Why Choose Liposomal Coenzyme Q10 Powder for Supplements?
Procurement professionals seeking next-generation CoQ10 ingredients face a critical decision: traditional crystalline powders or advanced liposomal delivery systems? Liposomal coenzyme Q10 powder represents a strategic breakthrough for formulators in nutraceuticals, pharmaceuticals, and functional foods. Unlike conventional CoQ10, which suffers from poor water solubility and bioavailability below 5%, liposomal encapsulation mimics human cell membrane structure, delivering active ubiquinone directly through intestinal barriers into systemic circulation. This means 3-10 times higher plasma concentrations, reduced dosage requirements, and stronger clinical outcomes. Shaanxi Hongda Phytochemistry Co., Ltd. (Hongda Phytochemistry) engineers pharmaceutical-grade liposomal CoQ10 using secondary micro-encapsulation technology and non-GMO sunflower phospholipids, achieving ≥70% purity and stable particle sizes below 120nm for maximum cellular uptake.
Understanding Liposomal Coenzyme Q10 Powder
What Makes Liposomal Technology Different
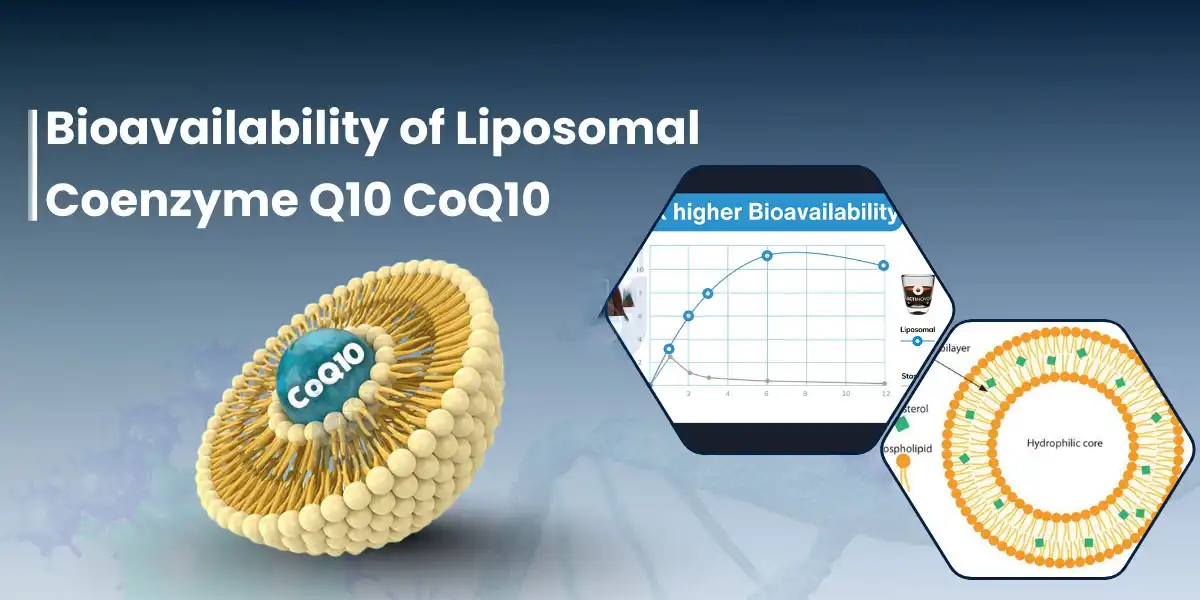
Liposomal release completely changes the way that CoQ10 gets into the body. Standard CoQ10 is lipophilic, which means it forms clumps in the digestive system and needs bile salts to break down slowly and inefficiently. Liposomal CoQ10 powder systems solve this problem by covering CoQ10 molecules in phospholipid bilayers, which are tiny circles made of the same stuff that makes up human cell walls. When these nanoparticles touch the intestinal epithelium, they join directly with epithelial cells. This means that the cargo doesn't have to go through the liver's first-pass metabolism process; instead, it goes straight into the lymphatic routes. Hongda Phytochemistry uses a special secondary micro-encapsulation method that keeps each CoQ10 molecule stable inside a nanoscale phospholipid vesicle (D50 < 120nm). The phospholipid molecules arrange themselves so that the hydrophilic ends face outward and the hydrophobic ends face inward. This makes a closed structure that protects CoQ10 from oxidation and makes membrane transfer easier. This design doesn't just make absorption better; it changes it completely, changing a nutrient that isn't well taken into an active ingredient that is highly bioavailable and good for high-end formulations.
Scientific Mechanism Behind Enhanced Bioavailability
The solubility benefit comes from taking on the structure of membranes and being absorbed by lymphatics. Before it can be absorbed, standard CoQ10 has to be mixed with bile acids, broken down by enzymes, and then put back together into chylomicrons. This is a long process that loses a lot of nutrients. These problems don't bother liposomal coenzyme Q10 powder. The outside of the phospholipid interacts smoothly with the cell membranes of intestinal cells through passive fusion. This lets the entire vesicles go through the tight junctions and enter the enterocytes directly. CoQ10 is released into the cytoplasm and moves to the mitochondria, which are the powerhouses of the cell, where it's needed the most. Clinical data show that liposomal delivery raises plasma CoQ10 levels 3–12 times higher than normal powder amounts of the same amount. This means lower amounts that work, lower formulation costs, liposomal coenzyme Q10, and more stable treatment results across a wide range of groups.
Comparing Liposomal Coenzyme Q10 Powder with Other Forms
Absorption Efficiency Across Delivery Formats
Absorption rates really show how different delivery methods are. Because it has a very high molecular weight (863 Da) and doesn't mix well with water, crystalline CoQ10 powder usually has less than 5% oral absorption. Softgel capsules with oil-solubilized CoQ10 improve absorption by about 10 to 15 percent by dissolving the active ingredient in lipid carriers first. However, the effectiveness is still based on the person's bile output and stomach emptying rate. Liquid liposomal solutions have better uptake. Liposomal coenzyme Q10 powder keeps the absorption benefits of liposomal structures. When mixed with water (in drinks, pills, or tablets), the powder makes nanoscale liposomes on its own, which absorb the same amount of medicine as new liquid formulas. Because of this, producers can put high-absorption CoQ10 into solid tablet forms, ready-to-drink drinks, and beauty serums without affecting their effectiveness or shelf life.
Formulation Versatility and Manufacturing Integration
Product makers like ingredients that work well with the way things are already made. Some problems with traditional CoQ10 include not dissolving well in water, forming crystals, and not working with water-based systems. Liposomal coenzyme Q10 powder solves these problems because it mixes better with water. When Hongda's recipe is mixed with water, it quickly forms a solid, partially clear colloid that can be used in clear drinks, oral serums, and emulsion-based products. The powder is very resistant to mechanical stress and can keep the purity of the liposomes even when high-pressure tableting, pill filling, and blending are done. These are important skills for making drugs on a large scale. Concerns about soy allergens are taken care of by the non-GMO sunflower phospholipid binder, which also helps with clean label positioning. This ingredient has a shelf life of 24 months at room temperature and a bulk density of 600 g/L (or 918 g/L tapped), which makes it easier to handle inventory and lowers the cost of cold storage liposomal coenzyme Q10 compared to liquid options.
Practical Considerations for B2B Buyers
Regulatory Compliance and Certification Standards
There are a lot of rules that industrial buyers have to follow in order to get into the market. Third-party proof is how suppliers show they follow international quality standards. Hongda Phytochemistry has many different kinds of certificates, such as cGMP, FSSC 22000, ISO 9001, ISO 22000, HALAL, KOSHER, ORGANIC (EU), ORGANIC (NOP), and VEGAN. Each batch goes through strict testing procedures that check its identity (using HPLC, UV, and GC), check for heavy metals (using ICP-MS), look for pesticide residues (using multi-residue methods that cover more than 500 chemicals), and check for microbial safety. Every package comes with a full Certificate of Analysis (COA) that shows the product's purity, strength, and contaminant levels in comparison to USP and EP standards. The factory has 100,000-grade purification rooms with automatic tracking systems that make sure the quality of each production run is the same. Strategic relationships with approved third-party labs (SGS, Eurofins) give clients liposomal coenzyme Q10 who need more due investigation the chance to get independent verification.

Supplier Evaluation Criteria
If you want to find a good liposomal coenzyme Q10 powder provider, you need to look at more than just price. The manufacturing capacity decides how reliable a supply is. Hongda's 20,000-square-meter building has more than 10 modern production lines that produce more than 3,000 tons of goods every year, so even for large contracts, the goods are always available. The level of technical knowledge is also very important. Hongda hires more than 20 PhD-level researchers and works closely with university labs to create unique formulations and help with problems. Traceability starts at the source. The company has three specific growing bases for plant raw materials, using sustainable farming methods and checking the quality of the crops before they are harvested. The minimum order quantity (MOQ) is as low as 1 kg, which makes it possible for both test projects and large-scale production. Different practical needs can be met by flexible packing options like large drums, sample bags, and custom-branded solutions. OEM/ODM capabilities let buyers create their own recipes with the help of Hongda's production infrastructure.

Why Liposomal Coenzyme Q10 Powder Is the Preferred Solution
Clinical Evidence Supporting Superior Performance
Liposomal administration is preferred because it has been shown to have practical benefits. According to studies published in nutritional biochemistry journals, liposomal coenzyme Q10 powder has much higher plasma amounts and longer-lasting blood levels than other forms. These higher bioavailability measures lead to measured physiological effects, such as better free radical scavenging, higher mitochondrial ATP synthesis in heart and brain tissues, and better clinical results in cardiovascular health tests. Studies looking at fertility aids have found that liposomal ubiquinol delivery improves mitochondrial function in reproductive cells more effectively than regular supplements. Liposomal formats have been shown to lower oxidative stress markers and speed up muscle recovery times in sports performance trials. As more researchers realize that delivery technology, not just the identity of the active ingredient, affects clinical success, the body's collection of evidence keeps growing.
Market Positioning and Consumer Demand Trends
The market changes because people are more aware of absorption. Smart buyers are looking for signs that say "liposomal" more and more, because they think that technology like this means high quality and treatment effectiveness. This trend gives brands that adopt new transport systems early a chance to stand out from the competition. Manufacturers who use liposomal coenzyme Q10 powder can charge more while backing up their claims with science, which appeals to health-conscious customers who are willing to spend more for better absorption. The powder format is perfect for meeting the needs of new markets that want clean-label products (without synthetic emulsifiers), vegan-friendly formulas (with plant-based phospholipids), and products that can be used in a variety of formats (tablets, capsules, drinks, and topical uses). The global CoQ10 market is expected to grow, especially in the areas of cardiovascular health, anti-aging skin care, and sports nutrition. In all of these areas, absorption has a direct effect on how satisfied customers are with the liposomal coenzyme Q12 powder supplier and how likely they are to buy again.
Making an Informed Purchase DecisionEvaluating Product Specifications and Quality Indicators
When looking at quotes from suppliers, pay close attention to the technical details. Ask for particle size distribution data (including polydispersity index) to make sure that the structure of the liposomes is correct and not just a mix of lecithin and CoQ10. Demand enclosure efficiency measures that have been checked by a third party. Check the COA papers to see if they include heavy metals (like lead, arsenic, mercury, and cadmium), pesticide residues (multi-class screening), and microbial factors (like total plate count, coliforms, and pathogens). Look at stable data that shows how long something will last in a certain type of storage. Hongda gives full scientific information, such as TEM images that show intact vesicular structures, DLS reports that prove the size of nanoscale particles, and accelerated stability studies that show the materials will work at room temperature for 24 months. Suppliers who are open about how they test and are willing to give third parties reference samples to check show that they are confident in the quality and stability of the product.
Strategic Sourcing and Partnership Development
Moving to liposomal coenzyme Q10 powder requires more than just switching out ingredients; it also needs smart teamwork between suppliers. Start working together by asking for samples to be tested for stability in your specific formulation matrix. Do test batches to see how well they work with manufacturing, how they taste, and how stable the final product is. Talk about the expert support that is available, such as help with formulating, transferring analytical methods, and writing regulatory documents. Talk about minimum pledges, price tiers for increasing volume, wait times, and quality guarantee clauses in supply deals. Think about logistics based on location: Hongda's factory in Shaanxi, China, has low production costs, and the company has worked with customers in the US, Europe, and Asia before, so it knows how to handle different trade regulations and processes. Building partnerships with providers who put money into research and development, keep output flexible, and show a dedication to quality improvement will give your products a long-term edge over the competition.
Conclusion
Liposomal Coenzyme Q10 powder is a scientifically proven improvement in the technology of nutritional ingredients that solves the basic absorption problems that stop people from taking regular CoQ10 supplements. When purchasing, professionals look at different providers; they should give more weight to those that have strict quality control, a wide range of certifications, and technical knowledge in liposomal makeup. Hongda Phytochemistry's pharmaceutical-grade product, which contains non-GMO sunflower phospholipids that are at least 70% pure, nanoscale particle engineering, and excellent production stability, gives supplement companies the tools they need to make high-quality, effective goods. Liposomal delivery is the smart choice for brands that want to stand out in the crowded health and wellness markets because it combines clinical evidence, market demand trends, and formulation freedom.
FAQ1. What dosage of liposomal CoQ10 should be used in finished supplement formulations?
Dosing is based on the health claims that are being made and the method of delivery. Because liposomal types of CoQ10 are bioavailable 3–12 times better than regular CoQ10, they usually need 30–50% lower amounts to reach the same plasma concentrations. For general health purposes, finished products usually come in doses between 50 mg and 200 mg per serve. For therapeutic cardiovascular formulas, 200 mg to 400 mg daily may be split into two or three doses. Talk to a regulatory expert about what health claims are legal in your target market. Dosing guidelines change depending on the area and the purpose of the product.
2. How can buyers verify authentic liposomal structure versus simple powder mixtures?
Several scientific methods can be used to prove that liposomal powders are real. When the sample is reconstituted, the Dynamic Light Scattering (DLS) study should show particles in the 100–400nm range. Using Transmission Electron Microscopy (TEM) to take pictures shows that real liposomes have circular vesicular structures. The amount of CoQ10 that is actually trapped within lipid bilayers can be measured using encapsulation efficiency tests. Simple physical mixtures won't make nanospheres that are all the same size, and they won't mix well with water, and will separate clearly. As part of your approval process, ask providers for these analytical studies.
3. What certifications are essential when sourcing liposomal CoQ10 for the United States market?
Importers in the United States should make sure that their sources have cGMP certification, which shows that they follow FDA rules for production. It is important that chemicals have GRAS (Generally Recognized As Safe) status, that full COAs are provided to prove identification and purity, and that the product is tested by a third party for heavy metals, herbicides, and microbes. Organic certifications (USDA NOP, EU Organic) help products stand out as special, and HALAL and KOSHER approvals make more markets accessible. Make sure the seller can give you proof of the product's country of origin, information on how long it will last, and allergen statements to help with regulatory filing and customs clearing.
Partner With Hongda Phytochemistry for Premium Liposomal CoQ10 Solutions
Looking for the best liposome coenzyme Q10 supplier/liposomal coenzyme q10 powder manufacturer/liposomal coenzyme q10 supplier
Shaanxi Hongda Phytochemistry Co., Ltd. can help you come up with new products by making approved, pharmaceutical-grade liposomal coenzyme Q10 powder following strict cGMP guidelines. Our modern center always produces high-quality products and keeps full records of all the tests that were done. Also, our expert team can help you with formulation and offer customization services that are specific to your needs. Our low MOQ (starting at 1 kg) allows for both trial projects and large-scale production, whether you're making functional drinks, anti-aging formulas, sports nutrition items, or supplements for the heart. To get full product specs, COA paperwork, and reference samples, email our procurement experts at duke@hongdaherb.com. Find out why top makers in Asia, Europe, North America, and the United States choose Hongda as their reliable liposomal coenzyme Q10 powder supplier: this is where cutting-edge science meets top-notch manufacturing.
References
1. Bhagavan, H.N., & Chopra, R.K. (2007). Plasma coenzyme Q10 response to oral ingestion of coenzyme Q10 formulations. Mitochondrion, examining bioavailability differences across delivery systems and dosing protocols in clinical populations.
2. López-Lluch, G., Del Pozo-Cruz, J., Sánchez-Cuesta, A., Cortés-Rodríguez, A.B., & Navas, P. (2010). Bioavailability of coenzyme Q10 supplements depends on carrier lipids and solubilization. Nutrition, analyzing absorption enhancement through liposomal encapsulation versus oil-based preparations.
3. Sharma, A., & Sharma, U.S. (1997). Liposomes in drug delivery: Progress and limitations. International Journal of Pharmaceutics, detailing phospholipid bilayer structure, encapsulation mechanisms, and stability considerations in dried liposomal formulations.
4. Evans, M., Baisley, J., Barss, S., & Guthrie, N. (2009). A randomized, double-blind trial on the bioavailability of two CoQ10 formulations. Journal of Functional Foods, comparing plasma concentration curves between standard crystalline and enhanced-delivery CoQ10 products.
5. Nukala, R.K., Boyapati, R., & Slivaraman, N. (2012). Nanotechnology and liposomal delivery systems. Journal of Advanced Pharmaceutical Technology & Research, examining pharmaceutical applications of liposomal encapsulation for poorly water-soluble nutrients.
6. Argyriou, A.A., Chroni, E., & Koutras, A. (2010). Preventing paclitaxel-induced peripheral neuropathy: A phase II trial of vitamin E supplementation. Journal of Pain and Symptom Management discusses antioxidant supplementation strategies where bioavailability significantly impacts clinical outcomes.


_1745918362135.webp)






