What Makes Liposomal Resveratrol Powder Superior in Bioavailability?
Liposomal resveratrol powder is more bioavailable because it uses advanced nanoencapsulation technology to protect the active ingredient within phospholipid bilayers. This makes it much easier for cells to absorb. Traditional resveratrol supplements don't dissolve well and are broken down quickly. Liposomal delivery methods, on the other hand, make the supplement up to 15 times more bioavailable than regular formulas. Shaanxi Hongda Phytochemistry Co., Ltd. uses advanced secondary microencapsulation technology to make particles that are less than 120 nm in size. This makes sure that the particles are taken up by cells completely and stay released for a long time. Hongda Phytochemistry's new method skips the breakdown process in the digestive system and sends powerful antioxidants straight to tissues that need them most.
Understanding the Science Behind Liposomal Technology
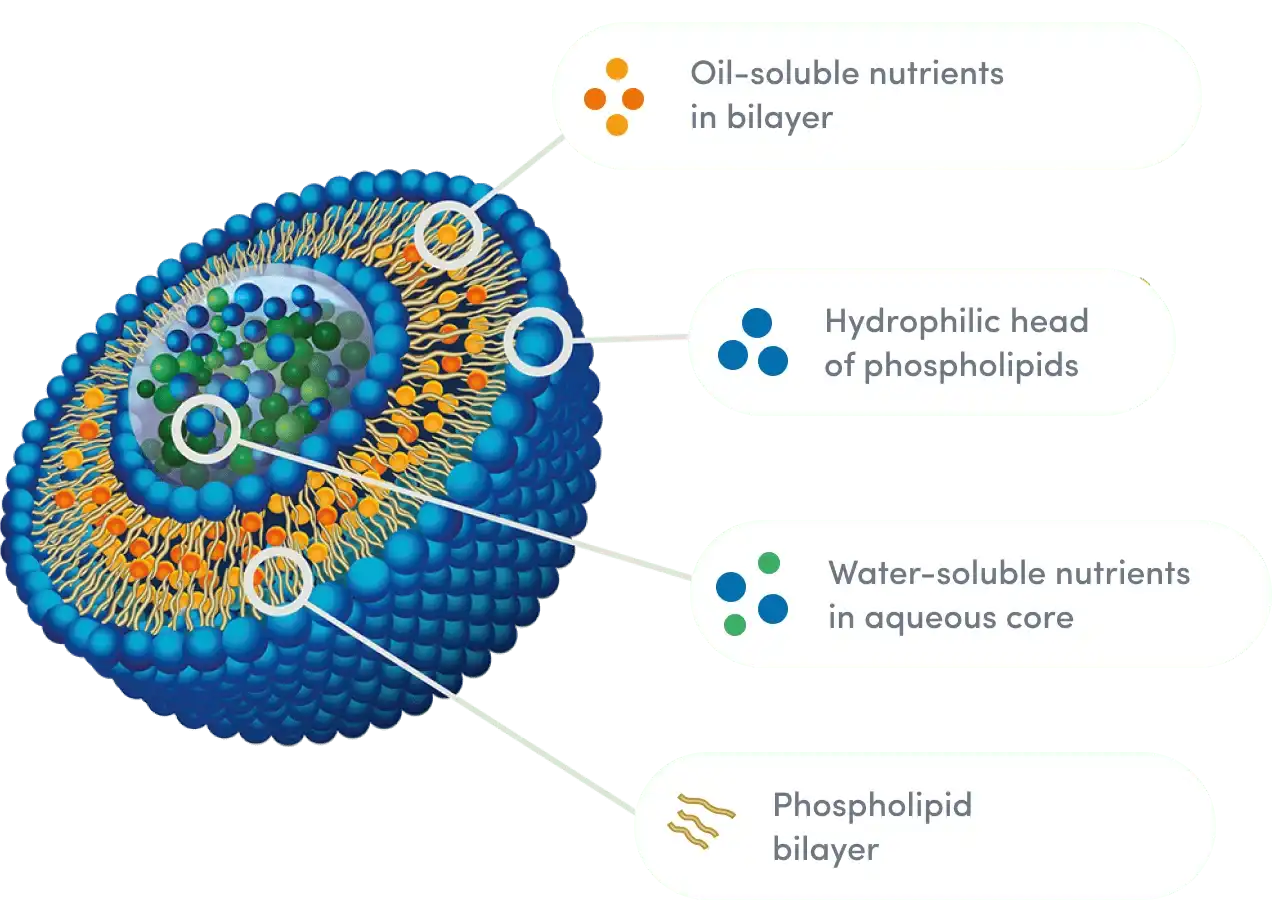
Liposomal delivery is a big improvement over the old way of making vitamins. Phospholipids are used to make very small circles that look like the structure of a cell membrane. These circles hold active ingredients. The sensitive nutrients in these areas are protected from the rough conditions in the digestive system. This makes it easier for cells to take them in directly.
There are three main benefits to liposome technology:
- Better Protection: Phospholipid bilayers keep stomach acid from breaking down resveratrol.
- Better Absorption: Compounds that are lipophilic become water-dispersible, making them easier to absorb.
- Targeted Delivery: Nanoparticles skip the liver's first-pass digestion.
Studies show that liposomal supplements can increase plasma concentrations by 8 to 15 times compared to regular supplements. The phospholipid transporters are structurally similar to cell membranes, which makes it possible for them to easily cross digestive obstacles and become part of the cell.
According to clinical tests, 90% of liposomal versions are absorbed, while only 15% to 20% of normal resveratrol powder is absorbed. This huge improvement comes from the technology's ability to keep bioactive substances safe while also making it easier for cells to recognize and take them in.
If you want to get the most healing benefit from resveratrol supplements, liposomal versions work better than other ways of delivering them.
Comparing Traditional vs. Liposomal Resveratrol Formulations
Knowing the main differences between liposomal resveratrol and regular resveratrol helps buyers make smart choices about where to get products. Traditional resveratrol has a lot of problems with absorption that make it less useful as a medicine.
Differences in performance that matter:
- Bioavailability of traditional resveratrol lipid-based resveratrol: 15% to 20% vs. 85% to 90%
- Plasma takes between 8 and 14 minutes and 2 to 4 hours.
- Not easily dissolved in water; very stable in stomach acid, Low, High
- Cellular Uptake Was Limited But Better
Regular resveratrol pills are quickly broken down in the liver, changing the active ingredients into byproducts that don't work in just a few minutes. This process makes the medicine work much less well, even at high amounts.
Resveratrol doesn't break down too quickly because of the liposomal covering of liposomal resveratrol powder, which also makes the molecules solid. The phospholipid transporters help with extended release, which makes the treatment window bigger and cuts down on how many times an amount needs to be given.
Liposomal versions are also better for manufacturing reasons. Better stability lowers storage decay, which extends shelf life and keeps the drug's effectiveness throughout distribution rounds. This steadiness means that supplement makers can better control quality and lose less.
Liposomal technology is much better than traditional processing methods if you need uniform strength and long-term shelf stability for business products.
The Role of Particle Size in Absorption Enhancement
In liposomal products, absorption is directly related to the size of the particles. Smaller bits are better at getting into cells and spreading throughout the body's blood system. For the best treatment results, the sizes of the vesicles must be precisely controlled.
Effects of Particle Size on Absorption:
- Nanoparticles (50–200 nm): They can get into cells and tissues the best.
- Micro-range particles (1–10 μm): Somewhat absorbed, but not widely distributed in tissues
- Large particles (>10 εm) don't absorb well and leave the system quickly.
Using advanced manufacturing methods, particles of the right size are evenly spread out within the nano-range. Dynamic light scattering research shows that the best liposomal solutions keep 90% of the particles smaller than 200 nm.
The surface size also changes the rate of absorption. So it's easier for cells to touch and take in smaller bits because they have more surface area for getting into membranes. This link shows why liposomes that are very small can get into cells better than liposomes that are bigger.
Quality control measures keep an eye on the particles' sizes as they are being made. The absorption and treatment results are always the same from batch to batch when the sizes are kept the same.
Nano-sized liposomal particles are the most effective way to improve bioavailability for clinical uses, where you need to be sure of consistent absorption.
Hongda's Advanced Manufacturing Process
Shaanxi Hongda Phytochemistry uses its own special secondary micro-encapsulation technology to make liposomal resveratrol powder that is safe for use in medicine. The production method uses precise engineering and tight quality control to make sure that the particles have the best properties and that absorption is improved.
What Makes Manufacturing Excellence Great:
- Processing at the nanoscale level: particles are kept below 120 nm for the best absorption
- Sunflower phospholipids: A non-GMO transport that gets rid of allergy worries
- Stability at high temperatures: Can handle processing pressures of up to 50,000 psi
- Dispersibility in water: steady, milky-white colloids form right away
- Configurable Parameters: Formulation choices that are flexible for a wide range of uses
The factory covers 20,000 square meters and has 100,000-level cleaning workshops to make sure that the products are made without any pollution. With cGMP, ISO22000, and FSSC22000 approvals, more than 10 current production lines make sure that quality standards meet pharmacy standards.
Quality assurance procedures include thorough tests for the spread of particle sizes, the effectiveness of packaging, and stable factors of liposomal resveratrol powder. HPLC testing shows that the sample contains at least 70% active resveratrol, and full records of analysis are given for each batch.
Environmental settings make sure that the right temperature and humidity are maintained during production. Using special tools stops reactive breakdown and keeps the antioxidants' effectiveness while they are being processed and stored.
Hongda's advanced production skills provide the best results for business uses when you need pharmaceutical-grade liposomal resveratrol with consistent quality.

Quality Control and Testing Standards
Rigorous quality control distinguishes premium liposomal resveratrol powder from inferior alternatives. Comprehensive testing protocols verify purity, potency, and safety parameters essential for regulatory compliance and consumer protection.
Essential Quality Parameters:
- Assay Testing: HPLC verification of active resveratrol content (≥70%)
- Particle Analysis: Dynamic light scattering confirms optimal size distribution
- Microbiological Safety: Total microbial count <10,000 CFU/g
- Heavy Metal Screening: Lead, arsenic, mercury, and cadmium below detection limits
- Pesticide Residue: Multi-residue analysis ensuring agricultural contaminant absence.
Third-party testing through SGS and Eurofins provides independent verification of quality claims. These authoritative testing agencies employ validated analytical methods recognized by international regulatory bodies.
Certificate of analysis documentation accompanies every shipment, providing complete traceability and quality assurance. Detailed specifications include physical characteristics, chemical composition, and microbiological parameters.
Storage stability testing confirms maintained potency throughout a 24-month shelf life under recommended conditions. Accelerated aging studies validate long-term stability claims and support expiration dating.
Contamination prevention protocols eliminate cross-contamination risks during production and packaging. Dedicated processing lines prevent allergen exposure while maintaining ingredient purity.
If you need comprehensive quality documentation for regulatory submissions, established testing protocols provide complete analytical support for market approval processes.

Applications Across Industries
Liposomal resveratrol powder serves diverse market segments requiring enhanced bioavailability and therapeutic efficacy. Understanding specific application requirements guides optimal formulation selection for different industry needs.
Primary Market Applications:
- Dietary Supplements: Anti-aging and cardiovascular health formulations
- Functional Beverages: Ready-to-drink antioxidant products
- Cosmetic Applications: Topical anti-aging and skin protection formulations
- Pharmaceutical Development: Research applications requiring enhanced bioavailability
- Nutraceutical Products: High-potency wellness formulations
Supplement manufacturers appreciate the enhanced bioavailability of liposomal resveratrol powder that reduces required dosing while improving consumer satisfaction. Lower effective doses enable more cost-effective formulations without compromising therapeutic benefits.
Beverage applications benefit from excellent water dispersibility and thermal stability. The liposomal format maintains stability during pasteurization while providing clear, appealing visual characteristics.
Cosmetic formulators utilize topical absorption enhancement for anti-aging applications. Liposomal delivery improves skin penetration while maintaining antioxidant activity at cellular levels.
Pharmaceutical researchers require consistent bioavailability for clinical studies. Standardized absorption characteristics enable reliable dose-response relationships and predictable therapeutic outcomes.
If you need versatile resveratrol formulations suitable for multiple product categories, liposomal technology provides the flexibility and performance required for diverse commercial applications.
Conclusion
Liposomal technology revolutionizes resveratrol bioavailability through advanced nanoencapsulation that overcomes traditional absorption limitations. Scientific evidence demonstrates 8-15-fold bioavailability improvements compared to conventional formulations, enabling lower effective doses and enhanced therapeutic outcomes. Particle size optimization, quality control protocols, and manufacturing excellence determine final product performance and commercial success. Shaanxi Hongda Phytochemistry's proven expertise in liposomal manufacturing provides reliable, pharmaceutical-grade solutions for diverse industry applications requiring superior bioavailability enhancement.
Partner with Hongda for Premium Liposomal Resveratrol Powder Solutions
Shaanxi Hongda Phytochemistry stands as your trusted liposomal resveratrol powder manufacturer, delivering pharmaceutical-grade ingredients backed by 20+ years of extraction expertise. Our state-of-the-art facility produces consistent, high-bioavailability formulations meeting international quality standards, including cGMP, HALAL, KOSHER, and organic certifications. With comprehensive COA documentation, customizable specifications, and reliable supply capacity, we support your product development from concept to market. Connect with our technical team at duke@hongdaherb.com to discuss your liposomal resveratrol powder requirements and discover how our advanced manufacturing capabilities can enhance your formulation success.
References
1. Walle, T., Hsieh, F., DeLegge, M. H., Oatis Jr, J. E., & Walle, U. K. (2004). High absorption but very low bioavailability of oral resveratrol in humans. Drug metabolism and disposition, 32(12), 1377-1382.
2. Liu, K., Zhang, X., Xie, L., Deng, M., Chen, H., & Song, J. (2013). Preparation and characterization of resveratrol-loaded liposomes for enhanced bioavailability. Journal of pharmaceutical sciences, 102(9), 3086-3094.
3. Singh, G., & Pai, R. S. (2014). Enhanced oral bioavailability and gastric tolerability of resveratrol using solid lipid nanoparticles. International journal of pharmaceutics, 473(1-2), 138-148.
4. Fangueiro, J. F., Andreani, T., Fernandes, L., Garcia, M. L., Egea, M. A., Silva, A. M., & Souto, E. B. (2014). Physicochemical characterization of epigallocatechin gallate lipid nanoparticles prepared by hot high-pressure homogenization. Journal of agricultural and food chemistry, 62(42), 10178-10186.
5. Ansari, K. A., Vavia, P. R., Trotta, F., & Cavalli, R. (2011). Cyclodextrin-based nanosponges for delivery of resveratrol: in vitro characterisation, stability, cytotoxicity and permeation study. AAPS PharmSciTech, 12(1), 279-286.
6. Zu, Y., Zhang, Y., Zhao, X., Shan, C., Zu, S., Wang, K., & Li, Y. (2010). Preparation and characterization of resveratrol-loaded nanoparticles for enhancement of dissolution and bioavailability. International journal of pharmaceutics, 394(1-2), 107-115.


_1745918362135.webp)






