What is diosmin used for?
Are you struggling with swollen legs, painful hemorrhoids, or chronic venous problems that affect your daily quality of life? Diosmin Powder from Hongda Phytochemistry and Shaanxi Hongda Phytochemistry Co., Ltd. offers a scientifically-backed natural solution derived from citrus fruits that addresses vascular health challenges. This comprehensive guide explores the therapeutic applications, clinical evidence, and quality standards that make Diosmin Powder an essential ingredient for pharmaceutical manufacturers and health supplement producers worldwide. Diosmin is a naturally occurring flavonoid compound primarily extracted from citrus fruits that has gained significant recognition in modern medicine for its remarkable ability to support vascular health and treat various circulatory conditions. As a biologically active plant compound, Diosmin Powder functions as a powerful venotonic agent that strengthens blood vessel walls, reduces inflammation, and improves overall circulation throughout the body. The pharmaceutical industry has embraced this natural therapeutic agent for decades, particularly in European and Asian markets where it has been extensively studied and clinically validated. Manufacturing high-purity Diosmin Powder requires sophisticated extraction techniques and rigorous quality control processes to ensure optimal bioavailability and therapeutic efficacy. Shaanxi Hongda Phytochemistry Co., Ltd. produces pharmaceutical-grade Diosmin Powder at 90% purity using HPLC standardization, meeting USP and EP pharmacopeial standards for use in clinical formulations. This citrus-derived compound has become increasingly important as populations age and vascular health challenges become more prevalent globally.

Understanding Diosmin Powder: Chemical Properties and Biological Mechanisms
Diosmin Powder represents a sophisticated bioflavonoid compound with the chemical designation 3',5,7-trihydroxy-4'-methoxyflavone 7-rutinoside, derived primarily from Citrus aurantium L. as its botanical source. The compound appears as a grayish yellow to light yellow powder with specific physicochemical characteristics that determine its pharmaceutical applications and formulation requirements. With a CAS number of 520-27-4, Diosmin Powder has been thoroughly characterized in scientific literature and regulatory databases worldwide. The molecular structure of this flavonoid glycoside features multiple hydroxyl groups that contribute to its antioxidant properties and ability to interact with biological membranes and vascular tissues. When manufactured to pharmaceutical specifications, Diosmin Powder exhibits excellent stability profiles and can be formulated into various dosage forms including tablets, capsules, and topical preparations. The standard specification for pharmaceutical-grade Diosmin Powder is 90% purity as determined by high-performance liquid chromatography, ensuring consistent therapeutic effects across different batches and production runs. Hongda Phytochemistry's production facilities utilize advanced extraction and purification technologies to achieve 99% purity levels that exceed standard pharmaceutical requirements, making the material suitable for the most demanding clinical applications in major drug formulations.
Pharmacological Actions of Diosmin Powder
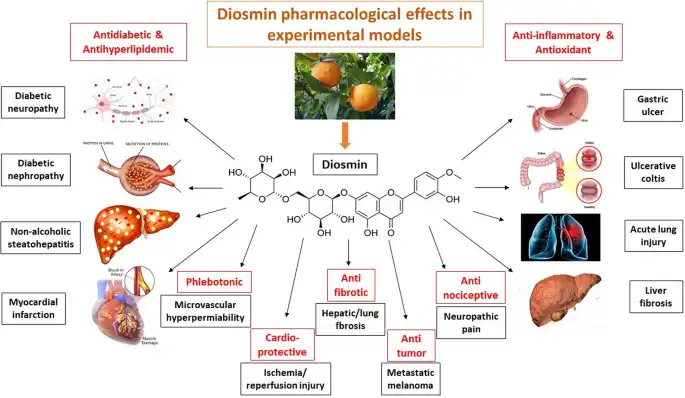
The therapeutic effects of Diosmin Powder stem from multiple complementary pharmacological mechanisms that work synergistically to improve vascular function and reduce inflammatory processes. As a venotonic agent, Diosmin Powder directly enhances venous tone by increasing the contractility of smooth muscle cells in vessel walls, thereby improving venous return and reducing venous stasis that leads to swelling and discomfort. The compound demonstrates significant anti-inflammatory properties by inhibiting the production and release of inflammatory mediators including prostaglandins, leukotrienes, and various cytokines that contribute to vascular inflammation and tissue damage. Research has shown that Diosmin Powder reduces capillary permeability and fragility by strengthening the endothelial barrier and reducing gaps between endothelial cells that allow fluid leakage into surrounding tissues. The antioxidant capabilities of this flavonoid compound provide additional protective benefits by neutralizing reactive oxygen species that damage vascular tissues and accelerate degenerative changes in blood vessels. Clinical pharmacology studies indicate that Diosmin Powder improves lymphatic drainage by stimulating lymphatic contractility and increasing the frequency of lymphatic pump function, which helps remove excess fluid and metabolic waste products from affected tissues. These combined mechanisms make Diosmin Powder particularly effective for treating conditions characterized by venous insufficiency, microcirculatory disorders, and inflammatory vascular pathologies. Pharmaceutical formulations typically combine Diosmin Powder with hesperidin in specific ratios to enhance bioavailability and therapeutic efficacy, creating micronized purified flavonoid fractions that demonstrate superior clinical outcomes compared to non-micronized forms.
Primary Clinical Applications of Diosmin Powder in Vascular Medicine
The most established and well-documented use of Diosmin Powder is in treating chronic venous insufficiency, a progressive condition affecting millions of people worldwide where weakened vein valves fail to adequately return blood from the lower extremities back to the heart. Clinical trials have consistently demonstrated that Diosmin Powder supplementation significantly reduces the hallmark symptoms of chronic venous insufficiency including leg pain, heaviness, swelling, cramping, and the sensation of restless or tired legs that interfere with daily activities and sleep quality. Patients taking pharmaceutical-grade Diosmin Powder typically report improvements within two to four weeks of initiating therapy, with continued benefits observed throughout long-term treatment protocols. The compound works by strengthening venous walls, improving valve function, and reducing the inflammatory cascade that contributes to disease progression and symptom severity. Dosing protocols for chronic venous insufficiency typically involve 900-1000mg of Diosmin Powder daily, often divided into morning and evening doses to maintain consistent therapeutic blood levels throughout the day. Shaanxi Hongda Phytochemistry Co., Ltd. produces Diosmin Powder specifically formulated for oral dosage forms that dissolve appropriately and demonstrate proper bioavailability profiles for systemic absorption. The compound has shown particular efficacy in reducing venous leg ulcers and preventing recurrence in patients with advanced venous disease, making it an important component of comprehensive wound care programs for vascular patients.
Diosmin Powder for Hemorrhoid Treatment
Hemorrhoidal disease represents another major clinical application where Diosmin Powder has demonstrated remarkable therapeutic value through numerous well-designed clinical studies and decades of clinical practice experience. Both internal and external hemorrhoids respond favorably to Diosmin Powder therapy, with patients experiencing reduced pain, bleeding, inflammation, and prolapse severity during acute hemorrhoidal episodes. The flavonoid works by strengthening the vascular structures in the anal region, reducing venous congestion that causes hemorrhoidal tissues to swell and become symptomatic, and decreasing inflammatory mediators that contribute to pain and discomfort. Clinical evidence indicates that Diosmin Powder is particularly effective when started at the onset of acute hemorrhoidal symptoms, potentially reducing the need for more invasive interventions such as rubber band ligation, sclerotherapy, or surgical hemorrhoidectomy. Treatment protocols typically involve higher initial doses during acute flare-ups followed by maintenance dosing to prevent recurrence, with many clinicians recommending periodic courses of therapy for patients with recurrent hemorrhoidal problems. The safety profile of Diosmin Powder makes it suitable for extended use in chronic hemorrhoid sufferers who require ongoing management of their condition. Topical formulations containing Diosmin Powder combined with local anesthetics and anti-inflammatory agents provide immediate symptomatic relief while the oral systemic therapy addresses the underlying vascular pathology. Hongda Phytochemistry supplies pharmaceutical manufacturers with Diosmin Powder suitable for both oral and topical hemorrhoid formulations, maintaining consistent quality standards across different application routes and delivery systems.
Lymphedema Management with Diosmin Powder
Lymphedema, characterized by chronic swelling due to impaired lymphatic drainage, represents a challenging clinical condition where Diosmin Powder offers significant therapeutic benefits as part of comprehensive decongestive therapy protocols. The compound enhances lymphatic contractility and pumping function, increasing the rate at which lymphatic vessels remove excess fluid and protein molecules from interstitial spaces back into the central circulation. Patients with lymphedema secondary to cancer treatment, chronic venous insufficiency, or primary lymphatic dysfunction have shown measurable improvements in limb circumference, tissue texture, and subjective symptoms when treated with Diosmin Powder supplementation. Clinical studies have documented reductions in both the frequency and severity of cellulitis infections that commonly complicate lymphedema, potentially due to improved tissue perfusion and enhanced immune cell trafficking in affected regions. The anti-inflammatory properties of Diosmin Powder help reduce the chronic low-grade inflammation that contributes to fibrotic tissue changes and progressive lymphedema worsening over time. Treatment approaches typically combine Diosmin Powder with compression therapy, manual lymphatic drainage, and exercise programs to achieve optimal therapeutic outcomes. Long-term supplementation appears safe and well-tolerated, making Diosmin Powder suitable for the chronic management required in lymphedema patients. Pharmaceutical formulations designed for lymphedema treatment often specify particular particle sizes and dissolution profiles to ensure adequate absorption and systemic distribution. Shaanxi Hongda Phytochemistry Co., Ltd. works closely with pharmaceutical developers to customize Diosmin Powder specifications that meet the unique requirements of lymphedema treatment products.
Advanced Therapeutic Uses and Emerging Applications
Beyond the primary established indications, Diosmin Powder has demonstrated promising results in various other clinical scenarios where vascular health, inflammation control, and antioxidant protection play important roles. Diabetic patients frequently experience microcirculatory complications including retinopathy, neuropathy, and impaired wound healing that may benefit from the vascular protective effects of Diosmin Powder supplementation. Preliminary research suggests that Diosmin Powder may help preserve capillary integrity and reduce oxidative stress in diabetic microvasculature, potentially slowing the progression of microvascular complications. Postoperative recovery represents another emerging application where Diosmin Powder may accelerate healing and reduce complications by improving tissue perfusion and reducing inflammation at surgical sites. Athletes and individuals engaged in intensive physical training have shown interest in Diosmin Powder for managing exercise-induced muscle damage and accelerating recovery between training sessions. The compound's anti-inflammatory and circulation-enhancing properties may help reduce muscle soreness, promote metabolic waste removal, and enhance nutrient delivery to recovering tissues. Some dermatological applications have explored Diosmin Powder for treating rosacea and other conditions involving facial vascular dysfunction and chronic inflammation. Research into cardiovascular protective effects continues to investigate whether Diosmin Powder might offer benefits for endothelial function and arterial health beyond its established venous applications.
Diosmin Powder Quality Standards and Manufacturing Excellence
Pharmaceutical-grade Diosmin Powder must meet stringent quality specifications that ensure therapeutic efficacy, safety, and batch-to-batch consistency for clinical applications and regulatory compliance. High-performance liquid chromatography serves as the gold standard analytical method for quantifying Diosmin Powder purity, with pharmaceutical specifications typically requiring minimum 90% purity for standard formulations and up to 99% purity for specialized applications. Comprehensive testing protocols examine physical characteristics including appearance, particle size distribution, bulk density, and solubility profiles that affect formulation performance and bioavailability. Chemical analysis evaluates moisture content, ash content, residual solvents, and potential degradation products that could compromise product stability or introduce impurities into final dosage forms. Heavy metal testing ensures that lead, arsenic, cadmium, and mercury levels remain below strict regulatory limits, typically less than 10 ppm total heavy metals for pharmaceutical applications. Microbiological testing confirms the absence of pathogenic organisms and ensures that total bacterial counts, yeast, and mold remain within acceptable specifications for oral pharmaceutical products. Pesticide residue analysis verifies that agricultural chemicals used during citrus cultivation do not persist in final Diosmin Powder products at levels that could pose health risks or violate regulatory standards. Shaanxi Hongda Phytochemistry Co., Ltd. maintains comprehensive quality management systems certified to cGMP, ISO 9001, ISO 22000, and FSSC22000 standards, ensuring that every batch of Diosmin Powder undergoes rigorous testing before release to customers. The company's SGS-standardized laboratory equipped with HPLC, GC-MS, and NMR instrumentation provides analytical capabilities that exceed typical industry standards.
Regulatory Compliance and International Certifications
The global pharmaceutical market demands extensive regulatory compliance and certification documentation to support product registration, importation, and commercial distribution across different jurisdictions with varying requirements. Diosmin Powder manufactured by Hongda Phytochemistry complies with United States Pharmacopeia (USP) and European Pharmacopoeia (EP) monographs, ensuring that material specifications align with recognized international standards used by regulatory agencies worldwide. The facility holds multiple organic certifications including ORGANIC (EU) and ORGANIC (NOP) designations that verify sustainable sourcing practices and prohibit synthetic pesticide use in raw material cultivation. Religious dietary certifications including HALAL and KOSHER enable Diosmin Powder use in products targeting specific consumer populations with dietary restrictions based on religious principles. Food safety management system certifications such as FSSC22000 and BRC demonstrate comprehensive controls over ingredient sourcing, manufacturing processes, storage conditions, and distribution practices that ensure product safety throughout the supply chain. The company's cGMP certification specifically addresses pharmaceutical manufacturing requirements including facility design, equipment qualification, process validation, personnel training, and documentation practices mandated by global regulatory authorities. Each shipment of Diosmin Powder includes complete documentation packages containing Certificates of Analysis (COA), Technical Data Sheets (TDS), Material Safety Data Sheets (MSDS), and stability study data that support regulatory submissions and facilitate customs clearance in destination countries. Third-party testing partnerships with SGS and Eurofins provide independent verification of quality claims and analytical results, adding additional credibility to product specifications. This comprehensive certification portfolio positions Hongda Phytochemistry as a trusted supplier for pharmaceutical companies operating in heavily regulated markets including the United States, European Union, and other regions with strict import requirements.
Manufacturing Infrastructure and Production Capabilities
Shaanxi Hongda Phytochemistry Co., Ltd. operates state-of-the-art manufacturing facilities specifically designed for natural plant extract production, with infrastructure investments exceeding industry standards for quality and capacity. The production complex covers approximately 20,000 square meters and houses more than 10 specialized production lines equipped with modern extraction, purification, drying, and packaging equipment. Class 100,000 purification workshops provide controlled environments where particulate contamination is minimized through advanced air filtration and pressure differential systems that prevent cross-contamination between different product lines. The facility's annual production capacity exceeds 3,000 tons of finished plant extracts, providing supply security for large pharmaceutical and nutraceutical customers with high-volume requirements. More than 150 trained workshop operation technicians execute standardized manufacturing procedures under strict quality supervision, ensuring consistent product quality across all production batches. Raw material sourcing begins at the company's three dedicated planting bases including specialized citrus cultivation areas where professional agronomists supervise growing conditions, harvest timing, and initial processing steps that preserve bioactive compound integrity. Vertical integration from raw material cultivation through final product packaging provides unprecedented control over quality parameters and traceability throughout the entire supply chain. Advanced extraction technologies including supercritical fluid extraction, membrane filtration, and column chromatography enable efficient isolation and purification of target compounds while minimizing environmental impact and solvent residues. The production facility maintains separate warehousing zones totaling 3,000 square meters with climate-controlled storage areas designated for different product categories including fruit and vegetable powders, natural plant extracts, vitamins, amino acids, and sample storage. This comprehensive infrastructure enables Hongda Phytochemistry to deliver Diosmin Powder with 3-7 working day lead times while maintaining flexible packaging options ranging from 1kg bags to 25kg drums suitable for different customer requirements.
Research and Development Capabilities
Innovation drives continued improvement in Diosmin Powder quality, application development, and formulation optimization through dedicated research and development programs supported by university partnerships and internal expertise. The company operates two specialized R&D institutions staffed by more than 20 experienced researchers recruited from universities and scientific research institutions across China, bringing diverse technical perspectives to product development challenges. Systematic research programs focus on optimizing extraction efficiency, enhancing bioavailability through particle size reduction and surface modification, developing novel delivery systems, and identifying synergistic combinations with complementary natural compounds. Collaborative relationships with university professors provide access to cutting-edge analytical techniques, clinical research networks, and fundamental scientific insights that inform practical product improvements. The R&D laboratories maintain extensive libraries of authenticated plant materials, chemical standards, and analytical methods that support new product development and quality troubleshooting initiatives. Ongoing stability studies evaluate how storage conditions, packaging materials, and formulation excipients affect Diosmin Powder shelf life and potency retention over time. Application research explores optimal combinations of Diosmin Powder with other vascular-protective compounds including hesperidin, rutin, and horse chestnut extracts to create synergistic formulations with enhanced clinical benefits. Formulation development services help customers translate raw Diosmin Powder into finished products by providing technical guidance on dissolution enhancement, tablet compression, capsule filling, and topical cream formulation. This comprehensive R&D infrastructure positions Hongda Phytochemistry as not merely a raw material supplier but as a technical partner capable of supporting customers throughout the product development lifecycle from concept through commercial launch.
Dosage Forms and Formulation Considerations
Diosmin Powder's versatility enables incorporation into multiple pharmaceutical dosage forms tailored to specific therapeutic applications, patient preferences, and market requirements. Oral tablets represent the most common formulation approach, typically containing 450-600mg of Diosmin Powder per tablet designed for twice-daily administration to achieve target therapeutic doses. Micronization technology reduces particle size to enhance dissolution rates and bioavailability, particularly important for poorly water-soluble compounds like Diosmin Powder where absorption may otherwise limit therapeutic efficacy. Sustained-release formulations using specialized polymeric matrices or coating systems provide extended drug release that maintains therapeutic blood levels throughout the day with once-daily dosing convenience. Hard gelatin capsules offer simpler manufacturing processes and flexibility for dose adjustments, with typical formulations containing 250-500mg of Diosmin Powder combined with appropriate flow agents and fillers. Soft gelatin capsules can incorporate Diosmin Powder in oil-based suspensions that potentially enhance absorption through the lymphatic system and bypass first-pass hepatic metabolism. Effervescent tablets or granules that dissolve in water provide alternative delivery formats for patients who have difficulty swallowing solid dosage forms, though careful formulation is required to maintain stability in the presence of acidic and alkaline effervescent components. Topical preparations including creams, gels, and ointments deliver Diosmin Powder directly to affected areas for hemorrhoid treatment and localized venous conditions, though transdermal penetration limitations may reduce systemic efficacy compared to oral administration. Powder blends and premix formulations enable convenient incorporation into functional foods, beverage products, and dietary supplements targeting wellness-oriented consumers rather than pharmaceutical patient populations. Hongda Phytochemistry provides technical support for all these formulation approaches, offering customized Diosmin Powder specifications including specific particle size distributions, coating treatments, and blend formulations that optimize performance in different dosage forms.
Global Market Distribution and Customer Support
International trade capabilities and customer service infrastructure enable Hongda Phytochemistry to serve pharmaceutical and nutraceutical manufacturers throughout Europe, North America, Asia, and other global markets with diverse regulatory requirements. Strategic inventory positioning in European warehouses provides local stock availability that reduces delivery times and shipping costs for customers in EU member states while simplifying customs procedures and regulatory compliance. Flexible packaging options accommodate different order volumes from small-scale trials requiring 1kg sample quantities to commercial production orders of multiple tons delivered in 25kg drums configured on standard pallets for efficient logistics handling. The company's established shipping networks ensure reliable delivery within 3-7 working days for most destinations, with tracking systems that provide real-time visibility into shipment status and estimated arrival times. Comprehensive documentation support includes preparation of certificates of origin, phytosanitary certificates, free sale certificates, and other export documentation required by destination country regulations and customs authorities. Technical support teams provide formulation guidance, stability recommendations, regulatory intelligence, and troubleshooting assistance to help customers successfully develop and commercialize products containing Diosmin Powder. Quality assurance commitments include rapid response to customer inquiries about specifications, testing results, or product performance, backed by robust complaint handling systems and corrective action procedures. Long-term supply agreements offer price stability and guaranteed capacity allocation for customers with predictable demand patterns and multi-year product life cycles. The company's business philosophy emphasizes building enduring partnerships rather than transactional relationships, with account management practices that proactively identify customer needs and deliver customized solutions that support their business objectives.
Conclusion
Diosmin Powder stands as a scientifically validated, clinically proven therapeutic agent for managing venous insufficiency, hemorrhoids, lymphedema, and various microcirculatory disorders affecting millions of patients worldwide. The compound's favorable safety profile, extensive clinical documentation, and multiple pharmacological mechanisms make it an indispensable ingredient for pharmaceutical manufacturers developing vascular health products for global markets. Shaanxi Hongda Phytochemistry Co., Ltd. has established itself as a leading China Diosmin Powder factory, China Diosmin Powder supplier, and China Diosmin Powder manufacturer through two decades of continuous innovation, quality excellence, and customer service commitment. Our China Diosmin Powder wholesale programs offer competitive pricing without compromising the High Quality Diosmin Powder specifications demanded by regulated pharmaceutical applications. For customers seeking Diosmin Powder for sale with comprehensive certification documentation, reliable supply chains, and technical support, our facilities deliver 99% purity material meeting USP/EP pharmacopeial standards. The competitive Diosmin Powder price combined with assured quality, extensive certifications including cGMP and FSSC22000, and responsive customer service creates compelling value for global buyers. Whether you require small development quantities or commercial-scale supply contracts, our team provides customized solutions tailored to your specific formulation requirements and regulatory needs. Contact our technical specialists at duke@hongdaherb.com to discuss your Diosmin Powder requirements and discover how our manufacturing excellence and application expertise can accelerate your product development timeline and support your business growth objectives in the expanding vascular health market.
References
1. Lyseng-Williamson KA, Perry CM. Micronised purified flavonoid fraction: a review of its use in chronic venous insufficiency, venous ulcers and haemorrhoids. Drugs. 2003;63(1):71-100.
2. Ramelet AA. Clinical benefits of Daflon 500 mg in the most severe stages of chronic venous insufficiency. Angiology. 2001;52 Suppl 1:S49-56.
3. Boyle P, Diehm C, Robertson C. Meta-analysis of clinical trials of Cyclo 3 Fort in the treatment of chronic venous insufficiency. International Angiology. 2003;22(3):250-262.
4. Kakkos SK, Nicolaides AN. Efficacy of micronized purified flavonoid fraction (Daflon) on improving individual symptoms, signs and quality of life in patients with chronic venous disease: a systematic review and meta-analysis of randomized double-blind placebo-controlled trials. International Angiology. 2018;37(2):143-154.


_1745918362135.webp)






