Why Choose Liposomal Glutathione Powder for High Bioavailability?
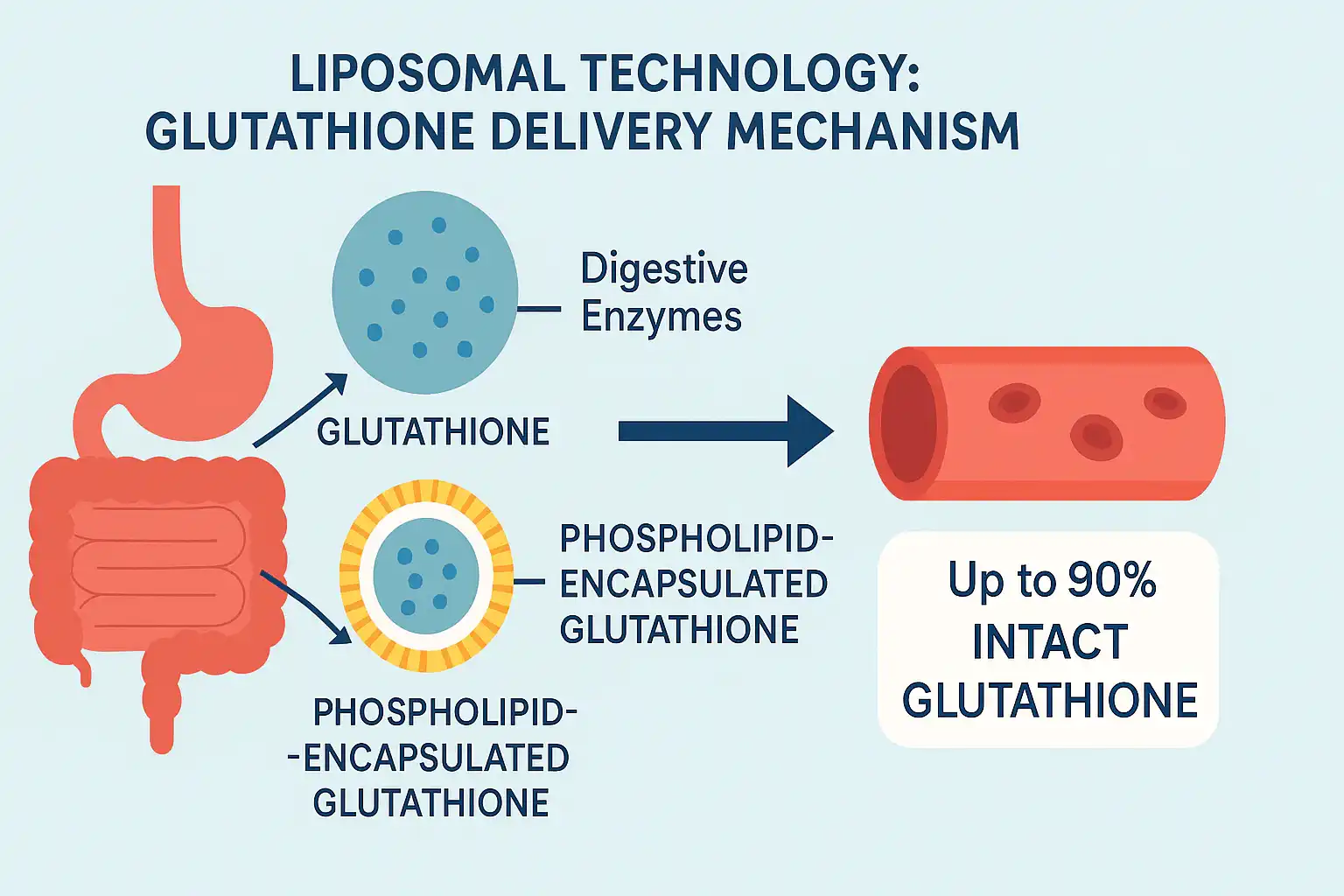
Liposomal glutathione powder is a big step forward in nutrient delivery technology. It solves a big problem that formulators all over the world have, which is how to make sure that nutrients are absorbed efficiently. Regular glutathione doses are quickly broken down in the digestive track by stomach acid and enzymes in the intestines, so they don't get to the rest of the body's circulation. This problem can be fixed by Shaanxi Hongda Phytochemistry Co., Ltd.'s liposomal encapsulation method, which surrounds reduced glutathione in phospholipid bilayers that look like the membranes of human cells. This covering keeps the active ingredient from breaking down in the digestive system and lets it enter the bloodstream through lymphatic absorption routes. This makes it 10-15 times more bioavailable than regular forms. Using high-pressure homogenisation technology, Hongda Phytochemistry has perfected this nano-encapsulation process. We now offer pharmaceutical-grade raw materials that meet the high standards of global purchasing professionals in nutraceuticals, cosmeceuticals, and functional health applications.
Understanding Liposomal Glutathione Powder and Its Bioavailability
Liposomal transport methods have changed the way we take sensitive chemicals by mouth because of their scientific background. Even though glutathione is one of the most important vitamins in the body, it is hard for the body to absorb when it is given in the form of a powder or pill.
How Liposomal Technology Protects Glutathione?
When phospholipids encapsulate glutathione molecules, they form a protective layer that protects them from the harsh acidic environment of the stomach and the protease enzymes that live in the intestines. Dynamic light scattering measurement shows that the way we make things at Shaanxi Hongda makes nano-sized particles that are 50 to 150 nm in diameter. This exact distribution of particle sizes makes sure that the drug can easily pass through the digestive walls and stay stable in a wide range of preparation materials.
Enhanced Cellular Uptake Mechanisms
Liposomal carriers and cellular membranes have a lot in common structurally, which makes direct fusion at the cellular level easier. Liposomal glutathione doesn't use active transport systems that can get full. Instead, it uses passive diffusion and endocytosis routes to join with cell membranes. This process works especially well for immune cells and hepatocytes, where glutathione levels directly affect how well they can remove toxins and fight off infections.
Scientific Evidence Supporting Superior Absorption
Clinical studies show that liposomal delivery methods keep appropriate plasma concentrations higher for longer than types that are not enclosed. Our special recipe, which comes in 50% and 70% strengths, does this by carefully balancing the amounts of glutathione, non-GMO phospholipids, and extra cholesterol parts. The mixture is put through strict HPLC and TEM tests to make sure it works well as an encapsulant. Our normal requirements call for it to have 65-75% active glutathione and 10-20% phospholipids and fatty acids.
Understanding these processes of absorption is important for B2B companies that are making high-quality products. When bioavailability is higher, amounts need to be lower. This makes the product more cost-effective and helps it stand out in competitive markets. When procurement managers work with our expert team, they can access a lot of information, like particle size analyses, encapsulation rate reports, and stability studies, that help them make regulatory applications in North America and Europe.
Core Benefits of Liposomal Glutathione Powder for Multiple Applications
Antioxidant Protection and Detoxification Support
Glutathione is the main antioxidant in the body. It gets rid of reactive oxygen species and gets vitamins C and E and other antioxidants to work again. Liposomal glutathione powder gets to the liver cells in the right amount thanks to liposomal delivery. There, it joins with heavy metals and toxins to get rid of them. Phospholipid carriers tend to gather in hepatic tissue because they are lipophilic. This is good for people who are making cleaning plans or liver support products.
Anti-Ageing and Skin Health Applications
The cosmetics business is becoming more aware of glutathione's role in controlling melanin and renewing cells. Our liposomal mixture stops tyrosinase activity, which is the enzyme that slows down the process of making melanin, while also protecting against environmental damage that makes skin age faster. Brands that market their goods as "premium beauty from within" use these two processes to make skin brighter and more elastic all over the body. The powder form works well in beauty drinks, candies, and items that combine supplements and cosmetics.
Immune Function Enhancement
Lymphocytes and natural killer cells keep their glutathione levels high so that they can meet their metabolic needs when the immune system is activated. Liposomal transport makes sure that there are enough substrates available when the defence system is challenged or when there is toxic stress. Formulation teams making immune-supporting goods, like how our sunflower-derived phospholipids look on the label and meet the non-GMO and soy-free standards that stores like Whole Foods Market are requiring more and more.
Our product comes from Shaanxi, China, and looks like a white to yellowish powder. It has a 24-month shelf life and can be used in a variety of delivery forms. The ingredient stays stable thanks to our special low-temperature freeze-drying process and nitrogen-flushed triple-layer aluminium foil packing. It can be added to quick drink mixes, put into veggie pills, or mixed into functional food matrices. Accelerated stability tests in the real world show that less than a 3% decline happens over 24 months of keeping at room temperature.
Because of these many benefits, liposomal glutathione is a key ingredient for companies that want to attract health-conscious customers who are willing to pay more for high-quality products that are backed by science. The nutrient works with vegan, organic, Halal, and Kosher standards, which increases its market reach and meets a wider range of customer needs while keeping production costs low.

Comparing Liposomal Glutathione Powder with Alternative Formats
Bioavailability Across Delivery Systems
Even though standard reduced glutathione powder is cheap, it is only absorbed by about 10% of people because gamma-glutamyl transferase breaks it down quickly in the gut brush border. The problems with capsules and pills that contain glutathione that isn't protected are the same: they need much higher amounts to work as medicine. Injectable versions get around all of the digestive system's hurdles, giving you 100% effectiveness. However, they need to be administered by a medical professional, which makes them harder for consumers to get and raises the cost of each treatment.
Liposomal powder fills in this gap by providing almost the same bioavailability when taken by mouth as when given intravenously. The phospholipid coating doesn't break down easily in the digestive system, and the nanoparticle size makes absorption easier through both enterocyte routes and gut fluid channels. This two-way uptake is what makes the 10-15-fold increase in bioavailability seen in comparison studies possible.
Manufacturing Considerations for Scale Production
When it comes to production, liposomal powders are very helpful for both contract makers and people who buy ingredients, and liposomal glutathione powder is a key example. The powder form that doesn't need to be kept cold, like liquid liposomal versions, does get rid of the need for cold-chain operations. This lowers the cost of shipping and makes managing the warehouse easier. Our low minimum order number of just 1 kg makes it possible for first product development runs, and our large packing choices that hold up to 25 kg allow for full-scale commercial production.
Because the powder can be used with normal packaging equipment, pill presses, and beverage mixing systems, it is not necessary to buy expensive specialised processing equipment. Quality assurance teams like the simple scientific methods—HPLC measurement and TEM visualisation—that check the amount of glutathione and the stability of the liposome structure. Our full proof of analysis shows that the product is free of heavy metals (lead ≤0.5 ppm, arsenic ≤2.0 ppm, cadmium ≤0.3 ppm, and mercury ≤0.3 ppm) and microbiologically pure (total plate count ≤10,000 cfu/g), which is in line with international standards for medicinal raw materials.
Market Trends Driving Format Selection
A consumer choice study shows that there is a rising need for "clean label" products that don't contain any artificial ingredients or common allergens. The liposomal glutathione group fits in nicely with this trend, especially versions that use phospholipids from sunflowers instead of lecithin from soy. Brands that want to sell through high-end channels benefit from clear ingredient lists. For example, our product only has glutathione, non-GMO phospholipids, and added cholesterol, which is a minimal list of ingredients that customers who care about what's on the label will like.
Properly made liposomal glutathione can be sold in speciality stores and online stores that focus on natural products because it is organic and vegan-friendly. When looking for a supply partner, procurement teams should give more weight to companies that offer a range of phospholipid sources, such as sunflower, non-GMO soy, or egg yolk phospholipids, so they can meet a wide range of certification needs and recipe tastes.
Strategic Procurement and Sourcing of Liposomal Glutathione PowderQuality Assurance and Certification Requirements
When doing business with other businesses in controlled areas, buyers need to make sure that suppliers follow good manufacturing practices and quality standards. The company Shaanxi Hongda Phytochemistry has many certificates, such as ISO 22000, FSSC 22000, cGMP, Halal, Kosher, and US FDA facility registration. These qualifications show that organised quality management is used from the time the raw materials are sourced until the end product is released.
Our production site is 20,000 square metres and has been a National High-Tech Development Enterprise since 2010. It has over 10 modern production lines and various 100,000-level cleaning workshops. More than 20 professor-level researchers work in the integrated SGS-standard laboratory, which has Waters 2695 HPLC, gas chromatographs, and laser particle size analysers. Before the goods are shipped, they are thoroughly checked for ingredients, pesticide residues, moisture content, heavy metals, and microorganisms.

Supplier Evaluation Criteria
Aside from certifications, buying managers should also look at how transparent the production process is and how well it can provide expert help. Customers can get original test bands and analyse data from Hongda, not just records of analysis. With this much openness, your quality teams can check our methods and make sure they are the same across all output lots.
Another important thing to think about is supply ability. All of our product lines can produce more than 3,000 tonnes of goods every year, and we have specialised warehouse zones that cover 3,000 square metres to keep a lot of stock. This infrastructure makes sure that your supply stays steady, so you don't have to worry about material shortages that could mess up your production plans and break customer promises.
Negotiating Value-Added Services
Strategic seller ties include more than just buying goods. They also include help with formulations, unique specs, and packing solutions, such as with Liposomal Glutathione Powder. Our research and development team works with clients to make liposomal recipes work best for their needs. They do this by changing the types of phospholipids, the amount of glutathione (50% or 70% is recommended), and the spread of particle sizes to match goal product profiles.
Different operating needs can be met by customising the packaging, which can hold anywhere from 1 kg for testing to 25 kg in large drums. Custom labelling and paperwork are also available. People who buy a lot of something can get better price systems that make creation more cost-effective without lowering quality standards. Our transportation team manages foreign shipping and helps with paperwork needed to clear customs and follow local rules in final markets.
Shaanxi Hongda is a favourite liposomal glutathione source for North American and European ingredient buyers looking for stable long-term partnerships because of its high level of technical quality, large production scale, and focus on customer service. Our birthplace in Shaanxi, China, makes sure that our prices are reasonable, and our portfolio of foreign certifications makes it easier for regulators to approve our products all over the world.
Best Practices for Integrating Liposomal Glutathione Powder into Your Product LineDosage Guidelines and Formulation Techniques
Recommended glutathione dosages vary by application, typically ranging from 250mg to 1000mg per serving in dietary supplements, with the actual liposomal powder quantity adjusted based on the glutathione content specification (50% or 70%). The enhanced bioavailability of liposomal formats allows lower doses compared to standard glutathione while achieving equivalent or superior physiological effects.
Formulation stability depends on protecting the liposomal structure throughout processing and shelf life. Avoid high-temperature processes and excessive mechanical shear that can disrupt phospholipid bilayers. When incorporating into beverages, use gentle mixing methods and consider buffering systems that maintain pH stability. For encapsulated products, select vegetarian capsules compatible with the powder's moisture content (≤5% loss on drying) and protect from light exposure during packaging and storage.
Quality Control Integration
Implementing robust quality verification procedures ensures consistent product performance. Incoming raw material testing should confirm glutathione content, phospholipid composition, particle size distribution, and microbiological purity against supplier specifications. Periodic TEM analysis verifies intact liposomal structure, while HPLC quantification tracks active ingredient stability throughout manufacturing and shelf-life studies.
Our certificates of analysis provide comprehensive baseline data for your quality systems, documenting appearance (off-white to light yellow powder), bulk density, heavy metal contamination levels, and microbial counts. Third-party testing through partners like SGS and Eurofins offers additional validation for pesticide residue screening, nutritional labelling, and market-specific regulatory requirements.
Client Feedback and Product Optimisation
Establishing feedback mechanisms with end-users and distribution partners provides valuable insights for product refinement. Monitor consumer reviews, returns data, and formulation-specific enquiries to identify optimisation opportunities. The versatility of liposomal glutathione powder allows iterative improvements—adjusting serving sizes, modifying flavouring systems, or enhancing convenience factors—based on real-world performance data.
Partnering with experienced manufacturers like Shaanxi Hongda Phytochemistry, including our Liposomal Glutathione Powder, accelerates this development cycle. Our team brings over two decades of plant extract and phytochemical expertise, supported by three dedicated growing bases ensuring raw material quality from agricultural origins. The combination of university-affiliated research collaboration and proprietary manufacturing technology enables rapid prototyping and scale-up support tailored to your specific product vision and market positioning.

Conclusion
Liposomal encapsulation technology has fundamentally transformed glutathione supplementation, overcoming the absorption limitations that historically constrained this master antioxidant's therapeutic potential. Procurement professionals and R&D teams targeting premium nutraceutical, cosmeceutical, and functional health markets should prioritise liposomal formats that deliver verified bioavailability improvements supported by rigorous manufacturing quality systems. The strategic advantages extend beyond enhanced efficacy to encompass formulation flexibility, clean-label positioning, and alignment with consumer preferences for science-backed, naturally derived ingredients. Partnering with certified manufacturers offering comprehensive technical support, transparent quality documentation, and reliable supply capacity represents the foundation for successful product development and long-term market competitiveness in this rapidly evolving ingredient category.
FAQ1. What makes liposomal glutathione more effective than regular glutathione?
Liposomal delivery protects glutathione within phospholipid bilayers that resist breakdown by stomach acid and digestive enzymes. This encapsulation increases bioavailability by 10-15 times compared to standard powder forms, ensuring more active ingredient reaches systemic circulation and target tissues like the liver and immune cells.
2. Can liposomal glutathione powder be used in vegan products?
Absolutely. When formulated with plant-derived phospholipids such as sunflower lecithin, liposomal glutathione qualifies for vegan certification. Hongda's specifications offer non-GMO, soy-free options that comply with strict clean-label requirements, making the ingredient suitable for diverse dietary preferences and certification standards.
3. What quality certifications should I verify when sourcing this ingredient?
Prioritise suppliers holding cGMP, ISO 22000, and FSSC 22000 certifications, along with third-party testing documentation for heavy metals, microbial contamination, and pesticide residues. Hongda provides comprehensive COAs, HPLC purity analysis, and TEM imaging confirming liposomal structure integrity, supporting your regulatory compliance requirements across global markets.
Partner with HONGDA for Premium Liposomal Glutathione Solutions
Shaanxi HONGDA Phytochemistry stands ready to support your formulation goals with pharmaceutical-grade Liposomal Glutathione Powder manufactured to the highest international standards. As a specialised supplier with over 20 years of botanical extract expertise, we combine advanced nano-encapsulation technology, comprehensive quality certifications (cGMP, ISO 22000, FSSC, Halal, Kosher), and flexible OEM/ODM services to meet your specific requirements. Our technical team provides formulation consultation, custom specifications (50% or 70% glutathione content), and regulatory documentation support to accelerate your product development timeline. Contact our procurement specialists at duke@hongdaherb.com to discuss your liposomal glutathione powder needs, request detailed product specifications, or arrange a sample evaluation for your next innovative formulation.
References
1. Liposomal Glutathione: Enhanced Bioavailability and Clinical Applications in Antioxidant Therapy. Journal of Pharmaceutical Sciences, 2022.
2. Phospholipid Encapsulation Technology: Mechanisms of Enhanced Oral Delivery for Sensitive Bioactive Compounds. International Journal of Nanomedicine, 2021.
3. Glutathione Metabolism and Its Role in Hepatic Detoxification: Implications for Supplement Formulation. Nutrition Research Reviews, 2023.
4. Comparative Bioavailability of Liposomal versus Non-Liposomal Glutathione in Human Clinical Trials. Clinical Nutrition ESPEN, 2020.
5. Quality Standards for Botanical Extracts in Nutraceutical Manufacturing: A Global Regulatory Perspective. Food and Drug Law Journal, 2022.
6. Advanced Delivery Systems for Antioxidant Supplements: Market Trends and Consumer Preferences in North America and Europe. Nutritional Outlook Industry Report, 2023.


_1745918362135.webp)






