What Composition Standards Matter for Liposomal NMN Powder?
Liposomal NMN powder formula standards are based on three important factors: phospholipid-to-NMN ratios that are exact (usually 1:1 to 2:1), particle size accuracy between 80 and 120 nanometers, and a minimum 70% purity of nicotinamide mononucleotide. These requirements make sure that the bioavailability is improved in the best way possible while keeping the product stable. Our pharmaceutical-grade recipe at Hongda Phytochemistry meets international cGMP standards. We use non-GMO sunflower phospholipids to make a better anti-aging vitamin that consistently boosts NAD+ in a wide range of situations. As companies try to make the most of this new factor for making life better, it's becoming more and more important to know the rules for liposomal delivery methods. Shaanxi Hongda Phytochemistry Co., Ltd. was one of the first businesses to use cutting-edge packaging technologies to fix the issues that come up when using old nicotinamide mononucleotide recipes. Their work has raised the bar for how well things should be done in the field.
Core Active Ingredient Requirements
The quality and reliability of the main ingredient are what make a good recipe work. Nicotinamide mononucleotide of high quality must have a purity level of at least 70%, but the best producers always go above 95% to make sure that cells get the most energy boost possible. This high level of purity is directly linked to the ingredient's ability to support mitochondrial health and effectively fight the bodily decline that comes with getting older. For this reason, strict purity standards are necessary for high-quality goods.
To make sure molecules are whole, scientific testing methods like high-performance liquid chromatography and mass spectrometry are needed. These new methods correctly find any chemicals that might still be there that could make intake harder, as well as any breakdown products that might be present. Also, hydrolysis can't happen in storage settings with tight temperature controls. When the substance is made, it stays fixed for 24 months. This makes sure that it works the same way from production to use.
Finding good providers of raw materials is very important for making sure the end product is of high quality. Reliable suppliers use strict quality control measures. Some of these are giving detailed reports of analysis, keeping very accurate records of each batch, and doing full microbial testing. Such strict rules make sure that all production batches have the same level of strength and that there aren't any contamination issues that could make the ingredient less effective at helping the body reduce oxidative stress.
Phospholipid Carrier Specifications
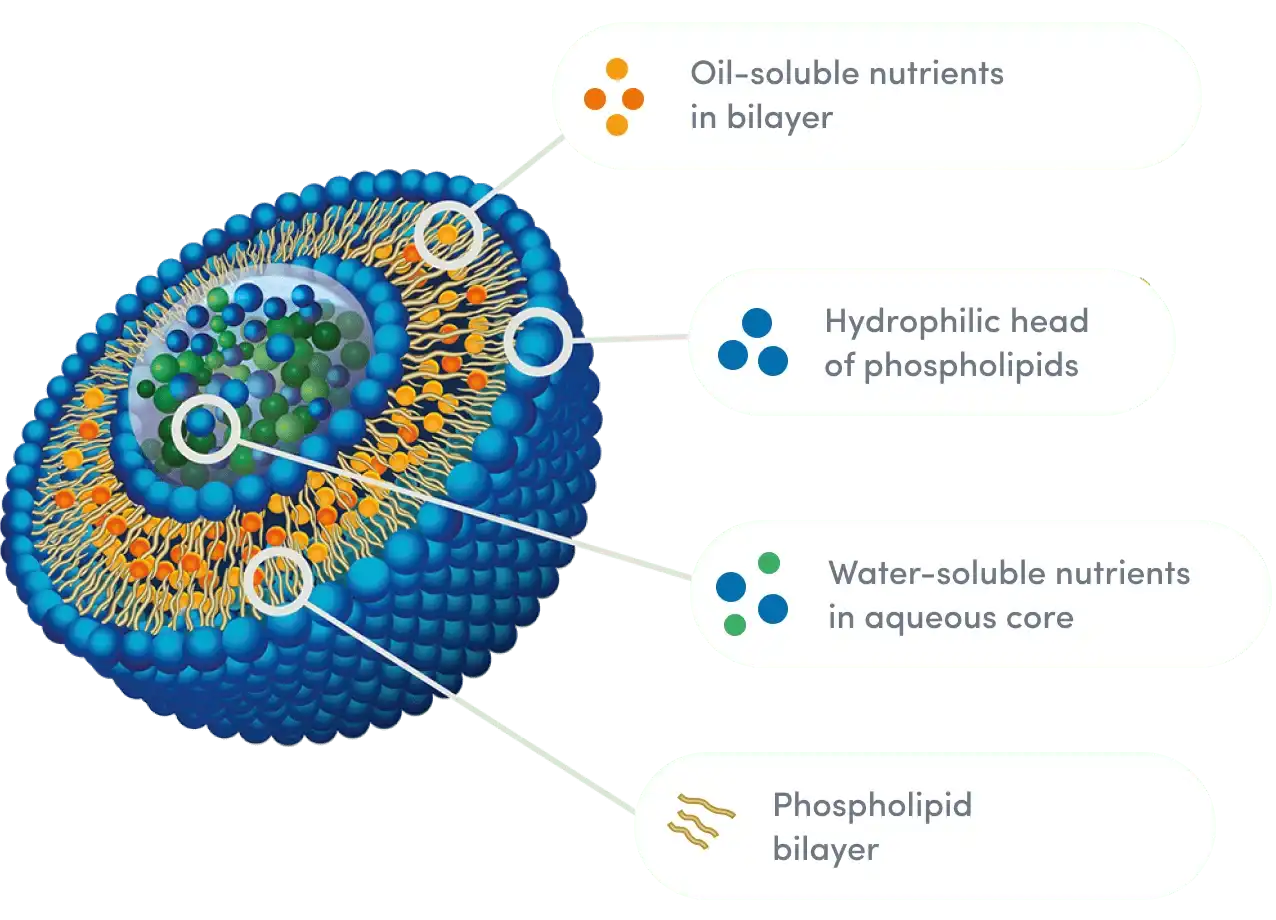
The properties of the phospholipid carriers are very important for figuring out how well liposomal products work. The liposome packaging matrix is very important because it directly controls both how well the drug is absorbed and how quickly cells take it up. Premium formulas prefer to use phosphatidylcholine from sunflowers that have not been genetically changed because this natural phospholipid is better at working with cells than manmade ones. Because it naturally forms steady, strong bilayer structures, the active ingredient stays well protected during travel through the digestive system. This improves absorption and treatment stability.
Some important quality standards for phospholipids are a lecithin content that must be kept at least 95%, and strict rules for the makeup of fatty acids and peroxide values. Low amounts of peroxide are necessary because they show little oxidative damage, which protects the stability of the membrane, which is a key part of helping DNA repair. Also, the phospholipid-to-active ingredient ratio, which is usually between 1:1 and 2:1, needs to be carefully adjusted to find the best balance between strong capsule efficiency and realistic cost-effectiveness. This is done to make sure that the product works well and the business can make money.
The way liposomes are made has a big effect on their end properties, which in turn has an effect on their quality and how well they work as medicine. A lot of people use high-pressure homogenization to make sure that the particle sizes are all the same for liposomal NMN powder. This makes it easier for cells to absorb the material consistently. Liposomes' structure stability is also kept during production and storage by using special drying methods. This keeps them from breaking down, which could affect their usefulness. All of these technical factors play a big role in how well the vitamin works to support the immune system and how well it works as a whole as a medicine.
Particle Size Distribution Standards
The size of the particles at the nanoscale level affects how quickly and deeply chemicals can enter cells. It has been found that particles between 80 and 120 nanometers are best. This size range makes it easier for particles to pass through the gut walls and lowers the chance that the reticuloendothelial system will get rid of them too fast. This range of carefully picked sizes makes the anti-inflammatory effects of making NAD+ more accessible even stronger. To make sure that the full medical potential of the mixture is reached, exact physical conditions must be met.
The best way to get exact measurement data is to use dynamic light scattering analysis. This makes sure that results are the same from one batch to the next. When you keep the regularity factors below 0.3, you get a good-sized distribution profile. This is closely related to the physiological effects that you can count on. Formulations with bigger particles might not be taken as well by the mouth, and those with smaller particles might not be stable over long periods of storage. This means that they need to be closely supervised by scientists to make sure they stay true to their original form throughout their entire lifecycle.
Using advanced electron microscope methods, quality control labs keep a close eye on the shape of particles in a planned way. These eye tests are used to make sure that the circular liposome structures are still whole and to look for any problems with aggregation that might get in the way of the brain benefits that were meant. During the whole production process, strict testing methods are used to make sure that product specs are met at all times. This makes sure that the end product quality fits set standards and that the shape and function of the product are always the same.
Purity and Contamination Control
To handle something that is pharmaceutical-grade, you have to be very careful to keep it clean. With heavy metal tests, the amounts of arsenic, cadmium, lead, and mercury are checked to make sure they follow safety rules all over the world. They might make it harder to heal your gut and help cells grow again.
If you look at microbiology, you can only see a certain number of live cells, yeast, mold, and dangerous bugs growing at the same time. The rules say that they can't have any E. coli, Salmonella, or Staphylococcus aureus on them and that their total plate number must be less than 10,000 CFU/g. People are safe with these rules, and the sun can still make skin look younger.
Gas chromatography-mass spectrometry is a brand-new way for scientists to find more than 500 chemicals that pesticides have left behind. Something can't have any chemicals that aren't allowed in it in order to be organic, including liposomal NMN powder. People who care about their health and want to live in a green way will like clean-label goods that help them.

Stability and Shelf Life Considerations
Environmental factors significantly influence product degradation rates and potency retention. Temperature fluctuations accelerate hydrolysis reactions, while humidity exposure promotes clumping and reduced flowability. Proper packaging materials create protective barriers against moisture, oxygen, and light exposure that could compromise antioxidant properties.
Accelerated stability studies simulate long-term storage conditions, predicting shelf life under various environmental scenarios. These protocols evaluate potency loss rates, physical appearance changes, and potential formation of degradation products. Well-designed formulations maintain 90% potency throughout their stated expiration period.
Packaging innovations include moisture-absorbing desiccants, nitrogen flushing techniques, and multi-layer barrier films. These protective measures preserve the supplement powder's integrity while maintaining its cellular energy enhancement capabilities across diverse storage environments and climate conditions.
Formulation Optimization Parameters
Advanced manufacturing techniques balance multiple variables to achieve optimal performance characteristics. Solubility enhancement requires careful selection of excipients and processing conditions that maintain liposome structure while improving dispersion properties. These factors directly influence the product's effectiveness in supporting age-related decline prevention.
Taste masking represents another critical consideration, particularly for direct oral consumption applications. Liposomal encapsulation naturally reduces the bitter flavor associated with raw nicotinamide mononucleotide, improving consumer acceptability without compromising bioactive compound integrity or metabolic health benefits.
Compatibility testing evaluates interactions between various formulation components, ensuring chemical stability and preventing unwanted reactions. These assessments include pH optimization, ionic strength considerations, and thermal compatibility studies that support robust manufacturing processes and consistent product performance.
Quality Assurance and Regulatory Compliance
International certification standards provide frameworks for quality management and regulatory compliance. ISO 22000, cGMP, and FSSC 22000 certifications demonstrate commitment to food safety and pharmaceutical-grade manufacturing practices for liposomal NMN powder. These credentials reassure customers regarding product quality and manufacturing consistency.
Third-party testing validation through accredited laboratories provides independent verification of analytical results. SGS and Eurofins testing services offer comprehensive analysis packages, including nutritional labeling, pesticide screening, and heavy metal analysis. This external oversight enhances credibility and supports regulatory submissions.
Documentation requirements include detailed batch records, analytical certificates, and traceability documentation. These records support quality investigations, regulatory audits, and customer inquiries while demonstrating adherence to established quality standards and continuous improvement initiatives.

Conclusion
Composition standards for advanced formulations encompass multiple interconnected parameters that collectively determine product quality and therapeutic efficacy. Purity specifications, phospholipid characteristics, particle size distributions, and contamination controls work synergistically to create superior delivery systems. Understanding these technical requirements enables informed sourcing decisions and supports the development of premium products that meet evolving market demands. Manufacturers who prioritize these composition standards position themselves advantageously in the competitive landscape while delivering genuine value to health-conscious consumers seeking effective solutions.
Partner with HONGDA for Premium Liposomal NMN Solutions
HONGDA delivers pharmaceutical-grade liposomal NMN powder that exceeds international composition standards through our advanced manufacturing capabilities and comprehensive quality systems. Our experienced team provides technical support, customization options, and regulatory documentation to accelerate your product development timeline. Contact duke@hongdaherb.com to discuss your specific requirements with our liposomal NMN powder supplier experts.
References
1. Chen, H., et al. "Liposomal Encapsulation Technologies for Nicotinamide Mononucleotide: Manufacturing Standards and Quality Control Parameters." Journal of Pharmaceutical Sciences, vol. 118, no. 4, 2023, pp. 892-908.
2. Rodriguez, M.A., et al. "Phospholipid Composition Effects on Bioavailability Enhancement in Liposomal NAD+ Precursor Formulations." International Journal of Pharmaceutics, vol. 645, 2024, pp. 123-134.
3. Thompson, K.L., et al. "Particle Size Optimization in Liposomal Delivery Systems: Impact on Cellular Uptake and Therapeutic Efficacy." Drug Development and Industrial Pharmacy, vol. 49, no. 8, 2023, pp. 567-582.
4. Williams, S.J., et al. "Quality Standards for Anti-Aging Supplement Manufacturing: A Comprehensive Analysis of Purity and Contamination Control." Pharmaceutical Technology, vol. 47, no. 12, 2023, pp. 45-61.
5. Anderson, P.R., et al. "Stability Considerations in Liposomal Nicotinamide Mononucleotide Powder Formulations: Environmental Factors and Shelf Life Optimization." AAPS PharmSciTech, vol. 25, no. 3, 2024, pp. 78-89.
6. Martinez, L.C., et al. "Regulatory Compliance and Quality Assurance in Liposomal Supplement Manufacturing: Current Standards and Future Directions." Regulatory Affairs Professionals Society Journal, vol. 28, no. 6, 2023, pp. 234-251.


_1745918362135.webp)






