What Properties Define High-Quality Liposomal Berberine Powder?
Superior encapsulation efficiency of more than 85%; precise particle size control within 80–150 nm measured by dynamic light scattering; phospholipid purity from non-GMO sources; full third-party testing verification; and pharmaceutical-grade manufacturing certifications such as cGMP and ISO 22000 are all signs of a high-quality liposomal berberine powder. We at Shaanxi Hongda Phytochemistry Co., Ltd. use high-pressure homogenization technology to get encapsulation rates of ≥90%. This gives us berberine in the form of a yellow powder that is more bioavailable and stable, and it stays effective for 24 months as long as it is stored properly.

Key Properties That Define High-Quality Liposomal Berberine Powder
To tell the difference between high-quality materials and less-than-perfect options, you need to carefully look at a lot of technical factors. Setting clear quality standards before negotiating with suppliers is helpful for procurement workers.
Raw Material Sourcing and Purity Verification
Quality starts with the plants that are used. The amount of berberine in raw materials changes a lot depending on when they were harvested, where they came from, and how they were handled afterward. To keep track of agricultural inputs and make sure they can be tracked, high-end makers keep specialized cultivation partnerships or direct planting bases. In Shaanxi, China, where our facilities are located, medicinal plants have been grown for hundreds of years. We run three specialized planting bases, including Chinese medicinal material farms where agronomists oversee the whole growing process, from choosing the seeds to harvesting them.
Before being put into a capsule, the berberine alkaloid should have purity levels higher than 95%, which can be checked using HPLC analysis. Heavy metal pollution is a very important issue that needs to be controlled. The levels of lead must be kept below 2 mg/kg, arsenic must be kept below 2 mg/kg, cadmium must be kept below 1 mg/kg, and mercury must be kept below 0.01 mg/kg. These limits must be checked using ICP-MS tests instead of less accurate screening methods.
Encapsulation Efficiency and Particle Characterization
Bioavailability and product stability are directly linked to how well the capsules are encapsulated. This variable measures the amount of berberine that is successfully trapped within liposomal structures compared to the amount that floats around in the matrix. Minimum standards in the industry usually say 85% effectiveness, but some advanced makers get 90% or higher by tweaking the homogenization pressure methods and the phospholipid ratio. To be sure, methods like ultracentrifugation must be used to separate the parts, and then a quantitative study of both parts is needed.
Equal care needs to be paid to particle-size variation. A dynamic light scattering study should show mean widths between 80 and 150 nm and polydispersity index values below 0.3, which means the sizes are spread out evenly. Particles bigger than 300nm lose their ability to penetrate tissue, and particles smaller than 50nm may become less stable when stored. Every output batch goes through DLS testing in our quality control labs. Along with standard certificates of analysis, these labs give customers full particle characterization data.
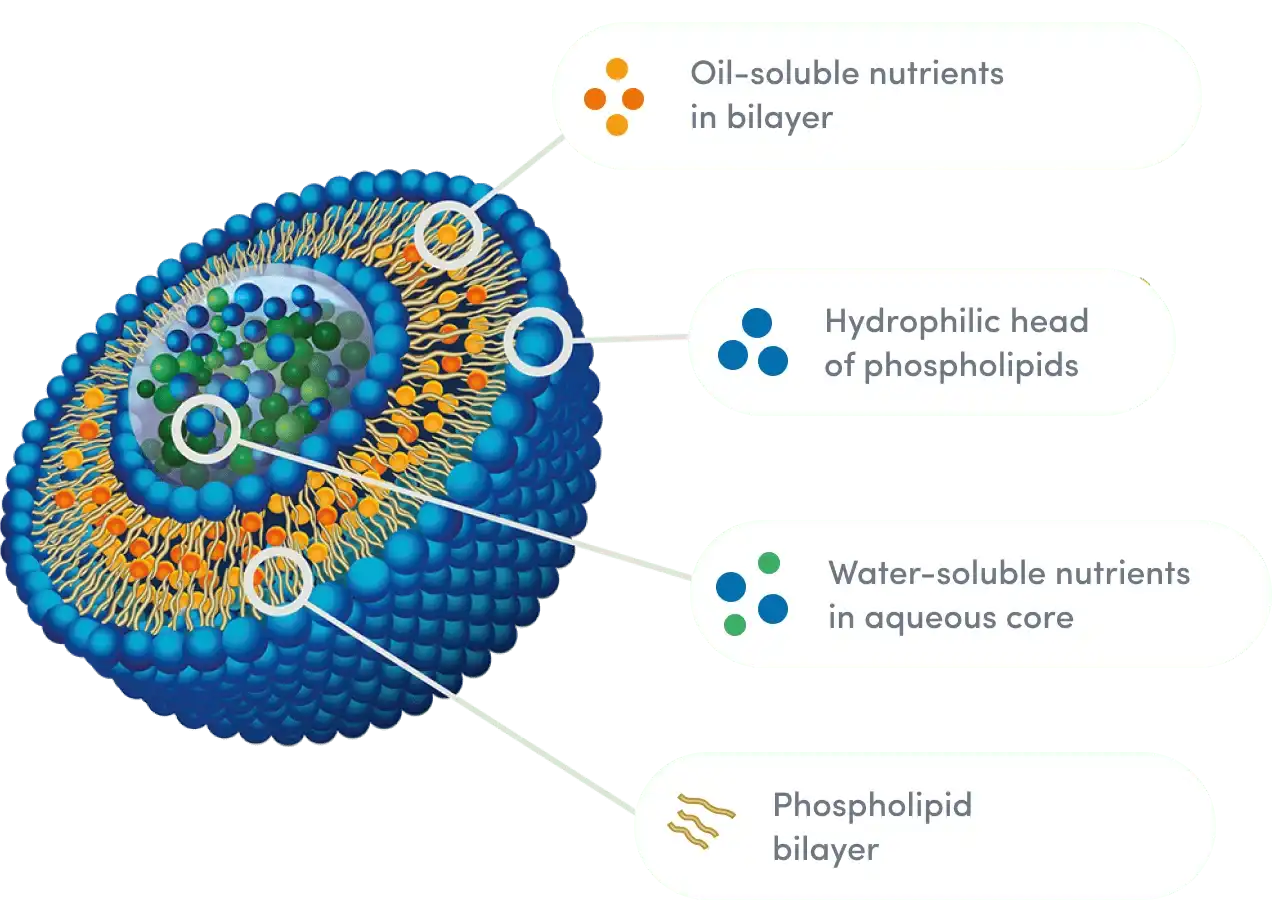
Stability Parameters and Shelf Life Validation
Stability testing tells the difference between companies that care about pharmaceutical standards and those that care more about cutting costs. Quality liposomal versions keep the amount of berberine in them between 95% and 105% of what's claimed on the label for the whole time they're on the shelf, which is usually 24 months under controlled circumstances. Accelerated stability studies at high temperatures and humidity show how things break down, while long-term tracking at room temperature and humidity confirms the expiration date.
Hongda Phytochemistry uses a special low-temperature centrifuge freeze-drying method that keeps the purity of the liposomes while the powder is being made. This process reduces the amount of oxidative degradation and structure failure that can happen with poor formulas. Choosing the right package also increases stability. For example, aluminum-lined bags with nitrogen flushing stop moisture entry and oxidation much better than regular plastic alone. Our rules say that the amount of wetness in the product can't be more than 5g/100g, and we suggest that it be stored in a cool, dry place that is out of direct sunlight.
Manufacturing Certifications and Compliance Documentation
For procurement teams that have to work in settings with a lot of rules, certifications help them control risk. cGMP approval shows that current Good Manufacturing Practices for making food supplements, such as liposomal berberine powder, are being followed. Both ISO 22000 and FSSC 22000 certify full food safety management systems that cover the whole supply chain. Certifications for HALAL and KOSHER food allow access to certain markets with particular types of customers.
Check out testing equipment and quality systems in addition to basic licenses. Manufacturers who run SGS-certified labs with a wide range of instruments, such as Waters HPLC systems, gas chromatographs, atomic absorption spectrophotometers, and microbial testing gear, show that they are committed to proof rather than just compliance. Over 20 professors work at our facility and take care of over 20 pieces of advanced testing equipment. This allows for full ingredient testing, pesticide residue screening, moisture analysis, heavy metal quantification, and microbiological validation before any lot is approved for shipment.
Comparing Liposomal Berberine Powder with Other Market Forms
Comparing things objectively across many aspects is needed to understand how competitors are positioned. To find the real value in certain situations, you have to weigh the benefits of success against the costs.
Bioavailability and Dosing Efficiency
To get effective plasma amounts of standard berberine hydrochloride powder, you need to take between 900 mg and 1500 mg of it every day, spread out over several doses. These high amounts are needed because of poor absorption, which causes tablet size problems and raises the cost per dose. Liposomal encapsulation changes this equation in a basic way; effective doses may drop to 300–500 mg per day while bloodstream levels stay the same or higher. This "low dose, high conversion" character is good for both makers and consumers: smaller capsules make it easier for people to take them, lower material costs cover the costs of encapsulation, and fewer stomach problems get rid of the main problem that keeps people from using regular berberine.
Whether to use a capsule or a powder depends on the end use. Finished-dose pills are convenient for brands that can't encapsulate their own products, but they limit the freedom of the formulation and add middle-market profits. Bulk powder lets you customize the dose, mix it with chemicals that work well together, like alpha-lipoic acid or chromium, and use it in a variety of delivery methods, such as packets, functional drinks, and medical foods. Our minimum order number of 1 kg allows for the development of the first formulation, and our 5-ton strategic reserve in both Xi'an and the US warehouses supports production needs as they grow.
Clean Label and Allergen Considerations
More and more, consumers want products that are easy to recognize and don't contain common allergens or genetic changes. Traditional liposomal recipes often use phospholipids that come from soy. This makes it hard for companies to make goods that aren't made with soy or that are aimed at people who are sensitive to legumes. Sunflower phospholipid alternatives solve this issue and meet Non-GMO Project proof standards, which lets them be sold in high-end shops like natural product stores with strict ingredient policies.
Organic approval is an extra way to set your product apart, and it's especially useful in European markets where more than 10% of supplement groups already use organic ingredients. To get certified organic, you need certified organic phospholipids and certified organic berberine source material. This limits your supplier choices and raises costs, but the market prices often make these investments worth it for brands that focus on being natural and sustainable.
Price-Value Analysis for Bulk Procurement
When products are truly different in quality and performance, price comparisons at the surface level are misleading. Standard berberine hydrochloride can cost between $45 and $65 per kilogram, while pharmaceutical-grade liposomal versions can cost between $180 and $350 per kilogram, based on the order size and specs. This seemingly higher cost needs to be rethought: when better absorption cuts the dose needed by 60–70%, the effective cost per accessible milligram becomes competitive. More savings come from fewer customer complaints, better compliance that leads to return purchases, and the possibility of higher margins from premium prices.
Price patterns are greatly affected by promises to volume for liposomal berberine powder. Tiered discounts are usually available for orders over 100 kg, and yearly supply agreements with forecasting clauses help makers get the best deals on raw materials and production schedules, which saves them money that they can share by lowering unit costs. Strategic buyers know that agreements with suppliers that offer technical support, formulation advice, and regulation paperwork create value that goes beyond just the price of the liposomal berberine powder.
Tips for Procuring High-Quality Liposomal Berberine Powder
Systematic review of suppliers lowers risk and finds partners who can help with long-term growth. We've seen hundreds of business-to-business relationships work or fail based on how well they were screened at the start.
Supplier Qualification Criteria
Before making a buy pledge, you should look over the paperwork. Ask for up-to-date copies of their production licenses, facility registrations with the right authorities (like FDA registration for goods going to the US), and active certifications such as cGMP, ISO 22000, FSSC 22000, HALAL, and KOSHER, depending on the markets you're targeting. Check the validity of the certification by searching the databases of the granting body. In foreign trade, fake credentials show up all too often, which is very concerning.
Use facility questionnaires to find out about technical skills by asking about things like output capacity, equipment specs, quality control processes, and R&D infrastructure. Manufacturers who run 100,000-grade cleaning workshops with specialized production lines show that they can control scale and contamination in a way that is good for pharmaceutical-grade ingredients. Our 20,000-square-meter building has ten modern production lines that can make more than 3,000 tons of goods each year. This makes sure that we always have enough supplies, even when demand is high at certain times of the year.
To get sample lots, make sure they come with full Certificates of Analysis that explain the test methods and results for identity confirmation (TLC, HPLC); potency (HPLC for berberine content); purity parameters (ICP-MS for heavy metals, residual solvents, and moisture); microbiological limits (total plate count, yeast/mold, and pathogens); and physical characteristics (appearance, particle size distribution, and filling efficiency). Check the manufacturer's stated specifications against an independent third-party analysis. Reliable makers welcome verification tests and may offer cost offsets for orders that meet certain criteria.

Sourcing Strategies for Volume Buyers
Strategic buying finds a mix between lowering costs and making sure the supply chain is stable. Dual-sourcing methods reduce reliance on a single provider, but the technical complexity of liposomal formulations makes approval hard. As an alternative, you could set up primary supplier relationships with confirmed backup choices for when something goes wrong. You could also keep in touch with the primary supplier and place small, regular orders to keep your qualification status.
When evaluating suppliers, ask about production wait times, normal order minimums, and capacity limits. Manufacturers who keep strategic inventory positions, like our 5-ton dynamic stock spread across various stores, can meet immediate needs within 48 hours. On the other hand, build-to-order operations may need 4 to 6 weeks from the time of the purchase order until the shipment. When deciding between domestic and international providers, you should look at the total landed costs, which include international freight, insurance, customs taxes, and inland transportation. When compared to North American or European producers, Chinese manufacturers often offer material cost benefits of 15 to 35 percent. However, longer transit times and paperwork requirements make things more complicated.
OEM and Private Label Partnership Development
Custom formulation skills add value to a provider beyond just providing raw materials. OEM partners should be able to show that they have experience making finished goods that meet your needs. This includes filling capsules, compressing tablets, packing powder, and helping with regulatory paperwork. Hongda Phytochemistry can customize soft capsules, hard capsules, tablets, premixed powders, and concentrated instant powders. We offer formulation freedom to support synergistic ingredient mixtures that are designed to achieve specific health results.
Private label services make it easier to get into new markets by using current formulas and adding your own branding, packaging, and labels. Check out the minimum order amounts, which for finished goods are usually between 5,000 and 10,000 units, as well as the different types of packing that are available, such as bottle sizes, label customization options, and other types of packaging. Comprehensive private label partners offer visual design help, regulatory review services to make sure labels meet FDA or other authority standards, and fulfillment services to handle inventory and shipping.
Creating a quality agreement makes sure that everyone knows what is expected of them when it comes to specifications, testing methods, batch release procedures, handling complaints, and managing legal changes, such as those involving liposomal berberine powder. These living papers change as relationships do, but they set clear rules for who is responsible for what, which stops mistakes that could threaten the supply chain. Long-lasting relationships that work well combine business needs with a shared dedication to quality and following the rules, knowing that your success means more business for your suppliers in the long run.

Conclusion
High-quality liposomal berberine powder is the result of a complex mix of plant knowledge, medicinal technology, and quality systems management. These qualities—high encapsulation efficiency of over 90%; precise particle size control between 80 and 150 nm; tested phospholipid purity from clean label sources; thorough testing documentation; and manufacturing certifications ranging from cGMP to FSSC 22000—set premium ingredients apart from cheaper alternatives that lower product performance and regulatory standing.
A systematic supplier review that focuses on technical skills, quality infrastructure, regulatory compliance, and a relationship approach rather than just lowering prices is helpful for procurement workers and product developers. Investing in better raw materials pays off because they improve bioavailability, which allows for "low dose, high conversion" product positioning; they also improve customer experiences, which builds brand loyalty; and they boost governmental trust, which makes it easier for companies to get into markets around the world. Concerns about metabolic health will continue to drive growth in the supplement category. Choices about which ingredients to use today will affect how competitive the category is in the years to come.
FAQ1. What certifications should I require from a liposomal berberine powder supplier?
Look for cGMP, ISO 22000, or FSSC 22000 certifications, plus HALAL or KOSHER if needed. Suppliers should also have proper business licenses, FDA registration for US markets, and third-party lab testing. Always verify certificates through official databases.
2. How does liposomal encapsulation compare cost-effectively to standard berberine?
Liposomal berberine costs 3–5 times more per kilogram than standard berberine, but its higher bioavailability allows 60–70% lower dosages. This can reduce the cost per absorbed dose while supporting premium pricing and better customer compliance.
3. What particle size range indicates optimal quality?
High-quality liposomal berberine powder typically has particle sizes between 80–150 nm with a polydispersity index below 0.3. This range supports better absorption, stability, and consistent product quality.
Partner with HONGDA for Superior Liposomal Berberine Powder Supply
HONGDA offers pharmaceutical-grade liposomal berberine powder with the highest encapsulation efficiency (>90%) in the market, precise particle sizes (80–150nm), and a number of certificates, such as cGMP, FSSC 22000, HALAL, and KOSHER. Our 20,000-square-meter factory in Shaanxi has ten modern production lines that can make 3,000 tons of goods every year. We also keep 5 tons of strategic inventory on hand to make sure that all orders are filled within 48 hours. We have been making liposomal berberine powder for a long time and can make it exactly how you want it. We offer private label services, unique formulations, and all the legal paperwork you need. Get in touch with our purchasing experts at duke@hongdaherb.com to talk about your specific needs and get full specs, prices, and sample evaluations for your next product development project.
References
1. Chen, W., Miao, Y.Q., Fan, D.J., et al. (2011). "Bioavailability study of berberine and the enhancing effects of TPGS on intestinal absorption in rats." AAPS PharmSciTech, 12(2), 705-711.
2. Kumar, A., Ekavali, Chopra, K., et al. (2015). "Current knowledge and pharmacological profile of berberine: An update." European Journal of Pharmacology, 761, 288-297.
3. Liu, Y.T., Hao, H.P., Xie, H.G., et al. (2010). "Extensive intestinal first-pass elimination and predominant hepatic distribution of berberine explain its low plasma levels in rats." Life Sciences, 87(17-18), 521-526.
4. Feng, X., Sureda, A., Jafari, S., et al. (2019). "Berberine in cardiovascular and metabolic diseases: From mechanisms to therapeutics." Theranostics, 9(7), 1923-1951.
5. Spinozzi, S., Colliva, C., Camborata, C., et al. (2014). "Berberine and its metabolites: relationship between physicochemical properties and plasma levels after administration to human subjects." Journal of Natural Products, 77(4), 766-772.
6. Maeng, H.J., Yoo, H.J., Kim, I.W., et al. (2002). "P-glycoprotein-mediated transport of berberine across Caco-2 cell monolayers." Journal of Pharmaceutical Sciences, 91(12), 2614-2621.


_1745918362135.webp)






