What Are Technical Attributes of Liposomal Coenzyme Q10 Powder?
Liposomal coenzyme Q10 powder is a pharmaceutical-grade ingredient that is made using nanoscale capsule technology (D50 < 120 nm), which ensures that at least 70% of the CoQ10 is pure within phospholipid carriers produced from sunflowers. Its technical features include better water dispersibility that forms stable colloids, extreme thermal stability that can withstand pressures of more than 50,000 psi during tabletting, uniform particle size through 20-mesh screening, and a longer shelf life of 24 months when stored properly. These standards make sure that bioavailability is high and that production is consistent for B2B uses, as provided by Hongda Phytochemistry.

Understanding Liposomal Coenzyme Q10 Powder: Technical Overview
Nutritional science has come a long way thanks to improvements in CoQ10 delivery methods. Liposomal technology is at the heart of these improvements. Our study team at Hongda Phytochemistry has come up with a new way to change regular CoQ10 into an ingredient that is easily absorbed by cells and can be used in tough industrial settings.
Liposome Encapsulation Technology Explained
When lipid droplets are used for movement, they are very small and look like the membranes of human cells. It works by putting two layers of phospholipids together so that the hydrophilic ends face outward and the hydrophobic ends face inward. In this way, a safe shell forms around the active CoQ10 molecule. The particles at Shaanxi Hongda Phytochemistry Co., Ltd are smaller than 120 nanometres thanks to secondary micro-encapsulation technology. This makes it much easier to get cell service than with standard recipes.
This way of packaging keeps CoQ10 from breaking down due to oxygen during shipping and storage, while also making it easier for the body to absorb directly into the bloodstream. The tight particle shape stops the active chemical from being released too early, so it gets to the target tissues whole. Our recipe keeps CoQ10 in both the fatty and watery parts of the liposome, which increases its loading capacity while keeping its structure strong.
Key Technical Specifications for B2B Procurement
There are a few key things to keep in mind when looking at ubiquinol liposomal types to buy in bulk. These will determine the quality of the product. When we tested the most recent batch, it was 73.1% pure, and the CoQ10 amount in our pharmaceutical-grade powder stays between 70 and 75%. We know the powder was handled properly because it is off-white to light yellow. It hasn't changed much in colour.
What makes production work better or worse is the range of particle sizes. A 20-mesh screen can let 90% of the powder through, making it very easy for automatic filler tools to use. It is accurate and reliable, with a bulk density of 600g/L and a cutting density of 918g/L. This means that less trash is made during production runs.
The moisture level stays below 1.2% (within the range of ≤5.0%), which stops microbes from growing and keeps the product stable for longer. Heavy metal testing doesn't find any amounts of lead, arsenic, mercury, or cadmium, which are important compliance factors for supplements that want to go into controlled markets. Microbial counts of 50 cfu/g (required ≤ 10,000 cfu/g) and coliforms at 0.31 MPN/g show that the product is very clean, even cleaner than GMP standards.
Carrier Selection and Its Impact on Performance
Both how well the drug works and how well it sells depend on the phospholipid bottle you pick. In place of regular soy lecithin, Shaanxi Hongda only uses sunflower seed phospholipids that are very pure. There are no more problems with GMOs when you choose this option. There are also fewer allergens for sensitive people, and walls are made more open so cells can pass through them better.
When it comes to phospholipids, sunflower phospholipids have more of the type that makes up cell walls. Because of this, the drug is more soluble and doesn't need to be broken down by enzymes as much before it can be taken. It's easier for brands that want to sell in North America and Europe to follow the rules when they use the non-GMO label because it doesn't contain GMOs.
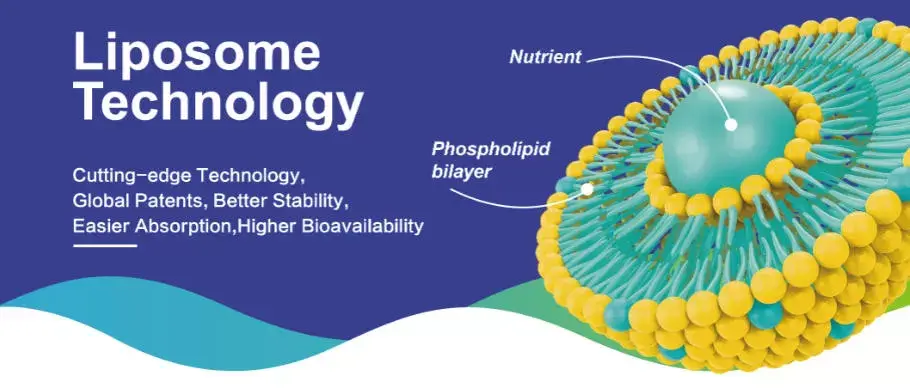
Core Technical Attributes Affecting Performance and Quality
Procurement teams can make better buying choices when they know how technology specs translate into real-world performance, such as for liposomal coenzyme q10 powder. The following factors have a direct effect on the success of the recipe, the ease of production, and the effectiveness of the final product.
Enhanced Bioavailability Through Nanoscale Engineering
The biggest benefit of liposomal delivery over regular CoQ10 powders is the faster absorption rate. When researchers compare regular ubiquinone supplements to liposomal versions, they find that the solubility is better, with peak plasma amounts that are 8 to 12 times higher. Hongda Phytochemistry's production process does this by carefully controlling the liposome width, keeping it below the important sub-120 nm level where absorption by the gut works best.
Several things work together to make this improvement possible. Before being absorbed, standard CoQ10 particles need to be broken down by bile acids and lipase enzymes, which are very different processes for different people with different gut health. Liposomal structures skip these steps and fuse directly with cells that line the inside of the gut by integrating membranes. This lets the active chemical get into the lymphatic system and then the bloodstream throughout the body without being broken down first by the liver. This means that a bigger part of the dose that was given is kept.
Water Dispersibility and Formulation Versatility
Traditional CoQ10 products are very slippery, which makes them hard to mix with other ingredients. Through designed surface chemistry, our liposomal powder gets around this problem. When the powder comes in touch with water, it quickly turns into a steady, milky-white colloid that doesn't separate into oil or stick to the walls of the container. This feature makes it possible to use forms in ways that weren't possible before.
Manufacturers of drinks can add this element to antioxidant drinks that are already made without using harsh chemicals or homogenising the mixture for a long time. The colloid doesn't change when the pH level of the drink changes, even when the drink is made with acidic fruit or neutral water. For oral beauty serums and liquid vitamin shots, this dispersibility makes it easy to describe the products as "oil-free" while still providing lipophilic nutrients well.
Thermal Stability and Mechanical Tolerance
In order to meet processing standards, foods are often put through hard conditions that can break down delicate compounds. When made into tablets, our liposomal coenzyme Q10 powder can survive pressures of up to 50,000 psi without the liposomes breaking or the active ingredient leaking out. The high mechanical tolerance comes from the well-balanced phospholipid makeup and controlled capsule ratios.
Heat stability testing shows that the drug's effectiveness is still there after being exposed to temperatures that are common during normal packaging processes. The equipment used to fill capsules usually works at temperatures between 40°C and 60°C, which is hot enough to break down exposed CoQ10 but not our liposomal structure. This makes the product more stable, which cuts down on production errors and rejections and makes sure that the amount is delivered consistently across all batches, all of which are important for brands that have to follow GMP guidelines.
Shelf Life and Storage Considerations
The security of raw materials has a direct effect on managing stockpiles and figuring out when to make new products. Our liposomal powder is pharmaceutical-grade and stays effective for 24 months when kept in sealed packages at room temperature (15–25°C) and out of direct sunlight. This longer shelf life is very different from reduced forms of CoQ10 like ubiquinol, which need to be kept cool and start to break down after 6 to 12 months of storage in the same circumstances.
The phospholipid layer keeps the encapsulated CoQ10 safe from oxygen in the air, which is the main way that quinone molecules break down. Studies that sped up the ageing process at high temperatures (40°C, 75% relative humidity) show that the drug loses less than 5% of its strength over six months. This means that it should be stable for much longer than the 24-month limit that is stated for normal storage conditions. This dependability lets you buy in bulk, which lowers the cost of buying things by taking advantage of high prices without worrying about running out of stock.
Quality Assurance and Supplier Credentials in the Liposomal CoQ10 Market
To build trusting supply chains, you need to look closely at the manufacturer's skills beyond just checking off the basic product requirements. To lower supply chain risks, procurement workers need to look at quality systems, testing procedures, and how open an organisation is.
Essential Certifications and Regulatory Compliance
Being able to trust high protection starts with making sure the factory is accredited. A building of 20,000 square metres is home to Shaanxi Hongda Phytochemistry Co., Ltd. It has several 100,000-level cleaning workshops used to make plant and medicine ingredients. cGMP, ISO 9001 for quality management systems, ISO 22000 for food safety management, and FSSC 22000, which is a mix of ISO standards and extra technical requirements, are all standards that we are qualified to follow.
They show that processes are recorded, equipment is checked, staff is trained, and there are regular checks by a third party, including for liposomal coenzyme Q10 powder. These badges are more than just things to hang on the wall. 21 CFR Part 111 says that supplies for food items that will be sold in the United States must be in line with cGMP, including liposomal coenzyme Q10 powder. The information that customers in Europe need is different, and most of the time, they want FSSC 22000 approval, which meets the Global Food Safety Initiative guidelines, also for liposomal coenzyme Q10 powder. Having one company handle licenses for more than one area makes it easy to get approvals and do checks.
Religious and health standards make the market bigger. With our Halal and Kosher approvals, brands can serve Muslim and Jewish customers, respectively, which means they can reach more customers without having to buy from more than one source. Vegan approval proves that the product or its processing tools don't contain any ingredients that come from animals. This is becoming a more important factor as the number of plant-based consumers grows across all age groups.

Laboratory Testing and Analytical Verification
Advanced mathematical skills are needed to make sure that quality paperwork is reliable. High-performance liquid chromatography (HPLC), gas chromatography (GC), UV-visible spectrophotometry, atomic absorption spectrophotometry, and other specialised tools are used in our SGS-certified lab to characterise things physically. More than 20 experts with the rank of professor work on developing methods, making sure they work, and regularly testing all of our products.
Every production batch is carefully looked over before it is made available. In the testing process, the amount of active ingredient is measured and identified by comparing it to a reference standard. Heavy metals like lead, arsenic, mercury, and cadmium are checked for, as well as the physical properties (such as appearance, particle size, and density). Pesticide residues that are appropriate for botanical sources are also tested for, and pathogen testing is used to count microbes. There are full Certificates of Analysis that come with each package that show what was found. This lets your quality control team look over the specs before sending the material to be made.
In addition to our own tests, we keep in touch with approved third-party labs like SGS and Eurofins for additional confirmation. This external evaluation backs up claims of quality and gives fair information that is useful when submitting to regulators. When your product needs specific testing that goes beyond standard parameters, like stability studies under fast conditions or compatibility testing with other ingredients, our analysis team works together to come up with the right procedures and gather the data that is needed.

Practical Applications and Customisation in B2B Procurement
Technical specifications achieve value only when aligned with practical application requirements. The versatility of pharmaceutical-grade liposomal powder enables diverse applications across industries, each with unique formulation considerations.
Dietary Supplement Applications
The nutraceutical sector represents the largest market for CoQ10 ingredients, driven by ageing populations seeking cardiovascular support and cellular energy enhancement. Liposomal delivery addresses the primary limitation of conventional CoQ10 supplements—poor absorption leading to inconsistent clinical outcomes. Brands can reduce dose requirements while maintaining efficacy, creating differentiated products with compelling value propositions.
Formulation approaches range from simple single-ingredient capsules emphasising maximum bioavailability to complex multi-nutrient formulas targeting specific health outcomes. Our powder integrates seamlessly with complementary ingredients like vitamin D3, omega-3 fatty acids, or resveratrol without compatibility issues common with oil-based CoQ10 suspensions. The dry powder format prevents oxidation reactions that occur when mixing CoQ10 with certain minerals or pH-sensitive vitamins in liquid or semi-solid matrices.
Dosage flexibility allows brands to target different consumer segments. Maintenance formulations might deliver 100-150 mg liposomal CoQ10 daily, while therapeutic protocols for individuals with increased needs could provide 300-600 mg. The 1kg minimum order quantity from Hongda Phytochemistry enables research and development teams to conduct formulation trials and stability studies before committing to production-scale quantities, reducing development risks and accelerating time-to-market.
Cosmetic and Dermatological Formulations
Topical applications of CoQ10, including liposomal coenzyme Q10 powder, have gained prominence in anti-ageing skincare, leveraging its antioxidant properties to combat oxidative stress in dermal tissues. Liposomal encapsulation proves particularly valuable in cosmetics, where the phospholipid structure enhances skin penetration and provides moisturising benefits beyond the active ingredient itself.
Unlike cosmetic-grade CoQ10 suspensions that require extensive emulsification and stabilisation systems, our powder disperses readily into both water-based serums and emulsion systems. The small particle size ensures smooth texture without grittiness, while the non-GMO sunflower phospholipid carrier appeals to clean beauty brands emphasising botanical ingredients. Formulators can incorporate the powder at 2-5% concentrations to deliver clinically relevant CoQ10 levels while maintaining elegant product aesthetics.
Stability considerations differ between ingestible supplements and topical cosmetics. Skincare products often face temperature fluctuations during shipping and storage, particularly in warm climates. Our liposomal powder's thermal resilience prevents degradation that would otherwise manifest as colour changes or rancid odours, extending product shelf life and maintaining brand reputation. Packaging customisation options allow brands to specify moisture barrier properties, light protection levels, and portion sizes appropriate to their distribution channels.
Conclusion
The technical attributes of liposomal coenzyme Q10 powder represent a significant advancement over conventional delivery systems, offering pharmaceutical-grade purity, nanoscale particle engineering, and superior bioavailability validated through both analytical testing and practical application performance. Key differentiators include sub-120nm liposome diameter enabling 12-fold absorption enhancement, sunflower phospholipid carriers eliminating GMO concerns, exceptional water dispersibility enabling diverse formulation approaches, and mechanical stability withstanding industrial processing conditions. For procurement professionals in nutraceutical, cosmetic, and functional food sectors, these technical specifications translate into reduced dose requirements, simplified formulation development, extended shelf stability, and enhanced end-product efficacy—critical factors supporting competitive positioning in quality-conscious markets.
FAQ1. What makes liposomal Is CoQ10 powder more effective than standard CoQ10?
The phospholipid bilayer encapsulation protects CoQ10 from degradation while facilitating direct cellular membrane fusion, bypassing digestive processes that limit conventional supplement absorption. Clinical comparisons show 8-12 times higher peak plasma concentrations with liposomal delivery, meaning lower doses achieve equivalent biological effects. This efficiency reduces manufacturing costs per effective dose while improving consumer outcomes.
2. How should liposomal CoQ10 powder be stored to maintain potency?
Store sealed containers at room temperature (15-25°C) away from direct sunlight and high-humidity environments. Under these conditions, pharmaceutical-grade material maintains potency for 24 months from the manufacture date. Once opened, minimise air exposure by resealing containers promptly and using desiccant packets if ambient humidity exceeds 60%. Refrigeration provides no additional benefit and may introduce moisture condensation issues.
3. Can liposomal powder be used in both capsules and tablets?
Yes, the powder's flow characteristics and compressibility suit both encapsulation and direct compression tabletting. The liposomal structure withstands pressures exceeding 50,000 psi without rupture, maintaining bioavailability advantages in compressed formats. Encapsulation requires no special handling beyond standard powder-filling procedures, while tabletting may benefit from standard excipients like microcrystalline cellulose to optimise hardness and disintegration properties.
Partner With HONGDA for Premium Liposomal Coenzyme Q10 Powder Supply
Shaanxi Hongda Phytochemistry Co., Ltd. stands ready to support your formulation and manufacturing requirements with pharmaceutical-grade liposomal coenzyme Q10 powder backed by comprehensive quality documentation and technical expertise. Our established position as a trusted liposomal coenzyme Q10 powder manufacturer stems from two decades of botanical extraction innovation, supported by cGMP-certified facilities, SGS-standardised laboratories, and a professor-level R&D team dedicated to continuous improvement. Whether you need research quantities starting at our 1kg minimum order or production-scale volumes with customised specifications, our team provides responsive communication, competitive pricing, and reliable logistics, ensuring your supply chain operates smoothly. Contact us at duke@hongdaherb.com to discuss your specific requirements, request detailed technical specifications, or arrange facility audits demonstrating our commitment to quality excellence and partnership success.
References
1. Bhagavan, H.N., and Chopra, R.K. (2006). "Coenzyme Q10: Absorption, Tissue Uptake, Metabolism and Pharmacokinetics." Free Radical Research, 40(5), 445-453.
2. Crane, F.L. (2001). "Biochemical Functions of Coenzyme Q10." Journal of the American College of Nutrition, 20(6), 591-598.
3. Hodgson, J.M., Watts, G.F., Playford, D.A., Burke, V., and Croft, K.D. (2002). "Coenzyme Q10 Improves Blood Pressure and Glycaemic Control: A Controlled Trial in Subjects with Type 2 Diabetes." European Journal of Clinical Nutrition, 56(11), 1137-1142.
4. Mortensen, S.A., Rosenfeldt, F., Kumar, A., Dolliner, P., Filipiak, K.J., Pella, D., Alehagen, U., Steurer, G., and Littarru, G.P. (2014). "The Effect of Coenzyme Q10 on Morbidity and Mortality in Chronic Heart Failure: Results From Q-SYMBIO: A Randomized Double-Blind Trial." JACC: Heart Failure, 2(6), 641-649.
5. Shults, C.W., Haas, R.H., and Beal, M.F. (1999). "A Possible Role of Coenzyme Q10 in the Etiology and Treatment of Parkinson's Disease." BioFactors, 9(2-4), 267-272.
6. Zhang, Y., Liu, J., Chen, X.Q., and Oliver Chen, C.Y. (2018). "Ubiquinol is Superior to Ubiquinone to Enhance Coenzyme Q10 Status in Older Men." Food & Function, 9(11), 5653-5659.


_1745918362135.webp)






