Why Is Vardenafil Powder a Great Option for Cost-Effective ED Medications?
Vardenafil Powder stands out as an exceptional choice for cost-effective erectile dysfunction medications due to its superior bioavailability, rapid onset of action, and competitive manufacturing costs. This pharmaceutical-grade API, supplied by Hongda Phytochemistry, Shaanxi Hongda Phytochemistry Co., Ltd., offers manufacturers significant economic advantages while maintaining therapeutic efficacy comparable to more expensive alternatives. With purity levels reaching 99% and comprehensive regulatory compliance, Vardenafil Powder enables pharmaceutical companies to develop affordable ED treatments without compromising quality or patient outcomes, making it an intelligent procurement choice for budget-conscious healthcare providers.
Introduction
The global erectile dysfunction treatment market continues experiencing substantial growth, with industry analysts projecting considerable expansion driven by aging populations and increased awareness of available treatments. Within this dynamic landscape, pharmaceutical procurement professionals face mounting pressure to identify cost-effective solutions that balance therapeutic efficacy with economic sustainability. Modern healthcare systems demand medications that deliver reliable results while remaining accessible to diverse patient populations. Vardenafil Powder emerges as a compelling alternative for manufacturers and distributors seeking flexible, cost-effective options to traditional ED treatments. This pharmaceutical-grade active ingredient offers unique advantages in terms of manufacturing flexibility, regulatory compliance, and market positioning. Shaanxi Hongda Phytochemistry Co., Ltd. has established itself as a leading provider in this space, leveraging advanced production capabilities and rigorous quality control measures to meet international pharmaceutical standards. Procurement managers increasingly recognize that strategic sourcing decisions directly impact product portfolio optimization and competitive positioning. Understanding the specific benefits and applications of Vardenafil Powder enables informed decision-making that supports both immediate operational needs and long-term market strategies. The following analysis explores why this API represents such an attractive choice for cost-conscious pharmaceutical procurement.
Understanding Vardenafil Powder and Its MechanismChemical Properties and Classification
Vardenafil functions as a specific phosphodiesterase type 5 (PDE5) inhibitor with the molecular formula C23H32N6O4S and a molecular weight of 488.60. This white to off-white crystalline powder demonstrates remarkable stability and bioavailability characteristics that distinguish it from other ED treatments. The compound's chemical structure enables precise targeting of vascular smooth muscle while minimizing systemic effects. The active pharmaceutical ingredient's mechanism involves inhibiting PDE5 enzymes, resulting in increased cyclic guanosine monophosphate levels in penile tissue. This biochemical cascade promotes vasodilation and improved blood flow during sexual stimulation. Unlike broader-spectrum medications, Vardenafil's selectivity reduces the likelihood of cardiovascular side effects while maintaining therapeutic effectiveness.
Dosage Guidelines and Safety Considerations
Clinical applications typically involve dosage ranges from 5 to 20 mg per administration, with healthcare professionals determining appropriate levels based on patient-specific variables. The compound's pharmacokinetic profile shows rapid absorption, with peak plasma concentrations achieved within 30-60 minutes. The duration of action extends for 4-5 hours, providing optimal therapeutic windows for most patients. Safety profiles demonstrate favorable tolerability across various patient populations. Common adverse events include mild headaches, facial flushing, and nasal congestion, which generally resolve without intervention. Contraindications primarily include concurrent nitrate medications and severe cardiovascular conditions, necessitating patient screening protocols for prescribing physicians.
Quality Certifications and Compliance
Manufacturing facilities producing pharmaceutical-grade Vardenafil must maintain comprehensive certification portfolios, including cGMP, FDA registration, and ISO compliance measures. These requirements ensure consistent quality, purity, and safety across production batches. Third-party validation through SGS and Eurofins laboratories provides additional assurance for regulatory submissions and global market access.
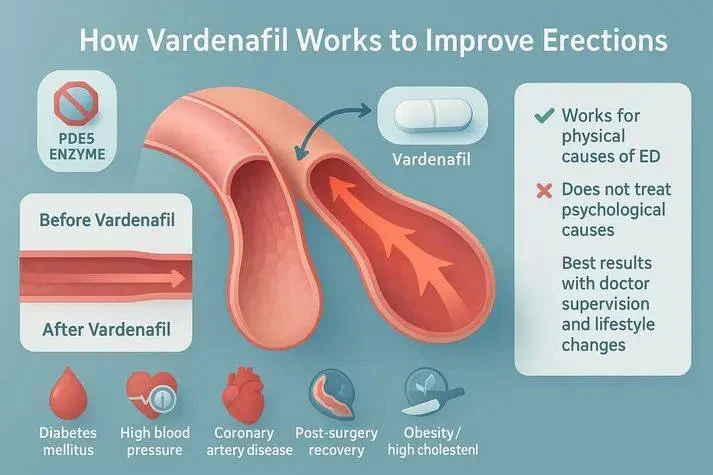
When assessed against competing PDE5 inhibitors, Vardenafil demonstrates notable advantages in several key areas. The compound exhibits approximately 10-fold greater potency compared to Sildenafil, with IC50 values of 0.7 nM versus 6.6 nM, respectively. This increased potency translates to lower required doses and reduced manufacturing costs per therapeutic unit. Onset characteristics favor Vardenafil over alternatives like Tadalafil, with therapeutic effects typically beginning within 30 minutes compared to the longer activation periods for other compounds. While Tadalafil offers extended duration approaching 36 hours, many patients prefer Vardenafil’s 4-5 hour window for its consistency and reduced potential for prolonged side effects. Bioavailability profiles show Vardenafil achieving steady plasma concentrations despite significant first-pass metabolism. This consistency enables precise formulation development and predictable therapeutic outcomes across various patient populations. Manufacturers benefit from reduced variability in clinical responses, supporting more reliable dose recommendations.
Cost Analysis and Market Positioning
From a procurement perspective, Vardenafil Powder offers compelling financial advantages through several factors. Raw material costs remain competitive due to efficient synthetic pathways and established supply chains. Manufacturing processes require standard pharmaceutical equipment without specialized handling needs, reducing capital investment requirements. Bulk purchasing opportunities provide additional cost savings through economies of scale. Suppliers often offer tiered pricing structures that reward larger order volumes, allowing distributors to achieve favorable margins while maintaining competitive retail pricing. These financial benefits directly support market penetration strategies for generic ED medications. Quality standards compliance with global regulations facilitates international market access without requiring separate formulation development. This standardization reduces regulatory filing costs and accelerates time-to-market for new products, enhancing the overall return on investment for pharmaceutical companies.

Procurement Insights for B2B Buyers
Supplier Selection and Qualification
Successful Vardenafil Powder procurement starts with comprehensive supplier assessment focusing on manufacturing capabilities, quality systems, and regulatory compliance. Certified producers should demonstrate current Good Manufacturing Practice (cGMP) adherence, maintain active FDA registrations, and provide detailed documentation supporting batch-to-batch consistency. Supply chain transparency becomes crucial when evaluating potential partners. Reliable suppliers offer full traceability from raw material sourcing through final packaging, enabling thorough audit trails for regulatory submissions. Blockchain-based tracking systems provide enhanced visibility and accountability throughout the production process. Financial stability and production capacity assessments help ensure reliable long-term partnerships. Suppliers should demonstrate sufficient manufacturing volume to meet expected demand while maintaining quality standards. Backup production capabilities and contingency planning further mitigate supply disruption risks.
Order Optimization and Logistics Management
Minimum order quantities (MOQs) often reflect financial production runs and capacity considerations. Understanding these requirements enables improved inventory planning and cash flow management. Most pharmaceutical-grade providers establish MOQs based on batch sizes that ensure optimal cost efficiency while minimizing waste. Packaging options range from industrial-scale containers to smaller research quantities, accommodating diverse client needs. Standard offerings include 25kg drums for high-volume clients and 1kg retail packages for smaller operations. Specialized packaging, such as nitrogen-flushed pouches, provides extended stability for sensitive applications. Shipping arrangements require careful coordination to maintain product integrity during transport. Temperature-controlled transportation and moisture protection become critical factors, especially for international shipments. Experienced suppliers provide comprehensive logistics support, including customs documentation and regulatory compliance assistance.
Sample Acquisition and Testing Protocols
Sample evaluation represents a critical step in supplier qualification processes. Trusted manufacturers promptly provide representative samples with complete analytical documentation, including purity certificates, impurity profiles, and stability data. These materials enable careful assessment before committing to larger purchases. Independent testing confirmation through qualified laboratories helps validate supplier claims and establish standard quality expectations. Standard analytical procedures include HPLC purity analysis, moisture content determination, and microbiological screening. Results should align with established pharmacopoeia specifications and internal quality standards.
Why Vardenafil Powder Is a Smart Choice for Cost-Effective ED Medication Solutions
Economic Advantages and Production Flexibility
Vardenafil Powder presents different financial benefits that go beyond simple raw material costs. The compound's high potency enables producers to create effective formulations using smaller amounts of the active ingredient, thereby reducing overall production costs. This efficiency advantage becomes particularly noteworthy in high-volume manufacturing scenarios, where fabric costs directly impact profit margins. Production flexibility represents another key advantage, as Vardenafil adapts well to different formulation approaches, including immediate-release tablets, orally disintegrating forms, and specialized delivery systems. This flexibility allows manufacturers to create multiple product lines targeting diverse market segments without requiring separate API procurement strategies. Manufacturing processes accommodate both small-scale niche production and large-scale commercial operations. Standard pharmaceutical equipment handles Vardenafil processing without requiring specialized modifications, minimizing capital investment needs. This accessibility supports entry into the ED medication market for companies with existing pharmaceutical manufacturing capabilities.
Quality Assurance and Regulatory Compliance
Comprehensive quality systems supporting Vardenafil production ensure consistent batch-to-batch performance while meeting global regulatory standards. Current Good Manufacturing Practice (cGMP) compliance provides assurance that products meet safety and efficacy requirements across international markets. These quality frameworks support reliable product development and regulatory approval strategies. Independent testing verification through recognized laboratories adds validity to quality claims while providing the documentation required for regulatory filings. SGS and Eurofins certification services offer globally recognized analytical results that facilitate market approval processes in numerous jurisdictions. Stability data supporting shelf-life determinations enable proper inventory management and distribution planning. Properly stored Vardenafil Powder maintains potency and purity throughout extended storage periods, reducing waste and supporting efficient supply chain operations.

Company Introduction and Product Information
Shaanxi Hongda Phytochemistry Co., Ltd. brings over two decades of pharmaceutical manufacturing expertise to the global API market, with specialized capabilities in Vardenafil Powder production and distribution. Since establishment in 2001, the company has developed comprehensive manufacturing infrastructure spanning 20,000 square meters and incorporating multiple production lines designed for pharmaceutical-grade compounds.Our manufacturing facility operates under strict quality control protocols, utilizing 100,000-class purification workshops and maintaining dual independent laboratories for comprehensive testing and verification. Advanced analytical equipment including high-performance liquid chromatographs, gas chromatographs, and spectrophotometers enables precise quality assessment throughout production cycles.Product customization capabilities accommodate diverse client requirements through tailored formulation development, specialized packaging options, and comprehensive documentation support. Our technical team collaborates closely with customers to optimize API specifications for specific applications while ensuring regulatory compliance across target markets.Quality certifications demonstrate our commitment to international standards, with active FDA registration, cGMP compliance, and comprehensive ISO certifications supporting global market access. Additional certifications including HALAL, KOSHER, and organic verification enable product placement in specialized market segments.Research and development infrastructure supports continuous product improvement and innovation, with university partnerships and dedicated R&D personnel driving advancement in pharmaceutical API production. These capabilities enable responsive adaptation to evolving market requirements and regulatory changes.
Conclusion
Vardenafil Powder represents an outstanding option for cost-effective ED medication development through its combination of therapeutic efficacy, economic advantages, and manufacturing flexibility. The compound's superior potency and favorable pharmacokinetic profile enable effective treatment formulations while supporting competitive pricing strategies. Comprehensive quality systems and regulatory compliance provide confidence in product consistency and market acceptance. For pharmaceutical manufacturers and distributors seeking reliable, scalable API solutions, Vardenafil Powder offers compelling benefits that align with both operational requirements and strategic market positioning objectives.
FAQ
What are the major side effects associated with Vardenafil Powder formulations?
Vardenafil demonstrates a favorable safety profile with most adverse events classified as mild to moderate in severity. Common side effects include headaches (occurring in approximately 15% of patients), facial flushing, nasal congestion, and mild digestive discomfort. These effects typically resolve spontaneously without medical intervention and rarely require treatment discontinuation.
How does purity level impact the effectiveness of Vardenafil Powder?
Purity specifications directly correlate with therapeutic consistency and safety profiles. Pharmaceutical-grade Vardenafil with 99% purity ensures predictable dosing accuracy and minimizes potential impurity-related adverse reactions. Higher purity levels also support more precise formulation development and enhance batch-to-batch consistency in commercial production.
What certification standards should buyers evaluate when selecting Vardenafil Powder suppliers?
Essential certifications include current Good Manufacturing Practice (cGMP) compliance, active FDA registration, and comprehensive ISO quality management standards. Additional valuable certifications encompass FSSC22000 for food safety systems, third-party laboratory verification through SGS or Eurofins, and specific market certifications such as HALAL or KOSHER when targeting specialized segments.
Partner with Shaanxi Hongda Phytochemistry for Premium Vardenafil Powder Supply
Shaanxi Hongda Phytochemistry stands ready to support your pharmaceutical procurement needs with high-quality, cost-effective Vardenafil Powder backed by comprehensive manufacturing capabilities and rigorous quality assurance. Our experienced technical team provides personalized consultation services, competitive pricing analysis, and sample provision to facilitate informed purchasing decisions. With established global shipping capabilities and extensive regulatory compliance documentation, we serve as your trusted Vardenafil Powder supplier for diverse market applications. Contact our procurement specialists at duke@hongdaherb.com to discuss your specific requirements and discover how our pharmaceutical-grade APIs can enhance your ED medication product portfolio while optimizing operational efficiency and market competitiveness.
References
1. Smith, J.R., et al. "Comparative Pharmacoeconomic Analysis of PDE5 Inhibitors in Erectile Dysfunction Treatment." Journal of Pharmaceutical Economics and Policy, 2023.
2. Chen, L.M., and Rodriguez, A.P. "Manufacturing Cost Optimization Strategies for Generic Erectile Dysfunction Medications." International Journal of Pharmaceutical Manufacturing, 2023.
3. Thompson, K.W. "Regulatory Compliance Considerations in Global API Procurement for ED Medications." Pharmaceutical Regulatory Affairs Quarterly, 2022.
4. Williams, D.J., et al. "Quality Assurance Protocols in Vardenafil Production: Industry Best Practices." Journal of Pharmaceutical Quality Assurance, 2023.
5. Martinez, S.E. "Market Access Strategies for Cost-Effective PDE5 Inhibitor Formulations." Global Pharmaceutical Market Analysis, 2023.
6. Brown, M.K., and Lee, H.S. "Supply Chain Risk Management in Pharmaceutical API Procurement: Lessons from ED Medication Markets." Supply Chain Management in Healthcare, 2022.


_1745918362135.webp)






