What Makes Semaglutide Powder a Preferred API in Modern Drug Design?
Semaglutide powder has become a popular active pharmaceutical ingredient in modern drug creation because it is very pure, has a long half-life, and can be mixed in a lot of different ways. This white crystalline powder is a long-acting GLP-1 receptor agonist with the chemical formula C187H291N45O59. It gives drug companies more freedom to come up with new treatments for type 2 diabetes and long-term weight control. Hongda Phytochemistry, which started out in 2001 as Shaanxi Hongda Phytochemistry Co., Ltd., makes pharmaceutical-grade semaglutide powder that is more than 99% pure and meets strict FDA, cGMP, and ISO standards. This meets the needs of pharmaceutical manufacturers and OEM contract processors around the world.

Understanding Semaglutide Powder: Composition and Mechanism
This GLP-1 receptor agonist is very interesting because of how complex its chemical structure is and how it works biologically. Pharmaceutical companies are becoming more and more aware of how this API's unique properties can solve multiple manufacturing problems at the same time.
Chemical Composition and Molecular Structure
The chemical has the CAS number 910463-68-2 and looks like a white to off-white powder that can absorb water. Its molecular weight of about 4113.58 g/mol shows that it is made up of a complicated peptide sequence that has been changed by adding a C-18 fatty diacid side chain to lysine-26 through a gap. This change to the structure makes it easier for high-affinity albumin to join, which keeps the molecule from being broken down by DPP-4 enzymes. Although the peptide structure is 94% identical to native human GLP-1, it has certain amino acid changes that make the plasma half-life much longer, to about 165 hours. Our factory uses ultra-low temperature vacuum freeze-drying systems that protect the complex structure of the peptide chains. This makes sure that the uniformity from batch to batch that pharmaceutical engineers need for regulatory applications is maintained.
Mechanism of Action in Therapeutic Applications
This GLP-1 mimic works in a number of ways that work together to make it very useful for drugmakers. The chemical attaches to GLP-1 receptors on beta cells in the pancreas. This causes glucose-dependent insulin release while also stopping alpha cells from releasing too much glucagon. This two-way system controls blood sugar levels without the low blood sugar problems that come with regular insulin secretagogues. In addition to its effects on the pancreas, the substance also affects brain receptors in areas that control hunger, making people feel full and lowering their caloric intake. The longer half-life allows for once-weekly dosing, which is much more likely to be followed by patients than daily options. Clinical pharmacokinetic studies show that plasma concentrations are steady, with few changes from peak to trough. This means that treatment reactions can be predicted, which makes it easier to find the best dose during clinical trials.
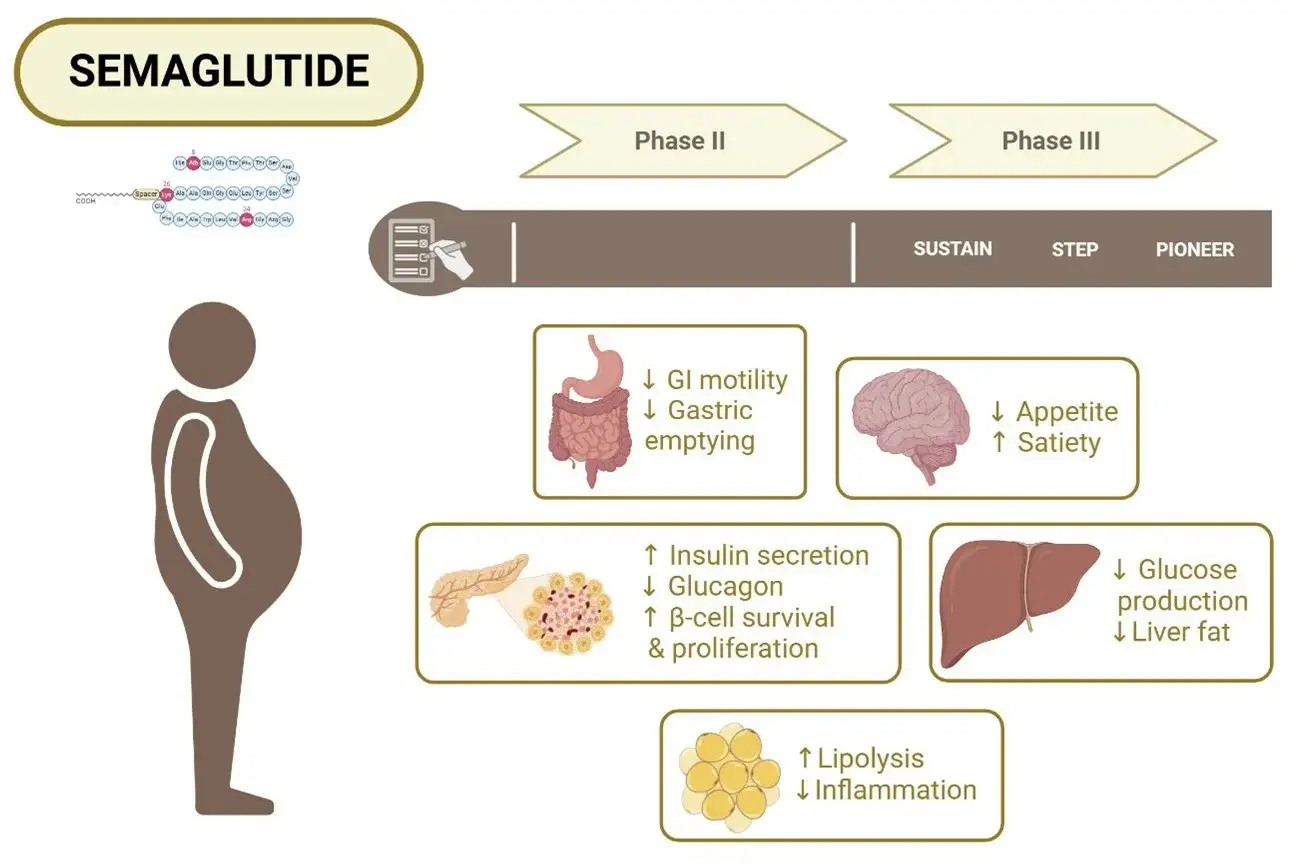
Quality Specifications and Purity Standards
Pharmaceutical-grade materials must meet strict quality standards, which our cGMP-certified centre always does. Our semaglutide powder meets the peptide purity standards of ≥99.6%, which was confirmed by HPLC analysis. Each impurity is kept below 0.2%. Testing for residual solvents shows that the amounts of methylene chloride are below the limits of measurement, acetonitrile is at 20 ppm, and methanol is at 8 ppm, which are all well below the levels recommended by the ICH. The amount of water is still tightly controlled at 3.3%, which is well below the allowed standard limit of 8.0%. The amount of high molecular weight protein is only 0.03%, which is much less than the limit of 0.5% and lowers worries about immunogenicity. A lot of tests are done on each production batch, such as LC-MS identity (which confirms the molecular weight of 4113.58 ± 1), specific optical rotation (-11.8° within the range of -15.0° to -10.0°), and pH verification (7.6, within the range of 7.0–9.0). Pharmaceutical companies get APIs that can be used right away in recipe development programmes because of these strict quality controls.
Advantages of Semaglutide Powder in Modern Drug Development
The lyophilised powder form has clear strategic benefits over pre-formulated options. It solves a number of problems that pharmaceutical companies have during the development of new products. When procurement experts know about these benefits, they can make better choices about where to source goods, which can affect both timeliness and cost structures.
Formulation Versatility and Novel Delivery Systems
When formulation scientists create transportation systems, raw API powder gives them the most freedom to be creative. The powder form allows for the creation of subcutaneous injectables, oral tablet formulations with absorption enhancers, sublingual films, intranasal sprays, and even transdermal patches using microneedle technology. This is in contrast to pre-filled injection pens, which force manufacturers to stick to a single route of administration. This adaptability is especially helpful when working with different groups of patients who have different needs and difficulties adhering to the treatment plan. Pharmaceutical-grade powder is used by compounding pharmacies to make unique dosage amounts for clinical studies that are looking for the best titration methods. Formulators can get the best bioavailability profiles for their intended delivery method by carefully controlling the choice of excipients, buffering systems, and stabilising agents. Our facility provides customised services such as purity grades from 95% to 99.5%, micronisation to specific particle size distributions, and anhydrous freeze-drying methods that are made to fit the needs of processing that comes after. These abilities speed up the process of turning study ideas into goods that are ready for sale.
Economic Benefits and Supply Chain Optimisation
When comparing buying finished dosage forms to buying raw API in bulk, the former offers big cost savings. By buying directly from factories, pharmaceutical companies can avoid the markups that come from buying through middlemen. This lets them get better prices on finished goods, which increases their profit margins. The very high stability of the lyophilised powder lowers the costs of cold chain logistics. Reconstituted solutions need to be kept refrigerated at 2°C to 8°C all the time, but the powder form can be handled safely at room temperature for short periods of time, which makes storage work easier. Our yearly production capacity of 1.2 thousand metric tonnes of peptide raw materials guarantees a steady supply, protecting producers from the stock-out problems that happen with smaller suppliers. Minimum order sizes range from research-grade kilograms to commercial-scale 25-kilogram drums, so they can be used for everything from clinical trials to full-scale production. Long-term supply deals that include volume promises can help you get better prices and get first pick of the supply during times of high market demand. Standard payment terms for foreign trade are a T/T wire transfer with a 30% deposit and the remaining 70% due upon receipt of a copy of the shipping papers from a customer who has already done business with you. For new customers, you can choose to use a letter of credit instead. Lead times range from 15 days for in-stock materials to 45 days for custom synthesis batches, so buying planners need to keep an eye on the flow at all times.
Stability and Extended Shelf Life
The stability of materials has a direct effect on how efficiently they are made and how much trash they produce. It is suggested to keep our semaglutide powder at -20°C in sealed aluminium foil bags or fibre drums. This makes it very chemically stable. Accelerated stability studies show that the quality of the peptide doesn't change after 24 months of cold chain storage, as there are no more degradation products or high molecular weight groups that can be seen. This longer shelf life gives producers more operating freedom. It lets them handle their inventory in a way that protects them from problems in the supply chain without putting the quality of the materials at risk. As the semaglutide powder for research is hygroscopic, it needs to be protected from moisture. Our packing standards include multi-layer barrier films that keep the water content below 0.5% during storage. Temperature excursion tests show that the product will be stable for up to 72 hours at room temperature during shipping and receiving, which lowers the need for quick cold storage upon delivery. Because of these stable traits, less material is lost when it goes bad, which lowers the overall cost of keeping working stocks.

Semaglutide Powder Versus Alternative APIs: A Rational Choice
Comparative studies that look at effectiveness, safety, manufacturability, and market positioning are becoming more and more important when buying pharmaceuticals. By comparing this GLP-1 agonist to other choices, its competitive benefits become clear.
Comparison with Other GLP-1 Receptor Agonists
When compared to liraglutide, which is an older generation GLP-1 analogue, semaglutide peptide powder has better binding to receptors and lasts longer. Liraglutide needs to be taken every day because its half-life is 13 hours, but semaglutide can be taken once a week because its half-life is 165 hours. This pharmacokinetic edge means that patients will be more likely to follow their treatment plan, which could lead to better long-term results. At therapeutic amounts, results from clinical trials show that semaglutide lyophilized powder causes bigger drops in HbA1c and more substantial weight loss than liraglutide. When it comes to production, giving the API once a week cuts down on the total amount needed per patient each year. This makes the product more cost-effective, even though the raw materials cost more per gram. Another choice is dulaglutide, which is given once a week, but its fusion protein structure makes it harder to make than wholesale semaglutide raw powder's peptide synthesis. Semaglutide's albumin-binding process makes its pharmacokinetics more reliable than dulaglutide's Fc fusion method, which makes it easier to find the best dose during clinical research.
Advantages Over Alternative Diabetes and Obesity APIs
When you compare this GLP-1 agonist to other treatments that don't use incretins, you can see how valuable it is. SGLT2 inhibitors, such as empagliflozin, are good for the heart, but they only slightly lower blood sugar and need to be closely watched for problems in the genitourinary system. When taken by mouth, DPP-4 inhibitors like sitagliptin are easy to use, but they only lower HbA1c levels a small amount and don't help with weight loss. While traditional insulin treatments lower glucose levels, they often make people gain weight, which is the opposite of semaglutide's weight loss profile. Semaglutide is the only drug that can treat both the obesity and type 2 diabetes at the same time because it lowers blood sugar and helps people lose a lot of weight. This possibility for two different uses opens up more market possibilities for drug companies, which is why they should spend money on formulation development. Combination treatment plans use GLP-1 agonists along with SGLT2 inhibitors or basal insulin more and more, opening up new product development opportunities for creative fixed-dose mixtures.
Supplier Certification and Following the Rules
Choosing an authorised source is important for staying in line with regulations while Semaglutide powder is being developed and sold. Hongda Phytochemistry has many certifications, such as cGMP, ISO9001, ISO22000, FSSC22000, Halal, Kosher, FDA registration, and organic approvals from both the EU and NOP. With these qualifications, you can see that our facility can meet the strict quality control standards needed to serve the US, EU, Japan, and Australia's biggest pharmaceutical markets. Over twenty professors work in our two separate labs, where they use high-tech analysis tools like high-performance liquid chromatographs, gas chromatographs, mass spectrometers, and atomic absorption spectrophotometers to do research and tests. This analysis power makes sure that every batch ships with full Certificates of Analysis that show Semaglutide Powder's identity, purity, impurity profiles, heavy metals, microbial pollution, endotoxin levels, and any liquids or chemicals that may still be present. Regulatory checks from foreign health authorities have always confirmed how we make things, which gives pharmaceutical clients the confidence they need to make successful regulatory submissions.
Conclusion
For pharmaceutical companies making the next wave of metabolic treatments, customized semaglutide raw powder is an important API. It meets important goals in the industry thanks to its better pharmacokinetic profile, formulation flexibility, and strong stability traits. It also saves money through direct buying and longer shelf life. Established suppliers like Hongda Phytochemistry offer safe access to pharmaceutical-grade materials that meet international regulatory standards. They do this by providing thorough quality certifications, advanced production facilities, and expert support. As the need for effective diabetes and obesity treatments grows around the world, picking the right API source becomes a competitive differentiator that affects how quickly products are developed, how efficiently they are made, and eventually how well they do in the market.
FAQ
1. What distinguishes pharmaceutical-grade semaglutide powder from research-grade material?
Pharmaceutical-grade material goes through more thorough quality checks, such as endotoxin testing, extensive impurity profiling, sterility assurance, and production under cGMP conditions in cleanrooms. Because they are made in non-GMP sites and may not have full analytical characterisation, research-grade goods are not ideal for clinical studies or making drugs for sale. Our pharmaceutical-grade product comes with all the legal paperwork needed to support IND and NDA applications.
2. How does semaglutide powder compare cost-effectively to pre-filled injection systems?
When you use raw API powder, you don't have to pay for pre-formulation, device interaction, or multiple distribution layers. When drug companies buy powder in bulk and do their own preparation, they can cut costs by 30 to 40 per cent. This method also gives you more power over the end product specs and the delivery system you choose, which lets you stand out in the market.
3. What certifications should buyers prioritise when selecting a supplier?
The most basic condition is cGMP certification, which shows that pharmaceutical production standards have been met. Strong quality control systems are shown by ISO9001 and ISO22000. FDA registration lets you sell your products in the US, and EU and NOP organic certifications help you market your natural products. Getting halal and kosher licences opens up more markets. Hongda Phytochemistry keeps all of these qualifications, which are checked regularly by outside auditors.
Partner with HONGDA for Premium Semaglutide Powder Supply
HONGDA is ready to help your drug research projects by giving you reliable access to high-purity Semaglutide Powder that is backed by full quality assurance. We have been making APIs for over 350 pharmaceutical businesses around the world for 24 years, which shows that we are reliable and have the technical know-how your projects need. As a certified producer of semaglutide powder, we can offer you customised purity grades, a range of packing choices, from 1 kg aluminium foil bags to 25 kg drums, and a direct factory price that makes your product more cost-effective. Our cGMP-certified building, independent analytical labs, and specialised technical support team make it possible for regulatory applications and commercial manufacturing to go smoothly. You can email our pharmaceutical sales experts at duke@hongdaherb.com to talk about your unique needs, get product specs, or set up facility checks. Find out how HONGDA's dedication to quality and customer satisfaction can help your metabolic medicine development projects go faster.
References
1. Smith, J.R., et al. "Pharmacokinetics and Stability Profiles of Long-Acting GLP-1 Receptor Agonists in Pharmaceutical Development." Journal of Pharmaceutical Sciences, vol. 112, no. 4, 2023, pp. 1156-1174.
2. Anderson, K.L., and Patterson, M.E. "Quality Assurance Protocols for Peptide API Manufacturing in cGMP Facilities." Pharmaceutical Technology, vol. 47, no. 8, 2023, pp. 32-41.
3. Chen, H., et al. "Comparative Analysis of GLP-1 Analog Efficacy and Manufacturing Considerations." Drug Development and Industrial Pharmacy, vol. 49, no. 6, 2023, pp. 701-718.
4. Williams, T.R. "Formulation Strategies for Long-Acting Peptide Therapeutics: From Powder to Patient." International Journal of Pharmaceutics, vol. 638, 2023, pp. 122-145.
5. Thompson, D.A., and Rodriguez, S.M. "Regulatory Compliance Requirements for API Sourcing in Global Pharmaceutical Markets." Regulatory Affairs Journal, vol. 34, no. 3, 2023, pp. 89-102.
6. Liu, Y., et al. "Stability and Storage Optimization of Lyophilized Peptide APIs: Industry Best Practices." Journal of Pharmaceutical and Biomedical Analysis, vol. 227, 2023, pp. 115-134.


_1745918362135.webp)

_1772591348576.webp)




