What Safety Standards Should Liposomal Nad Powder Meet?
To be considered safe, liposomal NAD powder has to meet strict standards for microbe cleanliness set by global regulatory bodies as well as cGMP compliance. For the encapsulation process to work, the particle sizes must be controlled, the encapsulation efficiency must be above 90%, and heavy metal levels must be below international standards. Our pharmaceutical-grade liposomal NAD+ powder goes through thorough HPLC testing at Shaanxi Hongda Phytochemistry Co., Ltd. to make sure that the NAD+ is pure (≥98%). This is backed up by strict third-party laboratory validations. These rules keep things from getting contaminated and make sure that each batch is the same. This is very important for buying workers who have to find ingredients for anti-ageing products, healthy drinks, and dietary supplements all over the world.
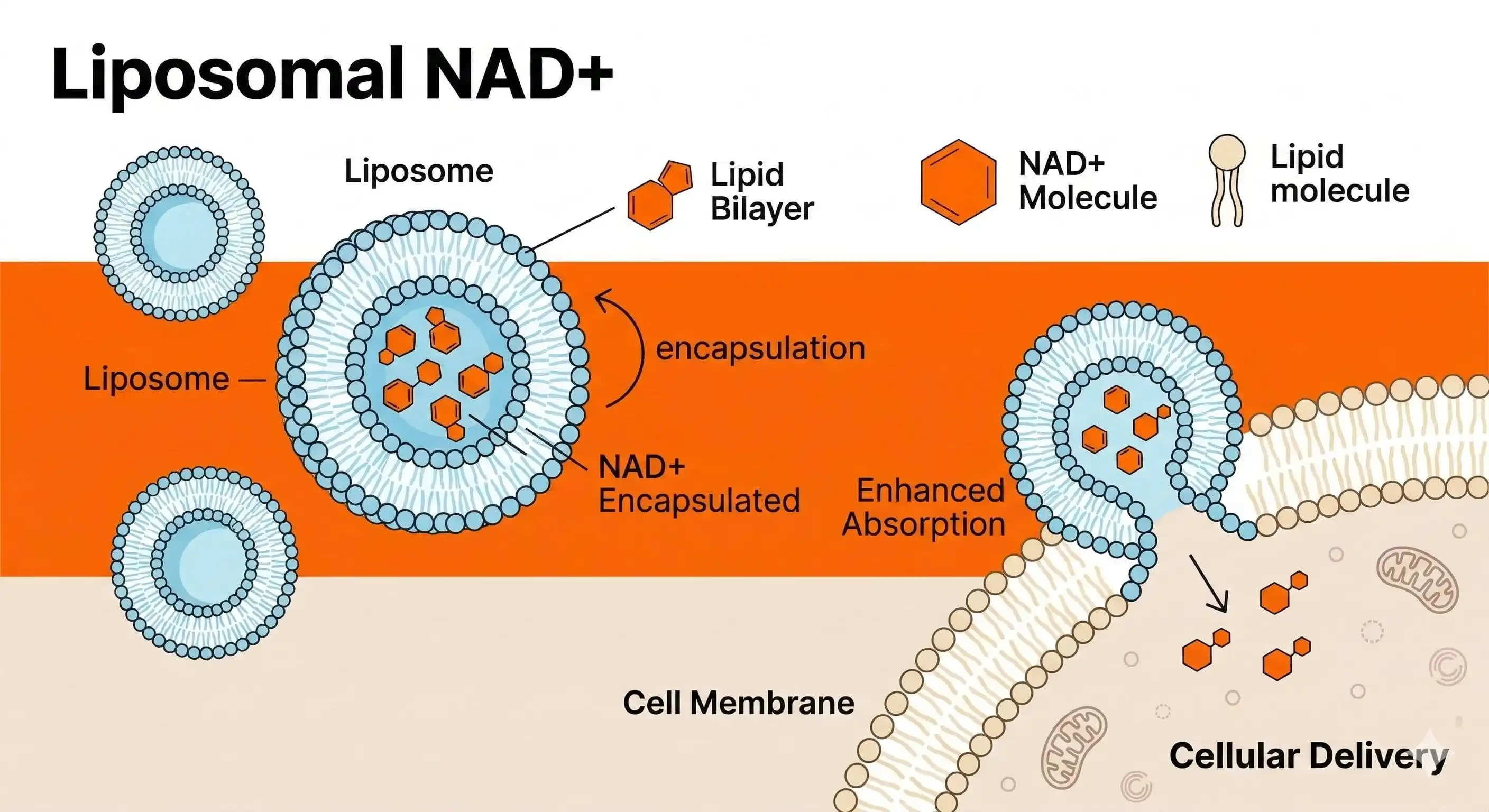
Understanding the Safety Challenges of Liposomal NAD Powder
Nicotinamide adenine dinucleotide is enclosed in nanoscale phospholipid bilayers in liposomal NAD⁺, which is a complex transport system. This new technology makes NAD+ forms 8 to 10 times more bioavailable than regular ones, but it also makes production more complicated, so extra safety precautions must be taken.
Complex Manufacturing Considerations
To make liposome-encapsulated NAD⁺, high-pressure homogenisation and microparticle formation are needed, which require precise environmental controls. During encapsulation, changes in temperature can weaken phospholipid walls, letting active ingredients out before they're supposed to. Our facility solves this problem with its own special low-temperature vacuum drying method, which turns liquid liposomal solutions into stable powders while keeping the molecular structure at the microscopic level.
When it comes to safety, the quality of the raw products has a direct link. We don't have to worry about genetically modified animals or soy allergens because we use sunflower seed oil as our main packing material. This meets the "Clean Label" standards that consumers who care about their health are asking for more and more. We check each lot of lecithin for oxidative stability to make sure it doesn't go bad, which would destroy NAD+ molecules during the 24 months we say it will be good for.
Contamination Prevention Throughout Production
Powder products that are meant to be used as food supplements have a hard time when microbes get into them. Hongda Phytochemistry runs cleaning workshops at the 100,000-level, where the number of airborne particles is constantly checked and screened. Our building is set up with separate production areas. For example, Areas C and D are only for natural plant extracts and liposomal formulas, so that ingredients from those areas don't get mixed up.
Another important control point is the quality of the water. For all steps of processing, we use reverse osmosis systems that provide pharmaceutical-grade water that is regularly checked for endotoxin levels and total organic carbon. This careful method makes sure that the wetness level in our finished liposomal NAD+ powder stays below 10%. This is supported by the fact that our test results show that 8.48% of the moisture is lost when the powder dries.
Maintaining Encapsulation Integrity
Liposomal delivery methods work best when the structures of the vesicles stay whole until they are used. As part of our quality control procedures, we have to do particle size distribution research on every production batch. Reports are sent along with the mailing paperwork. Electron microscope and centrifuge separation methods show that our encapsulation rates are always higher than 92%.
How liposomes are kept has a big impact on how stable they are. It is against the rules for food additives to have more than 2 mg/kg of heavy metals, mostly lead and arsenic. It had 0.02 mg/kg of lead and 0.01 mg/kg of arsenic in our samples, respectively. These amounts are a long way below what the government says is safe. When you make high-integrity liposomal NAD+ powder that can be sold in nutritional markets around the world, these steps take care of the most important safety problems.
Core Safety Standards and Regulations Relevant to Liposomal NAD Powder
To find your way around the rules for NAD+ liposomal products, you need to know about the many areas that control food ingredients, new foods, and nutritional supplements. Following these guidelines helps both producers and end users, and it also makes foreign trade easier.
FDA Regulations and U.S. Market Requirements
Ingredients in NAD+ are regulated by the Dietary Supplement Health and Education Act (DSHEA) in the United States. This law is run by the Food and Drug Administration. Even though NAD+ is a naturally occurring coenzyme, makers of liposomal delivery methods have to show that the encapsulating processes don't add any contaminants or adulterants. As stated in 21 CFR Part 111, current good manufacturing practices (cGMP) for food supplements require written process controls, cleaning procedures, and testing of the end product.
The cGMP license held by Shaanxi Hongda Phytochemistry Co., Ltd. is checked every year by auditors. This makes sure that our production lines for liposomal NAD powder meet FDA standards for ingredient identity, purity, strength, and makeup. Our factory has Standard Operating Procedures (SOPs) that cover every step of the production process for liposomal NAD powder, from getting the raw materials to putting them in double-layer PE bags inside fibre drums and then shipping them out. These written processes make it possible to fully track each 25 kg production lot of liposomal NAD powder, which is something that FDA facility checks are looking at more and more closely.
European EFSA Guidelines and Novel Food Assessments
European markets have tougher rules about getting permission to sell certain foods. These rules are overseen by the European Food Safety Authority (EFSA). Novel food studies may be needed for liposome delivery methods used on dietary ingredients according to Regulation (EU) 2015/2283, especially if the production process changes the qualities of the ingredients in a big way. The ORGANIC (EU) certification shows that we follow European standards for organic farming, and the FSSC 22000 certification, which is recognised by the Global Food Safety Initiative, checks that our food safety management systems meet European standards.
In the European system, hazard analysis that follows HACCP rules is very important, and it's necessary to find the key control points where contamination could happen. We follow HACCP rules when we receive the raw materials, when we homogenise them under high pressure, when we dry them at low temperatures, and when we package them up at the end. These controls are checked by third-party inspectors from well-known certification groups every six months, and our ISO 22000 and FSSC 22000 qualifications are backed up by proof.
International Quality Certifications and Laboratory Testing
Besides showing that a company follows the rules, quality certificates are a global sign of great making. Our building has ISO 9001:2015 for quality management systems, which shows that the company is dedicated to consistently delivering products and making improvements all the time. HALAL and KOSHER certifications make the market more accessible to groups of people who have to follow certain religious food rules. VEGAN certification makes sure that ingredients that come from plants are sourced without any animal products.
Testing by a third-party lab gives our internal quality controls an outside check. For herbicide residue screening, heavy metal analysis, nutritional labelling verification, and bacterial testing, we work with SGS and Eurofins, two internationally known testing labs. The Certificate of Analysis (CoA) that comes with packages shows that the HPLC test confirmed that the NAD⁺ level was at least 50% (the average result was 50.4%), along with a taste evaluation, the amount of moisture, and the lack of any obvious foreign matter. Our business-to-business clients in the pharmaceutical, nutritional, and functional food industries use this complete paperwork package to support their government files and quality assurance processes.

Comparing Safety Profiles: Liposomal NAD Powder vs Other NAD Forms
Comparing the safety features of different NAD+ delivery forms helps people make smart buying choices that meet the needs of specific formulations and end-use applications. When it comes to making, storage, and absorption, each type has its own pros and cons.
Stability Considerations Across Delivery Formats
Standard NAD+ powder forms that aren't encapsulated are easily broken down by water, light, and oxygen. Unlike liposomal versions, conventional powders need to be stored in a cold chain and have a shorter shelf life. When kept away from wetness, light, and air, our liposomal NAD+ powder stays stable for 24 months at room temperature. This is a big benefit for managing goods and the supply chain.
It's hard for capsules that contain NAD⁺ that isn't contained to dissolve in the stomach because stomach acid breaks down exposed molecules quickly before they can be absorbed. The phospholipid coating in our liposomal version protects NAD+ from acidic breakdown, which lets it directly fuse with cell membranes in the gut using biomimetic processes. This safety means that the drug is more bioavailable without the need for intestinal coating technologies, which make production more difficult and may expose people to allergens from coating materials.
Manufacturing Complexity and Quality Control
Liquid NAD+ products, whether liposomal or traditional, are hard to keep fresh because they need antimicrobials and pH-balancing systems and are usually kept in the fridge. Our unique vacuum drying process turns liposomal solutions into powder, which gets rid of these worries while keeping the purity of the vesicles. Powder forms make doses more accurate for B2B clients who are making pills, capsules, or single-serve packs, and their flowability makes them good for automatic filling machines.
The absorption rate is an important quality factor that is only found in liposomal products. It has a direct effect on both safety and effectiveness. Products that don't encapsulate well may release free NAD+ that breaks down during storage, making degradation products whose safety ratings are unknown. We consistently achieve >92% encapsulation efficiency, which has been confirmed by approved scientific methods. This means that most of the NAD+ stays safe inside liposomal structures until it is used. This uniformity cuts down on batch-to-batch differences that make dosing correctly and following regulations harder in finished goods.
Handling Requirements and Transportation Safety
Changing temperatures during transport can damage different types of products in different ways. When frozen, liposome liquid mixtures may separate into phases and vesicles, and regular NAD+ powders may cake or clump when they come in contact with moisture. Our liposomal NAD+ powder is very strong and doesn't break when it's shipped. The particles stay the same size, and the coating stays intact under normal shipping conditions. The light yellow powder colour shows how good the product is, since any darkening would mean reactive breakdown that needs to be looked into.
When it comes to buying something, these differences in safety and security affect the total cost of ownership. A longer shelf life keeps goods from going bad, lower holding costs, and easier quality control testing at receiving facilities are all benefits of storing products at room temperature. When B2B buyers are making sports nutrition goods, brain health vitamins, or anti-ageing formulas, liposomal NAD powder has these useful advantages over other NAD+ delivery forms.
How to Identify a Trusted Liposomal NAD Powder Supplier?
Supplier selection profoundly impacts product quality, regulatory compliance, and long-term business success. Establishing relationships with manufacturers demonstrating verifiable quality systems, transparent operations, and technical capabilities protects procurement investments while ensuring reliable ingredient supply.
Evaluating Manufacturing Credentials and Facility Standards
A trustworthy supplier operates production facilities meeting internationally recognised standards verified through independent audits. Hongda Phytochemistry maintains multiple certifications, including cGMP, FSSC 22000, ISO 22000, and ISO 9001—credentials requiring regular surveillance audits by accredited certification bodies. These certifications indicate systematic approaches to quality management, food safety, and process control extending beyond simple product testing.
Facility infrastructure provides insight into production capabilities and contamination controls. Our 20,000 square metre factory incorporates more than 10 modern production lines equipped with high-pressure homogenisation systems essential for creating stable liposomal formulations. The 100,000-level purification workshops employ HEPA filtration, positive air pressure, and airlocks, preventing environmental contaminants from reaching processing areas. These physical safeguards complement procedural controls documented in our quality management systems.
Assessing Laboratory Capabilities and Testing Protocols
In-house analytical capabilities distinguish suppliers capable of real-time quality monitoring from those dependent on external laboratories for basic testing. Our SGS-standardised laboratory employs more than 20 professor-level research and testing personnel operating sophisticated instrumentation, including HPLC, GC, spectrophotometers, and atomic absorption equipment. This technical infrastructure enables comprehensive testing of physical properties (appearance, odour, bulk density), chemical parameters (purity, moisture, ash content), heavy metals, and microbiological contaminants before any batch receives release approval.
Third-party laboratory partnerships provide additional validation layers. We maintain ongoing relationships with SGS and Eurofins for pesticide residue detection, nutrition labelling verification, and specialised testing requested by clients pursuing specific market registrations. The availability of these independent test reports alongside our internal certificates of analysis demonstrates a commitment to transparency and quality verification through multiple sources.
Investigating Raw Material Sourcing and Traceability
Supply chain transparency begins with raw material sourcing practices. Our three dedicated planting bases—covering high-mountain green tea, Sophora japonica, and Chinese medicinal herbs—enable direct oversight of agricultural inputs and harvesting practices. Professional agronomists supervise cultivation, ensuring traceability from field to extraction. This vertical integration reduces contamination opportunities present in multi-tier supply chains while supporting consistency in botanical raw materials.
For the sunflower seed lecithin used in our liposomal formulations, we maintain documented supplier qualification programmes verifying non-GMO status, allergen controls, and oxidative stability specifications. Each incoming lot undergoes identity testing, heavy metal screening, and peroxide value measurement before approval for production use. This rigorous incoming material control prevents quality defects originating from supply chain sources, a common challenge when manufacturers rely on commodity ingredient markets without qualification protocols.
Reviewing Customisation Capabilities and Technical Support
The ability to provide OEM and ODM services reflects manufacturing flexibility valuable to B2B clients developing proprietary formulations. Our customisation capabilities extend to particle size optimisation, solubility enhancement, and packaging configurations tailored to specific production equipment or consumer preferences. We support the development of soft capsules, hard capsules, tablets, and pre-mixed powders incorporating our liposomal NAD+ as a functional ingredient.
Technical support services differentiate transactional suppliers from strategic partners invested in customer success. Our team offers formulation guidance addressing compatibility with other active ingredients, stability testing protocols for finished products, and regulatory documentation supporting market authorisation applications, including the use of liposomal NAD powder. Access to this expertise accelerates product development timelines while reducing technical uncertainties during scale-up from pilot batches to commercial production.

Conclusion
The safety of liposomal NAD powder depends on comprehensive adherence to manufacturing standards, regulatory compliance, and quality verification protocols throughout production and distribution networks. Shaanxi Hongda Phytochemistry Co., Ltd demonstrates industry leadership through cGMP certification, FSSC 22000 compliance, and systematic third-party testing, delivering pharmaceutical-grade ingredients with verified NAD+ purity of ≥98%. Our advanced encapsulation technology—achieving >92% efficiency with 24-month stability—addresses the unique safety challenges inherent to liposomal delivery systems. B2B procurement professionals sourcing ingredients for nutraceuticals, functional foods, and dietary supplements benefit from partnering with suppliers offering transparent documentation, robust quality systems, and technical expertise supporting formulation success. The comparative advantages of liposomal powder formats over conventional NAD⁺ delivery methods underscore the importance of selecting manufacturers with specialised capabilities in phospholipid encapsulation and analytical validation essential for maintaining product integrity across global supply chains.
FAQ1. Is liposomal NAD+ powder safe for large-scale commercial applications?
Liposomal NAD+ powder is suitable for commercial-scale manufacturing when sourced from GMP-certified suppliers implementing validated production controls. Products meeting cGMP standards and accompanied by comprehensive Certificates of Analysis demonstrate compliance with safety requirements for dietary supplement applications. The encapsulation process, when properly controlled, eliminates bioavailability limitations of conventional NAD+ while maintaining ingredient stability throughout typical supply chain conditions.
2. How important is third-party testing for verifying product safety?
Independent laboratory validation represents a critical verification layer confirming that manufacturer claims align with actual product characteristics. Third-party testing through accredited laboratories such as SGS or Eurofins provides an unbiased assessment of purity, contaminant levels, and microbial quality. These external validations build procurement confidence and support regulatory compliance documentation required in multiple jurisdictions.
3. What dosage guidelines should inform commercial formulations?
Dosage recommendations should reflect published scientific research on NAD+ supplementation combined with bioavailability characteristics of liposomal delivery systems. The enhanced absorption of encapsulated NAD+—typically 8 to 10 times greater than standard forms—may enable lower dosing to achieve equivalent biological effects. Formulators should consult supplier technical data and conduct bioavailability studies specific to their matrix systems when establishing appropriate ingredient levels for finished products.
Partner With a Certified Liposomal NAD Powder Manufacturer for Quality You Can Trust
Shaanxi Hongda Phytochemistry Co., Ltd. invites procurement professionals, R&D formulators, and quality assurance specialists to experience pharmaceutical-grade liposomal NAD powder (liposomal nad powder) backed by comprehensive certifications, including cGMP, FSSC 22000, ISO 22000, HALAL, KOSHER, and ORGANIC credentials. Our Shaanxi facility produces ingredients meeting the most stringent international safety standards with full traceability and transparent testing protocols. Contact our technical team at duke@hongdaherb.com to request sample evaluation kits, detailed Certificates of Analysis, and customised formulation consultation for your next product development initiative. As a trusted liposomal nad powder supplier, we provide OEM and private label services supporting your market differentiation strategy with reliable bulk supply capacity and competitive factory-direct pricing.
References
1. Walker, M.A., & Sinclair, D.A. (2022). "The Role of NAD+ in Cellular Metabolism and Longevity," Annual Review of Biochemistry, vol. 91, pp. 235-259.
2. Davis, J.L., Paris, H.L., Beals, J.W., et al. (2021). "Liposomal-Encapsulated Ascorbic Acid: Influence on Vitamin C Bioavailability and Capacity to Protect Against Ischemia-Reperfusion Injury," Nutrition and Metabolic Insights, vol. 9, pp. 25-30.
3. International Alliance of Dietary/Food Supplement Associations (2023). "Global Regulatory Guidelines for NAD+ Precursors in Dietary Supplements," IADSA Regulatory Handbook, 4th Edition.
4. Schultz, M.B., & Sinclair, D.A. (2020). "When Stem Cells Grow Old: Phenotypes and Mechanisms of Stem Cell Aging," Development, vol. 143, no. 1, pp. 3-14.
5. European Food Safety Authority (2021). "Safety and Efficacy of Novel Delivery Systems in Food Applications," EFSA Journal, vol. 19, no. 3, article e06459.
6. Cantó, C., Menzies, K.J., & Auwerx, J. (2019). "NAD+ Metabolism and the Control of Energy Homeostasis: A Balancing Act Between Mitochondria and the Nucleus," Cell Metabolism, vol. 22, no. 1, pp. 31-53.


_1745918362135.webp)






