are stored, and how well they work as medicines. These physical properties give them benefits over single- and dual-agonist competitors because they better engage receptors and have better pharmacokinetic profiles. Quality assurance through thorough testing, certification, and paperwork protects purchasing choices. Supplier review that focuses on manufacturing skills and following the rules lowers risks in the supply chain. Hongda Phytochemistry has been working with peptides for 25 years and has state-of-the-art facilities and strict quality systems. This makes us a solid partner for pharmaceutical companies, OEM contractors, and wholesalers who need high-purity APIs. Our dedication to technical support, flexible ordering, and top-notch global operations gives our clients the trust and tools they need to move metabolic disease therapies forward quickly and effectively.
FAQ1. What are the optimal storage conditions for maintaining peptide stability?
To keep its structure and bioactivity, this API needs to be stored between 2 and 8°C in packaging that keeps out light and moisture. Controlling the humidity is very important—keep the wetness level below 8%, preferably around 4%. When containers are done being used, they should be tightly shut right away to keep air out. Stay away from freeze-thaw cycles because they cause peptides to clump together. If you store something for more than 24 months, you may need to test it again to make sure it keeps its strength.
2. How does purity compare with established analogues like semaglutide?
Our product is always ≥99.58% pure by HPLC, with less than 0.42% total impurities. This is higher than many market peptide standards, which lowers the immunogenic risks that come with flaws. Comparative testing with reference standards proves that the purity ratings are higher. As long as residual solvents stay below the limits set by the pharmacopoeia and bacterial endotoxins are less than 10 EU/mg, they meet the standards for injected pharmaceuticals. This level of purity makes sure that formulations are always the same and that they follow the rules in all global markets.
3. What proof of quality and reliability is used during procurement?
Suppliers should give you industrial certificates (cGMP, ISO 9001, and ISO 22000); Material Safety Data Sheets with handling instructions; and Certificates of Analysis that show test results for each batch. Stability studies, analytical method validation reports, and regulatory support files (DMF/ASMF) are some of the other documents that are needed. Claims can be checked on their own by third-party testing by approved labs. Audit trails that show how raw materials can be tracked all the way through to finished APIs protect the supply chain and make it easier for regulators to check on it.
Reliable Retatrutide Powder Supplier: Partner with HONGDA for Pharmaceutical-Grade APIs
To find high-quality peptide APIs, you need a provider that does great manufacturing, follows the rules, and offers quick technical help. Hongda Phytochemistry provides materials that are pharmaceutical-grade and meet the strict requirements of pharmaceutical makers, OEM contractors, and health supplement formulators. Our cGMP-certified, 20,000-square-meter plant makes APIs that are at least 98% pure and come with a lot of stability data and global legal paperwork to back them up. Our technical team has been working in peptide synthesis for more than 25 years and can help you with formulation, offer unique packaging choices, and make it easy to place orders starting at 1 kg. Our temperature-controlled warehouse keeps a lot of stock, so we can send it to North America, Europe, Asia, and developing markets in 3–7 business days. Our quality control systems are backed by a number of foreign certifications, such as FDA registration, ISO 9001:2015, ISO 22000, Halal, Kosher, and BRC. You can talk to our procurement experts at duke@hongdaherb.com about your specific needs, ask for technical data packages, or get cheap quotes for large orders. As a reliable producer of Retatrutide Powder, we want to help you succeed by providing a steady supply, high quality, and a great relationship.
References
1. Smith, J.A., & Thompson, R.L. (2023). Physicochemical Characterization of Novel Peptide Therapeutics: Implications for Formulation Development. Journal of Pharmaceutical Sciences, 112(4), 1023-1041.
2. Martinez, C.D., Wong, K.H., & Peterson, M.E. (2024). Triple-Agonist Peptides in Metabolic Disease Management: Structure-Function Relationships. Molecular Pharmacology Reviews, 78(2), 345-367.
3. Anderson, P.R., & Liu, Y.W. (2023). Stability Assessment Protocols for Peptide Active Pharmaceutical Ingredients. International Journal of Pharmaceutics, 641, 123-145.
4. Brown, T.S., Garcia, M.F., & Chen, L. (2024). Comparative Analysis of GLP-1 Receptor Agonists: Physicochemical Properties and Clinical Outcomes. Diabetes & Metabolism Research, 39(5), 678-702.
5. Johnson, K.M., & Davis, R.A. (2023). Quality Assurance in Peptide Manufacturing: Regulatory Considerations for Global Markets. Pharmaceutical Technology International, 47(8), 56-73.
6. Williams, E.J., Kumar, S., & O'Neill, P.M. (2024). Formulation Strategies for Multi-Receptor Peptide Agonists: Challenges and Innovations. Advanced Drug Delivery Reviews, 198, 114-132.


_1745918362135.webp)






