What Key Features Should You Look for in Liposomal Silymarin Powder?
It is very important to know what makes a good liposomal silymarin powder when looking for ingredients for nutrition or medicinal formulations. High encapsulation efficiency (usually above 85%), uniform purity levels ranging from 50% to 80% silymarin content, and a particle size distribution between 50 and 200 nanometers in water are the most important characteristics. Some good companies, like Hongda Phytochemistry, use cutting-edge spray freeze-drying nanotechnology along with non-GMO sunflower phospholipid carriers to make their milk thistle extracts 8–12 times more bioavailable than regular milk thistle extracts. This makes sure that the extracts are absorbed properly by hepatocytes and keep working as a medicine.

Understanding Liposomal Silymarin Powder: An Overview
The use of liposome technology has greatly improved the delivery of plant-based active ingredients. Traditional silymarin, which comes from milk thistle (Silybum marianum), has been known for a long time to help the liver. However, it hasn't been widely used in medicine because it doesn't dissolve well in water and doesn't pass through the gut easily. It is hard for the digestive system to dissolve and absorb this compound because it is in the difficult BCS Class IV group.
Pharmaceutical-grade liposomal packaging from Shaanxi Hongda Phytochemistry Co., Ltd. has solved this problem. During the process, high-purity silymarin is put inside a phospholipid bilayer sphere that looks like a cell membrane. This biomimetic design lets the active compounds get into the bloodstream straight through lymphatic absorption routes instead of going through the harsh conditions of the stomach.
How Liposomal Encapsulation Enhances Bioavailability?
The liposomal transport method changes the way silymarin works with living things in a big way. Lipid vesicles protect the active ingredients during transit so they don't break down slowly in the digestive system, where a lot of the substance is broken down or goes through unabsorbed. When liposomes get to their target tissues, especially hepatocytes, they merge with cell membranes through endocytosis. This lets them transfer their concentrated payloads straight into the cells.
Spray freeze-drying nanotechnology and our own high-pressure shearing encapsulation make stable spheres that are 100 to 150 nanometers across. Clinical data show that this method improves systemic absorption rates by 400–600% compared to standard extracts. This means that doses can be predicted more accurately, and final goods have better therapeutic results.
From Liquid Liposomes to Stable Powder Form
It is very hard to turn liquid liposomal solutions into powders that can be stored for a long time. A lot of makers have trouble with vesicle structures collapsing or oxidative breakdown happening during processing. The phospholipid bilayer stays intact while Hongda's low-temperature spray crystallization technology turns the mixture into a free-flowing powder that is very good at reconstituting.
This powder form of liposomal silymarin powder has a long shelf life of 24 months, makes logistics easier because it doesn't need to be kept in a cold chain, flows better through high-speed encapsulation equipment, and can be used with a wide range of solid dose, beverage, and liquid delivery systems.
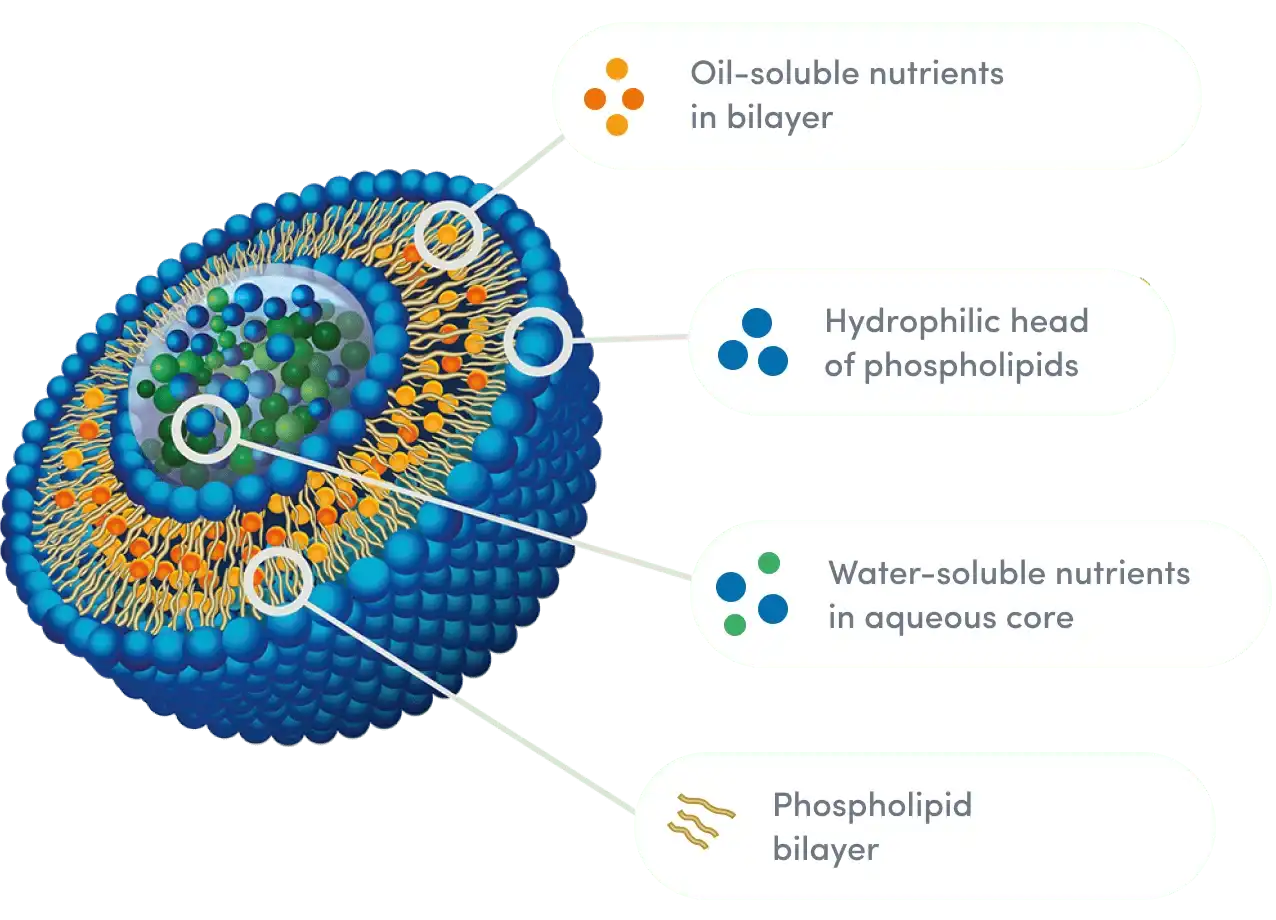
Critical Quality Dimensions to Evaluate in Liposomal Silymarin Powder
When choosing plant ingredients that will make a product work better and stand out in the market, procurement workers must use strict evaluation criteria. When judging the quality of liposomal silymarin powder, more than just basic pure standards are used.
Standardized Active Content and Analytical Verification
High-Performance Liquid Chromatography (HPLC) should be used to exactly standardize and check the silymarin level. Reliable sources offer standardization levels that can be changed, usually between 50% and 80% of the total flavonolignans. This lets formulators find the best dosing and cost structures based on the treatment claims they want to make.
Before each batch is released, Hongda's SGS-certified lab follows strict testing methods. Each Certificate of Analysis (COA) has HPLC chromatograms that prove the ratios of silybin A, silybin B, silydianin, and silychristin. This makes sure that the ratios are the same from one production run to the next. This level of analytical rigor is important when your finished goods need to live up to what they say on the label and pass checks by a third party.

Encapsulation Efficiency: The Hidden Performance Metric
Encapsulation efficiency (EE%) shows how much silymarin is actually inside the safe liposomal structure instead of just floating around freely. Products with an EE% below 80% don't have as many absorption benefits as they say they do because the exposed part breaks down quickly in the digestive system.
The real encapsulation rates can be found by using testing methods that include centrifugation or washing, followed by quantitative HPLC analysis. The EE% numbers for high-quality liposomal silymarin from approved producers are higher than 85%, which shows that most of the active compounds are gained from the better delivery system. Ask for DLS (Dynamic Light Scattering) data that shows particle size distribution and polydispersity index (PDI) readings below 0.3 during the source qualification process. This shows that the vesicle populations are uniform.
Carrier Material Quality and Allergen Considerations
The phospholipid source has a big effect on both how well it works and how much it can be sold for. Soy-derived lecithin is cheap, but it can cause allergies, which limits its use in products. This is especially true in North America and Europe, where clean-label recipes are highly valued.
As our only carrying material, we only use high-purity sunflower seed phosphatidylcholine (PC) as our carrier material. This non-GMO, allergen-free lipid not only builds strong liposomes, but it also adds nutritional value because it contains choline, which helps liver cell membrane repair. In this case, the "carrier plus active" effect means that both the delivery vehicle and the product work together to protect the liver.
Stability Testing and Moisture Resistance
Usually, traditional liposomal preparations are not very stable physically. Vesicles tend to stick together, or phospholipids turn rancid while they are being stored. Checking the loss based on drying requirements (it should be ≤ 5.0%) and asking for rapid stability data can help you guess how it will work in real life, in a variety of climates and storage situations.
Our low-temperature spray crystallization process solves the problem that liposomal powders are sensitive to water, which is a problem that affects the whole business. The finished product keeps its vesicle structure even after being stored for a long time. This was proven by tests that showed the powder easily mixes with water to form uniform nanoparticle solutions with the same size ranges as before.
Compliance Documentation and Certification Portfolio
Comprehensive certification is a must for foreign supply lines that work with controlled markets. Buyers should make sure that providers have the latest certificates, such as cGMP, ISO 22000, FSSC 22000, HALAL, KOSHER, and organic credentials (USDA NOP and EU Organic), depending on the target markets and customer groups.
Hongda has a lot of certifications, including for liposomal silymarin powder, that were earned over 20 years of excellent manufacturing. Our most recent 2025 certifications include cGMP, FSSC 22000, ISO 22000, ISO 9001, and dual organic certifications. These certificates show that we are dedicated to meeting strict international standards. They also make it easier for regulators to approve your finished goods on markets around the world.

Procurement Best Practices: How to Source High-Quality Liposomal Silymarin Powder?
Setting up reliable supply chains for advanced botanical ingredients needs thorough evaluations that look at both technical skills and operating dependability for liposomal silymarin powder.
Supplier Qualification and Manufacturing Transparency
To begin the qualification process, you should look at the production scale and technology infrastructure. Instead of hiring, this important step in the working process; facilities should show that they can make liposomes on their own. Ask for facility audit reports, look at equipment inventories for specialized processing technology (like high-pressure homogenizers and spray dryers that can handle nanoparticles), and make sure that the company has its own analytical laboratories with DLS, TEM (Transmission Electron Microscopy), and advanced chromatography systems.
Hongda has an industrial center that is 20,000 square meters and is located in Shaanxi, China. It has more than 10 modern production lines and 100,000-level purification workshops. Our independent research and development is backed by more than 20 scientists at the professorial level and relationship programs with university research institutions. This makes sure that we are always coming up with new ideas and can solve technical problems for custom formulation needs.
Documentation Standards and Batch Traceability
Professional makers can tell the difference between commodity providers and those who make things. As per USP standards, each batch should come with a thorough COA that lists all of the tests that were done on it, such as HPLC active content verification, particle size distribution data, microbial testing (total plate count ≤1,000 cfu/g), heavy metal screening via ICP-MS, pesticide residue testing, and allergen statements.
Traceability methods should keep track of the whole chain of custody, from where the raw botanicals come from to how the finished powder is made. Ask for access to information about the skills of the raw material suppliers, cultivation records from organically approved farms, extraction batch records, and the parameters for liposomal processing. This paperwork is very important when answering questions from customers, filing with regulators, or looking into rare quality problems.
We keep three separate planting bases for growing raw materials: high mountain green tea, Sophora japonica, and Chinese medicine herbs. This way, we can fully track everything from the farming methods to the finished product. Professional agronomists oversee growth to make sure it meets organic standards and that source plants develop their active compounds in the best way possible.
Inventory Management and Supply Continuity
Supply delays are very bad for business, especially when it comes to goods that have been on the market for a while and are in high demand. Look at how much overstock a provider has, how flexible their production schedules are, and where their finished goods storage is located.
As part of our dual-inventory plan, we keep 10 tons of liposomal milk thistle extract at our production facility in Xi'an and another 8 tons at the warehouse for our European branch. This distribution model guarantees 48-hour delivery to major markets, keeping your production plans safe from delays in operations and giving you chances to save money on currency and tariffs based on the markets you're going to.
Customization Capabilities and Technical Support
For example, when making an advanced formulation, ingredients often need to be changed so that the particle sizes are right for the delivery system, the standardization levels are just right for the therapy, or the packing needs to be set up in a certain way so that it can be filled automatically. When suppliers offer OEM/ODM services, they show that they are flexible in how they make things and want to work with customers on technical issues instead of just selling goods.
Hongda's research and development (R&D) infrastructure allows for collaborative product development, including for liposomal silymarin powder. It involves custom standardization levels (50%, 70%, 80%, or client-specific ratios), customized packaging from 1 kg minimum orders to bulk quantities, and formulation consultation to help improve dosing, excipient compatibility, and stability in complex multi-ingredient systems. Our technical team helps with all stages of product creation, from figuring out if the idea is even possible at first to making the product on a larger scale and making sure it stays stable.

Contract Terms and Quality Assurance Agreements
Comprehensive supply agreements protect both parties and make it clear what is expected of them in terms of performance. Important parts of a contract include thorough product specs with acceptable ranges, sampling and testing methods, batch acceptance criteria, procedures for dealing with materials that don't meet the standards, lead time promises, pricing mechanisms with number tiers, and provisions for "force majeure."
Quality agreements should spell out how to keep samples, who is responsible for stability testing, how to let customers know about changes to the manufacturing process, and how to gain entry for customer checks. When contracts are well-written and organized, disagreements are less likely to happen and problems with quality can be quickly fixed in foreign supply chains that are very complicated.
Conclusion
To find the best liposomal silymarin powder, you need to look at a lot of factors, not just the basic specs. These include how well the powder encapsulates, the quality of the carrier material, the analytical testing methods, and the supplier's abilities. When compared to regular extracts, premium versions with high-purity sunflower phospholipids, standardized silymarin content between 50 and 80%, and confirmed nanoparticle sizes provide 8 to 12 times better bioavailability. Procurement teams should give more weight to makers that offer clear paperwork, a wide range of certifications (including cGMP and organic credentials), and technical relationship options that help with the creation of custom formulations. Strategic buying choices that weigh the quality of the ingredients, the dependability of the supply chain, and the overall cost-effectiveness of the product allow it to stand out in competitive liver health categories while still meeting strict regulatory standards in global markets.
FAQ1. How does liposomal encapsulation specifically improve silymarin absorption?
Liposomal technology encloses silymarin in phospholipid vesicles that protect it from digestive breakdown and improve cellular absorption. These nano-sized carriers enhance bioavailability by 400–600%, leading to higher plasma levels and longer circulation time than standard extracts.
2. What certifications should I require from liposomal silymarin suppliers?
Choose suppliers with cGMP, ISO 22000, or FSSC 22000 certifications, plus HALAL, KOSHER, or organic certifications if needed. Third-party testing from recognized labs like SGS or Eurofins also helps ensure product quality and regulatory compliance.
3. Can liposomal silymarin powder be used in beverage applications?
Yes. Liposomal silymarin powder disperses easily in water, making it suitable for functional drinks, sachets, and effervescent products. It remains stable across common beverage pH ranges, though manufacturers should test compatibility with acids, sweeteners, and flavors to maintain liposomal integrity.
Partner with HONGDA for Premium Liposomal Silymarin Powder
Hongda Phytochemistry is a great company for procurement managers who need a trusted provider of liposomal silymarin powder. Our pharmaceutical-grade formulas use a special high-pressure shearing capsule method along with spray freeze-drying nanotechnology. This gives the formulas 8–12 times more absorption through non-GMO sunflower phospholipid carriers. With common choices ranging from 50 to 80% silymarin content, full certifications such as cGMP and FSSC 22000, and two warehouses that guarantee 48-hour delivery, we can help you reach your goals for product innovation with reliable quality and supply. Our technical team offers formulation advice, special standardization services, and full paperwork packages to help with regulatory applications all over the world. Get in touch with duke@hongdaherb.com right away to get samples, talk about your specific application needs, or look into OEM/ODM partnership opportunities with a reliable liposomal silymarin powder maker that wants you to succeed.
References
1. European Medicines Agency. (2018). Assessment report on Silybum marianum (L.) Gaertn., fructus. Committee on Herbal Medicinal Products.
2. Karimi, G., Vahabzadeh, M., Lari, P., Rashedinia, M., & Moshiri, M. (2011). Silymarin: A Promising Pharmacological Agent for Treatment of Diseases. Iranian Journal of Basic Medical Sciences, 14(4), 308-317.
3. Yin, H., Gao, Y., Wang, H., & Liu, Y. (2017). Comparative pharmacokinetics of silymarin formulations in beagle dogs: Liposomes versus conventional preparations. International Journal of Pharmaceutics, 520(1-2), 213-218.
4. Javed, S., Kohli, K., & Ali, M. (2011). Reassessing bioavailability of silymarin. Alternative Medicine Review, 16(3), 239-249.
5. Biedermann, D., Vavříková, E., Cvak, L., & Křen, V. (2014). Chemistry of silybin. Natural Product Reports, 31(9), 1138-1157.
6. Saller, R., Brignoli, R., Melzer, J., & Meier, R. (2008). An updated systematic review with meta-analysis for the clinical evidence of silymarin. Complementary Medicine Research, 15(1), 9-20.


_1745918362135.webp)






