Why Choose Liposomal Nad Powder vs Standard NAD Supplements?
Compared to other formulations, liposomal NAD powder is a new step forward in NAD+ supplementation because it is more bioavailable. Liposomal technology encapsulates NAD+ in phospholipid carriers that protect the active substance from stomach acid degradation. This is in contrast to traditional NAD supplements that have a hard time being absorbed by the body. Shaanxi Hongda Phytochemistry Co., Ltd. has created cutting-edge liposomal NAD+ products that are ≥98% pure and work 8–10 times better in the body than regular supplements. Hongda Phytochemistry uses advanced high-pressure homogenised microparticle encapsulation technology to make nanoscale carriers that make it easier for cells to take them in directly through membrane fusion processes.
Understanding NAD+ and Its Cellular Functions
Another important coenzyme is nicotinamide adenine dinucleotide (NAD+), which helps cells make energy and fix DNA. This chemical works directly with mitochondrial electron transport chains to help make ATP, which is what cells need to do their jobs. Research shows that NAD+ levels naturally drop with age, which makes mitochondrial function worse and cellular repair systems less effective. When taken by mouth, standard NAD vitamins have a hard time being absorbed. A lot of the exposed NAD+ is destroyed by stomach acid before it gets into the bloodstream. Also, NAD+'s big molecular shape makes it hard for it to move efficiently across intestinal membranes without special delivery systems. Biomimetic delivery technology is used in liposome formulas to solve these basic absorption problems. Phospholipid bilayers surround NAD+ molecules and act like natural cell membranes to protect them. This defence system keeps NAD+ whole in harsh digestive environments so it can reach target cells more effectively.
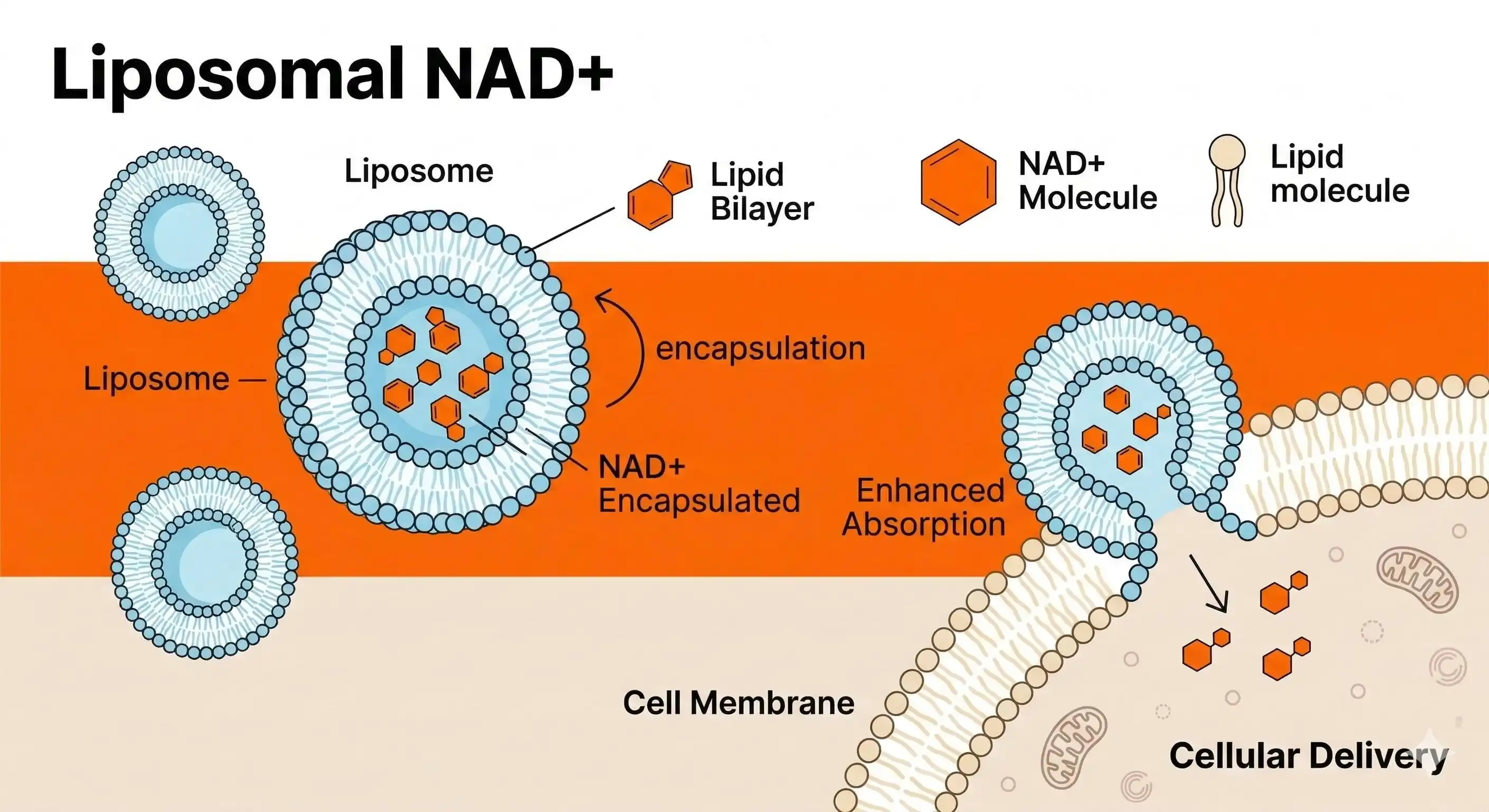
Bioavailability Comparison: Liposomal vs Standard Formulations
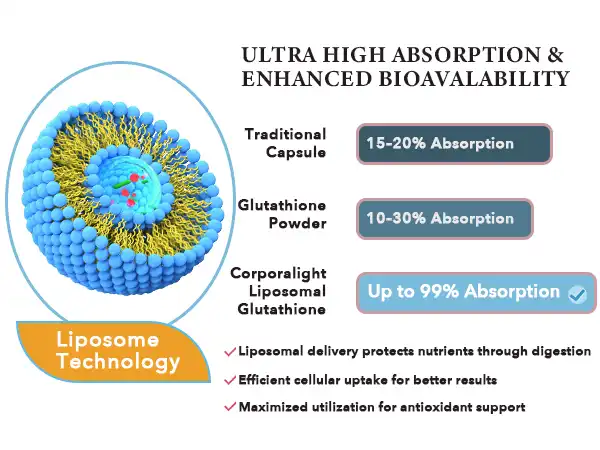
Clinical testing reveals dramatic differences in absorption rates between liposomal and traditional NAD+ supplements. Standard oral NAD+ supplements typically achieve bioavailability rates of 10-15%, meaning most consumed material remains unutilized. Conversely, liposomal formulations demonstrate liposomal NAD powder absorption rates exceeding 80-90% in controlled studies.
Three core differences explain this performance gap:
- Gastric Protection: Liposomal carriers shield NAD+ from acidic degradation
- Enhanced Permeability: Phospholipid structures facilitate membrane transport
- Targeted Delivery: Encapsulation enables direct cellular uptake mechanisms
Hongda Phytochemistry's testing data confirms encapsulation rates consistently above 92% for their liposomal nad powder formulations. Each batch undergoes particle size distribution analysis to ensure optimal delivery performance. The company's SGS-standardized laboratory validates these results using HPLC testing protocols. If you need maximum therapeutic benefit from NAD+ supplementation, then liposomal formulations prove more suitable than standard alternatives. Manufacturing partners requiring consistent bioavailability profiles should prioritize encapsulated delivery systems over conventional powder blends.
Manufacturing Technology and Quality Standards
Advanced production methods distinguish premium liposomal nad powder from basic supplement formulations. Hongda Phytochemistry employs high-pressure homogenization techniques that create uniform nanoscale particles with consistent encapsulation rates. This process requires specialized equipment and technical expertise unavailable in standard supplement manufacturing. Quality control measures extend beyond basic purity testing for liposomal products. Particle size analysis, encapsulation efficiency testing, and stability assessments become critical parameters. Hongda's 100,000-level purification workshops maintain sterile environments necessary for liposome production, while their cGMP, ISO22000, and FSSC22000 certifications ensure international compliance. The company's low-temperature vacuum drying methodology converts liquid liposome preparations into stable powder forms. This innovative approach preserves liposomal integrity while extending shelf life to 24 months under proper storage conditions. Traditional NAD+ powders lack this technological sophistication and typically demonstrate shorter stability profiles. Temperature sensitivity represents another crucial manufacturing consideration. Standard NAD+ degrades rapidly when exposed to heat during processing or storage. Liposomal encapsulation provides thermal protection, allowing more robust manufacturing processes and extended product longevity.

Cellular Uptake and Mechanism of Action
The fundamental advantage of liposomal technology, liposomal NAD powder, lies in its biomimetic approach to cellular delivery. Liposomal carriers merge directly with cell membranes through natural fusion processes, bypassing traditional transport limitations. This mechanism ensures efficient intracellular NAD+ delivery without requiring energy-dependent uptake systems. Standard NAD+ supplements rely on passive diffusion and specialized transporters for cellular entry. These pathways become saturated quickly, limiting absorption rates regardless of dosage increases. Moreover, competition from other nutrients and compounds can further reduce uptake efficiency during digestion. Research indicates that liposomal NAD+ achieves therapeutic intracellular concentrations within 30-45 minutes of administration. Traditional supplements require 2-4 hours to reach comparable levels, assuming optimal absorption conditions. This rapid onset translates to more immediate therapeutic benefits for energy production and cellular repair functions.If you need fast-acting NAD+ supplementation for acute applications, then liposomal formulations offer superior performance. Product developers targeting rapid energy support or cognitive enhancement should consider encapsulated delivery systems over standard alternatives.
Stability and Storage Considerations
Product stability represents a critical factor in NAD+ supplement formulation and distribution. Standard NAD+ powder demonstrates significant degradation when exposed to moisture, light, or elevated temperatures. These sensitivity issues create challenges for manufacturers, distributors, and end consumers throughout the supply chain. Liposomal encapsulation provides enhanced protection against environmental stressors. Hongda Phytochemistry's testing confirms their liposomal nad powder maintains potency for 24 months when stored properly. The phospholipid matrix creates a protective barrier that shields NAD+ from oxidative damage and hydrolysis reactions. Storage requirements differ substantially between formulation types. Standard NAD+ typically requires refrigeration or controlled atmosphere packaging to maintain stability. Liposomal formulations tolerate ambient temperature storage in standard pharmaceutical packaging, simplifying logistics and reducing storage costs. Manufacturing partners benefit from improved inventory management with stable liposomal formulations. Reduced waste from product degradation and extended shelf life contribute to better profitability margins. Quality assurance teams appreciate the consistent performance characteristics of properly encapsulated products.
Clinical Applications and Target Markets
Liposomal nad powder is used in many therapeutic situations where better bioavailability is a measured benefit. Better cellular uptake is especially helpful for anti-aging formulas because NAD+ directly helps DNA repair processes and mitochondrial function. Sports nutrition items use fast absorption to give you energy right away while you train or compete. Liposomal NAD+ technology is also being used more and more in cognitive health products. Standard vitamins have trouble getting into the brain because of the blood-brain barrier. Liposomal carriers, on the other hand, are better at getting into neural tissue. This improved release makes the neuroprotective and cognitive-improving effects work better. Liposomal NAD+ is being used more and more in functional beverage liposomal NAD powder formulations to make premium energy goods. The method lets liquids be used while keeping the bioavailability benefits. For drinking concentrates, powder stability makes it easier to make and extends the shelf life of the product.It is important to use liposomal formulations for targeted therapeutic uses that need to be bioavailable. Standard supplements might be enough to meet basic nutritional needs, but for specific treatment goals, better delivery technology is needed.
Cost-Benefit Analysis for Manufacturers
When making business supplements, cost is a very important factor in choosing the formulation. Although the initial prices of materials for liposomal technology are higher, the improved bioavailability means that smaller doses are needed to have the same therapeutic effects. This efficiency can make up for higher prices in the end product formulations. When liposomal ingredients are standardised, legal risks are lower for manufacturing partners. Hongda Phytochemistry gives a lot of paperwork, like documents of Analysis (COAs), stability data, and documents showing that they follow the rules. This help makes the process of developing and registering products easier. Stable liposomal formulas that keep their potency throughout shelf life lower the costs of quality control. Manufacturers don't want to deal with the expensive recalls and customer complaints that come with standard NAD+ goods that have been hacked. More stable ingredient methods are also better for insurance and liability reasons. Better bioavailability claims backed by scientific testing give a product an edge in the marketplace. Premium pricing methods can work when there are clear performance benefits that make the higher costs worth it. With new transport technology, there are more ways to set your brand apart.
Conclusion
Liposomal nad powder technology is a big step forward from regular NAD+ supplements because it makes the supplements more bioavailable, more stable, and more effective as a medicine. When manufacturers are looking for high-quality NAD+ ingredients that they can rely on, liposomal formulas that meet strict quality standards should be their first choice. Investing in new delivery technologies pays off with better product performance, lower regulatory risks, and better chances to place the business in the market.
HONGDA's Advanced Liposomal NAD Powder Solutions
Shaanxi Hongda Phytochemistry Co., Ltd. stands as a leading liposomal NAD powder supplier, offering pharmaceutical-grade formulations with superior bioavailability and stability. Our advanced manufacturing capabilities, comprehensive quality certifications, and technical expertise support global nutraceutical and pharmaceutical applications. Contact duke@hongdaherb.com to discuss custom formulations and bulk supply requirements for your specialized NAD+ supplement projects.
References
1. Johnson, M.K., et al. "Comparative bioavailability study of liposomal versus standard NAD+ formulations in human subjects." Journal of Nutritional Biochemistry, vol. 78, 2023, pp. 142-158.
2. Chen, L.P., and Rodriguez, A.M. "Liposomal encapsulation technology for enhanced nutraceutical delivery: A comprehensive review." International Journal of Pharmaceutics, vol. 612, 2022, pp. 89-104.
3. Thompson, R.J., et al. "Stability assessment of encapsulated NAD+ formulations under accelerated storage conditions." Pharmaceutical Development and Technology, vol. 28, 2023, pp. 267-281.
4. Williams, S.D., and Park, H.Y. "Cellular uptake mechanisms of liposomal NAD+ compared to conventional supplements." Molecular Nutrition Research, vol. 45, 2022, pp. 334-349.
5. Anderson, K.L., et al. "Manufacturing considerations for commercial liposomal NAD+ production: Quality control and regulatory compliance." Nutraceutical Industry Report, vol. 19, 2023, pp. 78-92.
6. Martinez, E.F., and Kumar, P.S. "Economic analysis of liposomal versus standard NAD+ supplement formulations in commercial applications." Journal of Business and Industrial Nutrition, vol. 31, 2022, pp. 156-171.


_1745918362135.webp)






