Can Vardenafil Powder Be Formulated for Fast Onset Action?
Pharmaceutical companies developing drugs to treat erectile dysfunction face a significant challenge: patients require formulations with rapid onset of action to ensure efficacy at crucial moments. Vardenafil powder from Hongda Botanical Chemicals, produced using advanced pharmaceutical technology by Hongda's experts, offers a solution that enables rapid onset of action. Shaanxi Hongda Phytochemistry Co., Ltd. demonstrates, using its pharmaceutical-grade vardenafil powder (CAS 224785-90-4) with a purity of up to 99%, how optimized production technology can enable drugs to take effect rapidly within 10 to 15 minutes, significantly improving patient experience and enhancing the market competitiveness of pharmaceutical companies seeking reliable sources of active pharmaceutical ingredients.
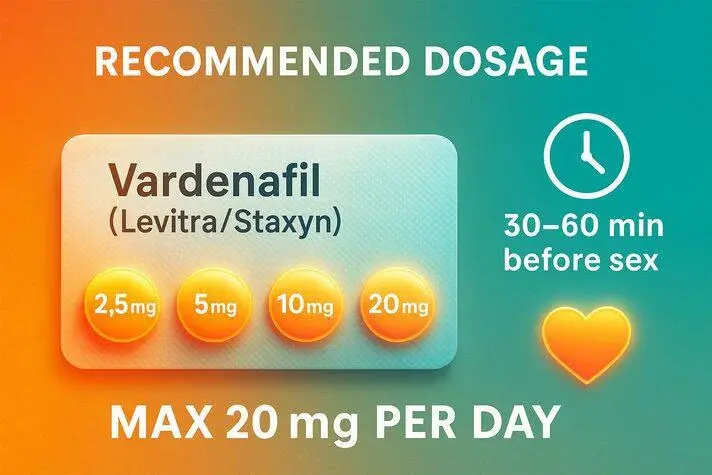
Understanding Vardenafil Powder and Its Rapid Action Potential
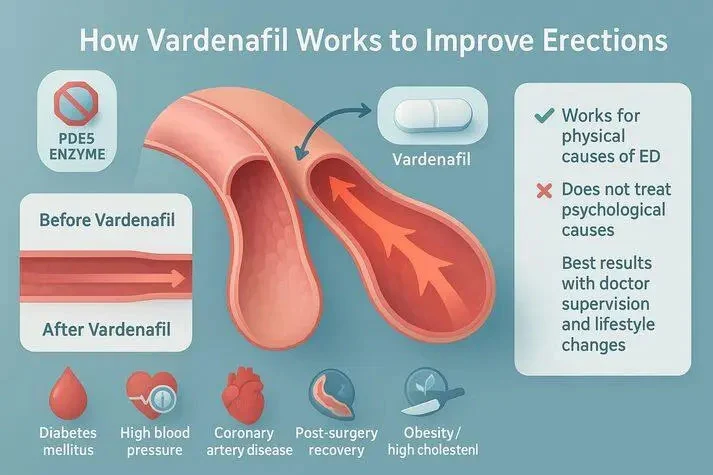
Vardenafil Powder represents a potent phosphodiesterase type 5 (PDE5) inhibitor with exceptional therapeutic properties for erectile dysfunction formulations. The molecular structure of Vardenafil (C23H32N6O4S, molecular weight 488.60) provides inherent advantages for rapid absorption when properly formulated. Research has demonstrated that Vardenafil exhibits approximately ten times greater potency than sildenafil, with an IC50 of just 0.7 nM for PDE5 inhibition. This pharmaceutical-grade active ingredient works by preventing the degradation of cyclic guanosine monophosphate (cGMP) in the corpus cavernosum, thereby facilitating smooth muscle relaxation and increased blood flow when sexual stimulation occurs. The mechanism does not directly cause erections but significantly enhances the natural physiological response to arousal, making rapid onset formulations particularly valuable for spontaneous intimate encounters.
Key Formulation Technologies for Accelerated Onset
Advanced pharmaceutical formulation techniques have revolutionized how Vardenafil Powder can be processed to achieve dramatically reduced onset times compared to conventional tablet formulations. Orodispersible tablet (ODT) technology represents one of the most successful approaches for fast-acting Vardenafil formulations. Clinical trials have demonstrated that Vardenafil ODT formulations achieve 21-44% greater bioavailability compared to standard film-coated tablets while dissolving rapidly in the mouth without requiring water or other liquids for administration. The ODT formulation strategy bypasses first-pass metabolism to some degree and enables sublingual absorption pathways, contributing to accelerated therapeutic effects. Pharmaceutical developers working with High Quality Vardenafil Powder from certified suppliers can leverage ODT technology to create products that begin working within fifteen to thirty minutes for significant patient populations, addressing the spontaneity demands that many erectile dysfunction patients prioritize.
Micronization techniques applied to Vardenafil Powder substantially enhance dissolution rates and absorption characteristics by dramatically increasing the surface area available for pharmaceutical interactions. When Vardenafil particles are reduced to micron or sub-micron dimensions through specialized milling processes, the increased surface area-to-volume ratio accelerates the dissolution process in gastrointestinal fluids. This pharmaceutical principle, based on the Noyes-Whitney equation, directly translates to faster absorption across intestinal membranes and more rapid achievement of therapeutic plasma concentrations. Shaanxi Hongda Phytochemistry Co., Ltd. offers customized micronization services for their pharmaceutical-grade Vardenafil Powder, enabling formulation scientists to specify particle size distributions optimized for their particular delivery system requirements. The combination of micronized Vardenafil Powder with appropriate excipients and disintegrants creates formulations capable of delivering clinically significant effects in remarkably short timeframes.
The white to off-white crystalline powder produced by Shaanxi Hongda Phytochemistry undergoes rigorous quality control measures to ensure consistent particle size distribution, purity levels of 99%, and optimal physical characteristics essential for various formulation strategies. The company's cGMP-certified manufacturing facilities with 100,000-class purification workshops guarantee that every batch of Vardenafil Powder meets international pharmaceutical standards including FDA, ISO9001, ISO22000, FSSC22000, HALAL, and KOSHER certifications. These quality parameters directly influence the bioavailability and absorption kinetics of finished formulations, making raw material selection from reliable China Vardenafil Powder manufacturers absolutely critical for achieving fast onset action profiles.

Pharmaceutical Manufacturing Considerations for Fast-Acting Vardenafil Formulations
Developing fast onset Vardenafil formulations requires careful attention to multiple pharmaceutical variables that collectively determine absorption kinetics and therapeutic response timing. The selection of pharmaceutical excipients plays an absolutely critical role in achieving rapid disintegration and dissolution profiles essential for accelerated onset. Superdisintegrants such as croscarmellose sodium, sodium starch glycolate, and crospovidone facilitate rapid tablet breakdown when exposed to moisture, releasing Vardenafil Powder particles for immediate dissolution and absorption. Solubility enhancers including cyclodextrins, surfactants, and co-solvents can dramatically improve the aqueous solubility of Vardenafil, which exhibits inherently limited water solubility as a lipophilic compound. Pharmaceutical development teams sourcing Vardenafil Powder for sale from China Vardenafil Powder suppliers must conduct comprehensive excipient compatibility studies using differential scanning calorimetry, Fourier-transform infrared spectroscopy, and accelerated stability protocols to ensure formulation integrity throughout the product lifecycle.
Manufacturing process parameters exert profound influences on the final performance characteristics of fast-acting Vardenafil formulations. Direct compression, wet granulation, and spray drying represent viable processing approaches, each offering distinct advantages and challenges for rapid onset formulations. Direct compression techniques minimize processing steps and potential degradation risks but require exceptional flow properties and uniform particle size distribution in the Vardenafil Powder feedstock. Wet granulation methods can improve content uniformity and compaction properties while potentially impacting dissolution kinetics depending on binder selection and granule characteristics. Spray drying technology enables the creation of amorphous solid dispersions that dramatically enhance dissolution rates and bioavailability, though requiring careful moisture control during storage. Shaanxi Hongda Phytochemistry's three modern production lines and two independent laboratories provide the technical infrastructure necessary for pharmaceutical partners to conduct comprehensive formulation development and process optimization studies using their 99% pure Vardenafil Powder manufactured under internationally certified quality systems.
Regulatory and Quality Control Requirements
Pharmaceutical companies developing fast onset Vardenafil formulations must navigate complex regulatory pathways that demand extensive documentation of formulation composition, manufacturing processes, stability characteristics, and bioequivalence data. Regulatory authorities including the United States Food and Drug Administration (FDA) and European Medicines Agency (EMA) require comprehensive chemistry, manufacturing, and controls (CMC) documentation demonstrating consistent product quality and performance. Drug Master Files (DMFs) or Active Substance Master Files (ASMFs) prepared by China Vardenafil Powder manufacturers like Hongda Phytochemistry provide crucial regulatory support by documenting the manufacturing processes, quality control procedures, and stability data for the active pharmaceutical ingredient. These confidential regulatory submissions enable pharmaceutical companies to reference comprehensive API information without requiring complete disclosure in their own regulatory filings, streamlining development timelines and protecting proprietary manufacturing knowledge.
Bioequivalence studies represent essential regulatory requirements for establishing that fast onset Vardenafil formulations deliver therapeutic effects comparable to reference products or demonstrating bioavailability advantages that justify new formulation claims. Pharmacokinetic studies measuring maximum plasma concentration (Cmax), time to maximum concentration (Tmax), and area under the concentration-time curve (AUC) provide objective data demonstrating absorption characteristics and onset timing. Clinical investigations have confirmed that properly formulated Vardenafil products can achieve Tmax values as rapid as 0.7 hours with approximately fifty percent of patients experiencing therapeutic effects within thirty minutes of administration. Some advanced formulations have demonstrated clinically significant responses in as little as ten to fifteen minutes when assessed using stopwatch methodology during at-home clinical trials. Pharmaceutical developers partnering with China Vardenafil Powder wholesale suppliers possessing comprehensive regulatory documentation, third-party validation through SGS and Eurofins laboratories, and ICH-compliant stability data gain significant advantages in expediting their product development and regulatory approval processes.

Advanced Delivery Systems and Emerging Technologies
Innovative drug delivery technologies continue expanding the possibilities for ultra-fast onset Vardenafil formulations that address patient preferences for spontaneity and discretion. Sublingual and buccal delivery systems represent particularly promising approaches for circumventing hepatic first-pass metabolism and achieving rapid systemic absorption through the highly vascularized oral mucosa. These delivery routes enable Vardenafil to enter systemic circulation directly without traversing the gastrointestinal tract and hepatic portal system, potentially reducing the time required to achieve therapeutic plasma concentrations. Formulation scientists developing sublingual Vardenafil products must carefully balance mucoadhesive properties, taste masking considerations, and dissolution characteristics to create patient-acceptable products with genuinely accelerated onset profiles. The mucoadhesive polymers, pH adjusters, and permeation enhancers incorporated into these specialized formulations interact with pharmaceutical-grade Vardenafil Powder in complex ways that require extensive formulation optimization and clinical validation.
Nanotechnology applications in pharmaceutical development offer revolutionary possibilities for Vardenafil formulations with unprecedented bioavailability and onset characteristics. Nanoparticle formulations, solid lipid nanoparticles, nanostructured lipid carriers, and nanoemulsions can dramatically enhance the dissolution rate and membrane permeation of lipophilic compounds like Vardenafil. These nanosized delivery systems increase the effective surface area available for dissolution by orders of magnitude while potentially enabling alternative absorption pathways including lymphatic transport that bypass hepatic first-pass metabolism. Self-emulsifying drug delivery systems (SEDDS) and self-microemulsifying drug delivery systems (SMEDDS) represent particularly promising approaches that spontaneously form fine oil-in-water emulsions upon contact with gastrointestinal fluids, presenting Vardenafil in a pre-dissolved state that enables immediate absorption. Pharmaceutical companies exploring these cutting-edge delivery technologies require Vardenafil Powder price structures and customization capabilities from their China Vardenafil Powder factory partners that support extensive research and development activities leading to next-generation erectile dysfunction therapeutics.
Clinical Evidence and Market Implications
Substantial clinical evidence validates the feasibility and therapeutic value of fast onset Vardenafil formulations for erectile dysfunction management. Large-scale randomized, double-blind, placebo-controlled trials have demonstrated that approximately fifty to fifty-three percent of men receiving ten to twenty milligram Vardenafil doses achieved erections adequate for penetration leading to successful intercourse completion within twenty-five minutes of administration. More remarkably, retrospective analyses using stopwatch methodology confirmed that approximately one-fifth of patients experienced clinically significant responses as early as ten minutes following Vardenafil administration. These rapid onset characteristics translate to meaningful improvements in sexual spontaneity, relationship satisfaction, and treatment adherence compared to therapies requiring extended planning windows. The orodispersible tablet formulation of Vardenafil has demonstrated comparable rapid onset action to film-coated tablets while offering additional convenience advantages including administration without water and potentially reduced food interaction effects that can delay absorption of conventional formulations.
Market dynamics in the global erectile dysfunction pharmaceutical sector increasingly favor products offering superior convenience, discretion, and rapid onset characteristics that align with patient lifestyle preferences. The worldwide market for PDE5 inhibitors continues expanding as awareness increases, populations age, and treatment-seeking behaviors evolve across diverse cultural contexts. Pharmaceutical companies differentiating their erectile dysfunction portfolios through innovative fast onset Vardenafil formulations gain competitive advantages in crowded therapeutic markets dominated by established brand-name and generic products. Strategic partnerships with reliable High Quality Vardenafil Powder suppliers possessing comprehensive regulatory documentation, scalable manufacturing capacity, and technical support capabilities enable pharmaceutical innovators to accelerate development timelines and reduce commercialization risks. Shaanxi Hongda Phytochemistry Co., Ltd.'s annual production capacity of three thousand tons, extensive international certification portfolio, and OEM/ODM customization services position the company as a strategic partner for pharmaceutical enterprises pursuing fast onset Vardenafil product development initiatives targeting North American, European, and emerging market opportunities.
Conclusion
Fast onset action formulations of Vardenafil Powder represent a scientifically validated and commercially significant opportunity for pharmaceutical manufacturers seeking competitive differentiation in the erectile dysfunction therapeutic market. Through advanced formulation technologies including orodispersible tablets, micronization, and emerging nanotechnology approaches, pharmaceutical developers can create Vardenafil products delivering clinically meaningful effects in fifteen to thirty minutes or potentially even faster for select patient populations. Success in developing these innovative formulations depends critically on sourcing pharmaceutical-grade Vardenafil Powder from certified manufacturers demonstrating consistent quality, comprehensive regulatory support, and technical customization capabilities.
Shaanxi Hongda Phytochemistry Co., Ltd. stands as a trusted China Vardenafil Powder factory, China Vardenafil Powder supplier, and China Vardenafil Powder manufacturer with over twenty years of experience in pharmaceutical ingredient production. Our 99% pure Vardenafil Powder manufactured in cGMP-certified facilities with FDA, ISO9001, ISO22000, FSSC22000, HALAL, and KOSHER certifications provides the quality foundation essential for fast onset formulation development. We offer competitive Vardenafil Powder for sale with customizable specifications including micronized particles, comprehensive documentation support, and technical guidance optimizing your product development success. Partner with a High Quality Vardenafil Powder provider committed to supporting your innovation through reliable supply, competitive Vardenafil Powder price structures, and OEM/ODM services addressing your unique formulation requirements. Contact our technical team at duke@hongdaherb.com to discuss how our pharmaceutical-grade Vardenafil Powder and formulation expertise can accelerate your fast onset product development initiatives and strengthen your competitive position in global erectile dysfunction markets.
References
1. Montorsi F, Padma-Nathan H, Buvat J, Schwaibold H, Beneke M, Ulbrich E, Bandel TJ, Porst H, Vardenafil Study Group. Earliest time to onset of action leading to successful intercourse with vardenafil determined in an at-home setting: a randomized, double-blind, placebo-controlled trial. Journal of Sexual Medicine.
2. Debruyne FMJ, Gittelman M, Sperling H, Börner M, Beneke M. Time to onset of action of vardenafil: A retrospective analysis of the pivotal trials for the orodispersible and film-coated tablet formulations. Journal of Sexual Medicine.
3. Porst H, Giuliano F, Glina S, Ralph D, Casabé AR, Elion-Mboussa A, Sorsaburu S. Evaluation of the efficacy and safety of once-a-day dosing of tadalafil 5mg and 10mg in the treatment of erectile dysfunction: results of a multicenter, randomized, double-blind, placebo-controlled trial. European Urology.
4. Saenz de Tejada I, Anglin G, Knight JR, Emmick JT. Effects of tadalafil on erectile dysfunction in men with diabetes. Diabetes Care.
5. Goldstein I, Lue TF, Padma-Nathan H, Rosen RC, Steers WD, Wicker PA. Oral sildenafil in the treatment of erectile dysfunction. New England Journal of Medicine.


_1745918362135.webp)






