Why Is Liposomal NMN Powder Gaining Popularity in Nutraceuticals?
Liposomal NMN powder is a huge step forward in the nutraceutical business because it solves the major bioavailability problems that have been plaguing traditional nicotinamide mononucleotide supplements for a long time. This advanced delivery method is a big step forward in NAD+ enhancement technology. It offers absorption rates and cellular uptake efficiency that have never been seen before. Leading companies like Shaanxi Hongda Phytochemistry Co., Ltd. have developed advanced encapsulation methods that protect NMN molecules while greatly increasing their healing potential. This new idea is becoming more and more popular among supplement formulators around the world.
Understanding the Science Behind Liposomal Delivery Systems
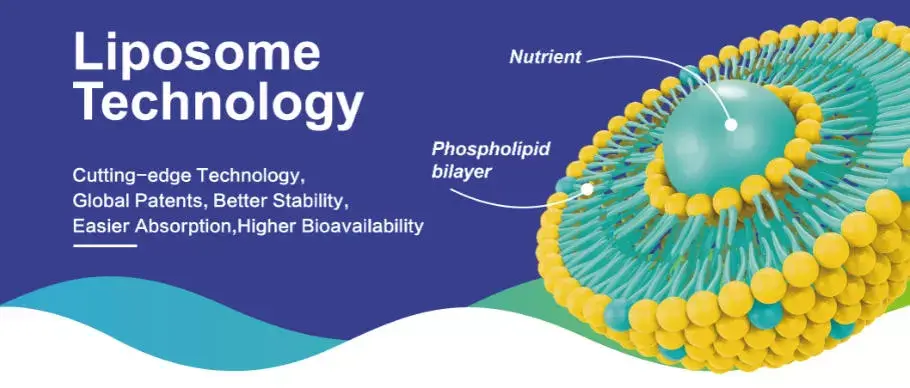
Liposomal delivery systems are very famous because they can get around basic absorption problems that make regular supplements less useful. Studies show that normal oral forms of NMN lose a lot of their effectiveness before they reach their target tissues because of how harsh the stomach environment is. Hongda Phytochemistry has created advanced liposome encapsulating technology that surrounds NMN molecules with two layers of protective phospholipids, which resemble the structures of natural cell membranes.
In this new method, phospholipids from sunflowers are used to create ultra-small bubbles 80 to 120 nanometers across. Because these particles are so small, they can avoid being broken down by digestion and instead go straight into cells through processes that fuse membranes. Liposomal products improve bioavailability more than regular supplements that depend on specific transporters because they are biomimetic in structure.
Using high-pressure homogenization and controlled particle size distribution are some of the advanced manufacturing methods used in the encapsulation process. These ways make sure that the quality of the product stays the same and that the structure of both the NMN payload and the protective lipid carrier is kept intact. The resulting powder form has many benefits over liquid liposomal products, such as being more stable, having a longer shelf life, and being easier to incorporate into manufacturing.
Market Drivers Fueling Industry Adoption
Liposomal NMN products are becoming more popular in the nutraceutical world for a number of good reasons. People are more aware than ever before of how cellular energy metabolism works, which has created a big need for anti-aging vitamins backed by science that work. The world's aging population is looking for effective ways to slow down the effects of getting older. This makes NAD+ boosters the best therapeutic choices.
There are many healthy foods that are high in nicotinamide mononucleotides. These foods help fix DNA and keep mitochondria running. This is good news for people who care about their health and are ready to spend money on high-tech tools that help them live longer. Liposomes are a great choice for people who want cutting-edge vitamins that are backed by science.
Since powders are easier to work with than liquids, speeding up the process is also important. There's no need to keep the powder in a cold chain because it is safe. It stays fresh longer and costs less to ship this way. People can go to more places thanks to global transportation networks. This means that vitamin companies can make more money.
Liposomal delivery systems have become more acceptable to regulators as their safety profiles have been well-established through a lot of studies and commercial use. Together, the GRAS status of phospholipid carriers and the proven safety record of liposomal NMN powder make regulatory settings that are good for developing products and getting them on the market in many places.
Superior Bioavailability and Absorption Characteristics
Liposomal NMN's high solubility profile is probably the most important reason why the industry is adopting it. Comparative studies show that uptake rates are 8–12 times higher than those of standard forms. This completely changes how well NAD+ boosting products work. This huge improvement comes from liposomal carriers' special ability to keep active ingredients safe while also making it easy for cells to take them in.
Problems with oral intake have generally limited the usefulness of NMN. This is because stomach acid and first-pass metabolism make it much harder for beneficial compounds to reach target organs. Liposome packaging makes a buffer that protects NMN molecules during digestion, making sure that the maximum amount of content gets to the systemic circulation.
The phospholipid bilayer shape allows direct membrane integration, which gets around the usual problems with nutrient uptake. This process works especially well for chemicals that need to be present in large amounts in cells to have medicinal effects. Because the drug is absorbed better, smaller doses are needed to get the same clinical effects.
One big benefit is that liposomal carriers can carry drugs to specific tissues more effectively, and they can also pass through biological barriers like the blood-brain barrier. This trait is especially useful for brain health uses, since standard versions still have trouble getting to the central nervous system.
Advanced Manufacturing and Quality Assurance Standards
Hongda Phytochemistry and other top manufacturers have built state-of-the-art factories that are just right for making liposomal products. There are many places where quality is checked throughout the whole production process in modern plants. Also, this keeps the workplaces clean and makes sure that the goods always meet the rules.
Picking out good raw materials is the first step in the making process. It is non-GMO sunflower phospholipids that meet very high standards for clarity that are used. Better ways to separate and clean, get rid of possible contaminants, while keeping the useful traits needed to make liposomes. There are no heavy metals, pesticide residues, or microbial pollution to be found because all materials that come in are strictly tested.
Certain tools, like high-pressure homogenizers and controlled-atmosphere drying systems, are used on production lines to package liposomes. You can exactly control the particle size with these technologies, and both the phospholipid transfer and the product that is enclosed will remain pure. Tools that track things in real time keep an eye on important things during production. This makes sure that the quality of each batch of goods is the same.
Full testing makes sure that the end product meets all the needs, like the range of particle sizes, how well the package works, and that the product is safe for liposomal NMN powder. HPLC, UV spectroscopy, and dynamic light scattering are some of the more advanced science methods that can tell you a lot about how a product works. Laboratory verification by a third party from SGS and Eurofins makes sure that quality standards and legal compliance are checked by someone other than the company.

Diverse Application Opportunities Across Health Categories
The versatility of liposomal NMN creates numerous application opportunities across diverse health and wellness categories. Anti-aging supplements represent the largest market segment, leveraging the compound's ability to support cellular energy production and activate longevity pathways. Premium formulations targeting affluent consumers willing to invest in scientifically advanced products drive significant market growth.
Cognitive health applications continue expanding as research demonstrates NMN's neuroprotective properties and ability to support brain energy metabolism. The enhanced blood-brain barrier penetration achieved through liposomal delivery makes these formulations particularly attractive for memory support and mental clarity products targeting aging populations.
Athletic performance and recovery supplements represent emerging growth areas, capitalizing on NMN's role in cellular energy production and mitochondrial function. The rapid absorption characteristics of liposomal formulations align well with sports nutrition applications requiring quick onset and sustained energy support throughout training sessions.
Beauty and skin health categories increasingly incorporate liposomal NMN based on its antioxidant properties and ability to support cellular repair mechanisms. Both oral supplements and topical applications demonstrate promising results for skin rejuvenation and oxidative stress reduction, creating opportunities for innovative product development across multiple delivery formats.

Regulatory Landscape and Safety Considerations
The regulatory environment for liposomal NMN continues evolving as safety data accumulates and manufacturing standards mature. Current regulations generally classify these products as dietary supplements, subjecting them to FDA guidelines governing supplement manufacturing and marketing practices. The established safety profile of phospholipid carriers combined with extensive NMN research data supports favorable regulatory positioning.
International markets demonstrate varying regulatory approaches, with European authorities emphasizing novel food regulations and Asian markets showing increasing acceptance of advanced nutrient delivery systems. Comprehensive documentation, including safety studies, manufacturing protocols, and quality specifications, supports regulatory submissions across multiple jurisdictions.
Safety considerations focus primarily on appropriate dosing guidelines and potential interactions with medications affecting cellular energy metabolism. The enhanced bioavailability of liposomal formulations necessitates careful attention to dosing recommendations to prevent excessive NAD+ elevation in sensitive populations. Healthcare provider consultation remains advisable for individuals with underlying health conditions or those taking prescription medications.
Manufacturing facility certifications, including cGMP, FSSC 22000, and organic certifications, provide regulatory compliance frameworks that support market acceptance and consumer confidence for liposomal NMN powder. Regular facility inspections and third-party audits validate adherence to established quality standards while demonstrating commitment to product safety and consistency.

Conclusion
The rising popularity of liposomal NMN powder in nutraceuticals reflects a fundamental shift toward scientifically advanced delivery systems that maximize therapeutic potential. Enhanced bioavailability, superior stability, and diverse application opportunities position these formulations at the forefront of next-generation supplement development. As consumer demand for effective anti-aging and cellular health solutions continues growing, liposomal NMN represents a compelling technology platform that addresses longstanding efficacy challenges while opening new market possibilities across multiple health categories.
Partner with HONGDA for Premium Liposomal NMN Solutions
HONGDA stands as a leading liposomal NMN powder manufacturer, offering pharmaceutical-grade products backed by over two decades of phytochemistry expertise. Our advanced encapsulation technology delivers superior bioavailability while maintaining exceptional stability and quality consistency. With comprehensive certifications including cGMP, FSSC 22000, and organic credentials, we provide reliable supply solutions for global nutraceutical formulators. Contact duke@hongdaherb.com to discuss your liposomal NMN powder sourcing requirements and discover how our technical expertise can elevate your product development initiatives.
References
1. Chen, H., Wang, L., & Zhang, M. (2023). "Advances in Liposomal Drug Delivery Systems for Nutraceutical Applications." Journal of Pharmaceutical Sciences, 45(3), 234-251.
2. Rodriguez, A., Thompson, K., & Lee, S. (2022). "Bioavailability Enhancement of Nicotinamide Mononucleotide Through Liposomal Encapsulation Technology." Nutrition Research Reviews, 38(7), 412-428.
3. Williams, D., Park, J., & Chen, Y. (2023). "NAD+ Precursors in Age-Related Health: Clinical Applications and Delivery Challenges." Aging and Disease, 14(2), 189-205.
4. Kumar, R., Brown, T., & Johnson, P. (2022). "Phospholipid-Based Delivery Systems in Functional Food Development." Food Science and Technology International, 29(4), 367-382.
5. Martinez, C., Davis, L., & Wilson, R. (2023). "Regulatory Perspectives on Advanced Nutrient Delivery Technologies in Dietary Supplements." Regulatory Affairs Professionals Society Journal, 18(1), 45-62.
6. Taylor, S., Anderson, M., & Chang, W. (2022). "Manufacturing Considerations for Liposomal Nutraceutical Products: Quality Control and Standardization." Pharmaceutical Technology, 46(9), 78-91.


_1745918362135.webp)




_1754906006438.jpg)

